Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
IMMUNOLOGY / RESEARCH PAPER
Amplification of Cord Blood-Derived Cytotoxic T Lymphocytes using HL-60 cell Derived Exosomes
1
Qilu Hospital of Shandong University, China
2
Yidu Central Hospital of Weifang, China
Submission date: 2020-03-17
Final revision date: 2020-05-25
Acceptance date: 2020-06-05
Online publication date: 2021-04-13
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Dendritic cell (DCs) based cytotoxic T lymphocytes (CTLs) are commonly used in immunotherapy due to their specificity. The selection of appropriate cell origin and tumor antigen is the key point. The objective was to culture DCs and CTLs simultaneously from cord blood, and deliver antigen information using tumor derived exosomes.
Material and methods:
Exosomes were collected from the human promyelocytic leukemia cell line HL-60 using ultracentrifugation. Prepared DCs from adherent cord blood mononuclear cells (MNCs) using SCF, GM-CSF, and IN-4. TNF-α and microRNA removed tumor-exosome were used to induce DCs maturation. DCs matured in the presence of HL-60 cell membrane protein extract or no antigen were set as control. CTLs was cultured from non-adherent MNCs by adding IFN-γ, IL-15, SCF, FLT-3L, anti-CD3, anti-CD28 and IL-2. The CTLs were analyzed by flow cytometry, cytotoxicity experiments and ELISA.
Results:
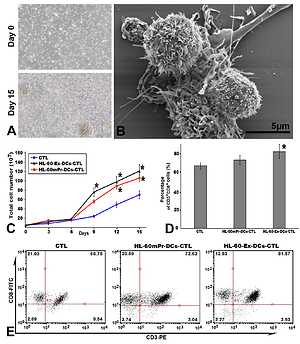
DCs can be obtained from cord blood and express costimulatory molecules. After 15 days, the total number of the cells expanded 26.3 times, and more than 82% of the cells expressed CD3+CD8+ in the most amplified HL-60-Ex-DCs-CTL group. These CD3+CD8+ T cells generated by HL-60-Ex-DCs displayed specific cytotoxicity towards HL-60 and low lethality towards unrelated BALL-1 cells. ELISA results showed that the expressions of TNF-α and IFN-γ in HL-60-Ex-DCs or HL-60mPr-DCs activated CTLs were upregulated compared with the control group.
Conclusions:
Cord blood CTLs generated by HL-60 derived exosome activated DCs displayed specific cytotoxicity towards HL-60 promyelocytic leukemia cells. Therefore cord blood and tumor derived exosomes provided a good source for adoptive immunotherapy.
Dendritic cell (DCs) based cytotoxic T lymphocytes (CTLs) are commonly used in immunotherapy due to their specificity. The selection of appropriate cell origin and tumor antigen is the key point. The objective was to culture DCs and CTLs simultaneously from cord blood, and deliver antigen information using tumor derived exosomes.
Material and methods:
Exosomes were collected from the human promyelocytic leukemia cell line HL-60 using ultracentrifugation. Prepared DCs from adherent cord blood mononuclear cells (MNCs) using SCF, GM-CSF, and IN-4. TNF-α and microRNA removed tumor-exosome were used to induce DCs maturation. DCs matured in the presence of HL-60 cell membrane protein extract or no antigen were set as control. CTLs was cultured from non-adherent MNCs by adding IFN-γ, IL-15, SCF, FLT-3L, anti-CD3, anti-CD28 and IL-2. The CTLs were analyzed by flow cytometry, cytotoxicity experiments and ELISA.
Results:
DCs can be obtained from cord blood and express costimulatory molecules. After 15 days, the total number of the cells expanded 26.3 times, and more than 82% of the cells expressed CD3+CD8+ in the most amplified HL-60-Ex-DCs-CTL group. These CD3+CD8+ T cells generated by HL-60-Ex-DCs displayed specific cytotoxicity towards HL-60 and low lethality towards unrelated BALL-1 cells. ELISA results showed that the expressions of TNF-α and IFN-γ in HL-60-Ex-DCs or HL-60mPr-DCs activated CTLs were upregulated compared with the control group.
Conclusions:
Cord blood CTLs generated by HL-60 derived exosome activated DCs displayed specific cytotoxicity towards HL-60 promyelocytic leukemia cells. Therefore cord blood and tumor derived exosomes provided a good source for adoptive immunotherapy.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.