Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
HEMATOLOGY / CLINICAL RESEARCH
Comparison of the efficacy of vindesine and leurocristine for pediatric acute lymphoblastic leukemia and their effects on quality of life
1
Department of Hematology, Yinzhou People’s Hospital, Ningbo City,
Zhejiang Province, China
Submission date: 2020-02-25
Final revision date: 2020-06-29
Acceptance date: 2020-07-20
Online publication date: 2021-04-18
Publication date: 2026-04-30
Corresponding author
Xiaowei Shi
Department of Hematology, Yinzhou People’s Hospital, No. 251 Baijiang East Road, Ningbo City 315040, Zhejiang Province, China, Tel.: 0574-87017565
Department of Hematology, Yinzhou People’s Hospital, No. 251 Baijiang East Road, Ningbo City 315040, Zhejiang Province, China, Tel.: 0574-87017565
Arch Med Sci 2026;22(2):815-821
KEYWORDS
vindesineleurocristinepediatric acute lymphoblastic leukemiaquality of lifeinterleukin 6tumour necrosis factor-α
TOPICS
ABSTRACT
Introduction:
The present study aimed to compare the efficacy of vindesine with that of leurocristine for pediatric acute lymphoblastic leukemia and their effects on the quality of life.
Material and methods:
We included 94 patients with pediatric acute lymphoblastic leukemia admitted to our hospital for treatment and categorized them into two groups: treatment with vindesine (group A, n = 48) and treatment with leurocristine (group B; n = 48). The effective rate, adverse reactions (thrombocytopenia, anemia, peripheral neurotoxic reaction, infection, nausea, and vomiting) and basic clinical conditions (medical expenses and length of hospital stay) of the patients were observed. In addition, interleukin 6 (IL-6) and tumour necrosis factor-alpha (TNF-α) expression levels were determined by ELISA before and after treatment, posttreatment quality of life score was determined by the Karnofsky Performance Scale (KPS), and the 5-year overall survival was determined in both groups.
Results:
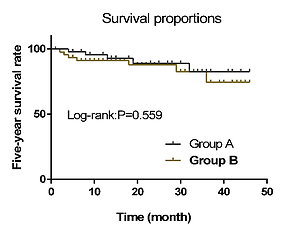
Group A showed no significant difference in the total efficacy rate and 5-year overall survival after treatment (p > 0.05) and had lower medical expenses, length of hospital stay, IL-6 and TNF-α expression, and total incidence of adverse reactions and higher KPS scores than group B (p < 0.05).
Conclusions:
Although no significant difference was observed between vindesine and leurocristine in treating pediatric acute lymphoblastic leukemia, patients administered vindesine had fewer adverse reactions, shorter length of hospital stay, lower medical expenses, and higher quality of life than those administered leurocristine, indicating a potential association with decreased serum IL-6 and TNF-α expression.
The present study aimed to compare the efficacy of vindesine with that of leurocristine for pediatric acute lymphoblastic leukemia and their effects on the quality of life.
Material and methods:
We included 94 patients with pediatric acute lymphoblastic leukemia admitted to our hospital for treatment and categorized them into two groups: treatment with vindesine (group A, n = 48) and treatment with leurocristine (group B; n = 48). The effective rate, adverse reactions (thrombocytopenia, anemia, peripheral neurotoxic reaction, infection, nausea, and vomiting) and basic clinical conditions (medical expenses and length of hospital stay) of the patients were observed. In addition, interleukin 6 (IL-6) and tumour necrosis factor-alpha (TNF-α) expression levels were determined by ELISA before and after treatment, posttreatment quality of life score was determined by the Karnofsky Performance Scale (KPS), and the 5-year overall survival was determined in both groups.
Results:
Group A showed no significant difference in the total efficacy rate and 5-year overall survival after treatment (p > 0.05) and had lower medical expenses, length of hospital stay, IL-6 and TNF-α expression, and total incidence of adverse reactions and higher KPS scores than group B (p < 0.05).
Conclusions:
Although no significant difference was observed between vindesine and leurocristine in treating pediatric acute lymphoblastic leukemia, patients administered vindesine had fewer adverse reactions, shorter length of hospital stay, lower medical expenses, and higher quality of life than those administered leurocristine, indicating a potential association with decreased serum IL-6 and TNF-α expression.
REFERENCES (40)
1.
Ross ME, Zhou X, Song G, et al. Classification of pediatric acute lymphoblastic leukemia by gene expression profiling. Blood 2003; 102: 2951-9.
2.
Nordlund J, Bäcklin CL, Wahlberg P, et al. Genome-wide signatures of differential DNA methylation in pediatric acute lymphoblastic leukemia. Genome Biol 2013; 14: r105.
3.
Gaipa G, Basso G, Biondi A, Campana D. Detection of minimal residual disease in pediatric acute lymphoblastic leukemia. Cytometry B Clin Cytom 2013; 84: 359-69.
4.
Ravindranath Y. Recent advances in pediatric acute lymphoblastic and myeloid leukemia. Curr Opin Oncol 2003; 15: 23-35.
5.
Aplenc R, Thompson J, Han P, et al. Methylenetetrahydrofolate reductase polymorphisms and therapy response in pediatric acute lymphoblastic leukemia. Cancer Res 2005; 65: 2482-7.
6.
Carroll WL, Bhojwani D, Min DJ, et al. Pediatric acute lymphoblastic leukemia. Hematology Am Soc Hematol Educ Program 2003; 102-31.
7.
Gralla RJ, Tan CT, Young CW. Vindesine a review of phase-II trials. Cancer Chemother Pharmacol 1979; 2: 271-4.
8.
Kelsen D, Bains M, Cvitkovic E, Golbey R. Vindesine in the treatment of esophageal carcinoma: a phase II study. Cancer Treat Rep 1979; 63: 2019-21.
9.
Kelsen D, Gralla R, Cheng E, Martini N. Vindesine in the treatment of malignant mesothelioma: a phase II study. Cancer Treat Rep 1983; 67: 821-2.
10.
Duan YT, Sangani ChB, Liu W, Soni KV, Yao Y. New promises to cure cancer and other genetic diseases/disorders: epi-drugs through epigenetics. Curr Top Med Chem 2019; 19: 972-94.
11.
Albert DM, Wong VG, Henderson ES. Ocular complications of vincristine therapy. Arch Ophthalmol 1967; 78: 709-13.
12.
Rosenthal S, Kaufman S. Vincristine neurotoxicity. Ann Intern Med 1974; 80: 733-7.
13.
Silverman JA, Deitcher SR. Marqibo® (vincristine sulfate liposome injection) improves the pharmacokinetics and pharmacodynamics of vincristine. Cancer Chemother Pharmacol 2013; 71: 555-64.
14.
Douer D. Efficacy and safety of vincristine sulfate liposome injection in the treatment of adult acute lymphocytic leukemia. Oncologist 2016; 21: 840-7.
15.
Mora E, Smith EML, Donohoe C, Hertz DL. Vincristine-induced peripheral neuropathy in pediatric cancer patients. Am J Cancer Res 2016; 6: 2416.
16.
Tay CG, Lee VWM, Ong LC, Goh KJ, Ariffin H, Fong CY. Vincristine-induced peripheral neuropathy in survivors of childhood acute lymphoblastic leukaemia. Pediatr Blood Cancer 2017; 64: e26471.
17.
Xu P, Jiang Y, Zuo H, et al. Vincristine-loaded platelets coated with anti-CD41 mAbs: a new macrophage targeting proposal for the treatment of immune thrombocytopenia. Biomater Sci 2019; 7: 4568-77.
18.
Vardiman JW, Thiele J, Arber DA, et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood 2009; 114: 937-51.
19.
Cui L, Li ZG, Chai YH, et al. Outcome of children with newly diagnosed acute lymphoblastic leukemia treated with CCLG-ALL 2008: the first nation-wide prospective multicenter study in China. Am J Hematol 2018; 93: 913-20.
20.
Terret C, Albrand G, Moncenix G, Droz JP. Karnofsky performance scale (KPS) or physical performance test (PPT)? That is the question. Crit Rev Oncol Hematol 2011; 77: 142-7.
21.
Gao RW, Dusenbery KE, Cao Q, Smith AR, Yuan J. Augmenting total body irradiation with a cranial boost before stem cell transplantation protects against post-transplant central nervous system relapse in acute lymphoblastic leukemia. Biol Blood Marrow Transplant 2018; 24: 501-6.
22.
Cooper SL, Brown PA. Treatment of pediatric acute lymphoblastic leukemia. Pediatr Clin North Am 2015; 62: 61-73.
23.
Davidsson J, Lilljebjörn H, Andersson A, et al. The DNA methylome of pediatric acute lymphoblastic leukemia. Hum Mol Genet 2009; 18: 4054-65.
24.
Sinnett D, Krajinovic M, Labuda D. Genetic susceptibility to childhood acute lymphoblastic leukemia. Leuk Lymphoma 2000; 38: 447-62.
25.
Kawamata N, Ogawa S, Zimmermann M, et al. Molecular allelokaryotyping of pediatric acute lymphoblastic leukemias by high-resolution single nucleotide polymorphism oligonucleotide genomic microarray. Blood 2008; 111: 776-84.
26.
Rivera G, Pui CH, Abromowitch M, et al. Improved outcome in childhood acute lymphoblastic leukaemia with reinforced early treatment and rotational combination chemotherapy. Lancet 1991; 337: 61-6.
27.
Nadeau G, Ouimet-Grennan E, Aaron M, et al. Identification of genetic variants associated with skeletal muscle function deficit in childhood acute lymphoblastic leukemia survivors. Pharmgenom Pers Med 2019; 12: 33.
28.
Suárez-Llanos JP, Vallejo-Torres L, García-Bello MÁ, et al. Cost-effectiveness of the hospital nutrition screening tool CIPA. Arch Med Sci 2019; 16: 273-81.
29.
Dancey J, Steward WP. The role of vindesine in oncology – recommendations after 10 years’ experience. Anticancer Drugs 1995; 6: 625-36.
30.
Renbarger JL, McCammack KC, Rouse CE, Hall SD. Effect of race on vincristine-associated neurotoxicity in pediatric acute lymphoblastic leukemia patients. Pediatr Blood Cancer 2008; 50: 769-71.
31.
Mao W, Wu F, Lee RJ, Lu W, Wang J. Development of a stable single-vial liposomal formulation for vincristine. Int J Nanomed 2019; 14: 4461-74.
32.
Polikandrioti M, Panoutsopoulos G, Tsami A, et al. Assessment of quality of life and anxiety in heart failure outpatients. Arch Med Sci Atheroscler Dis 2019; 4: e38-6.
33.
Polikandrioti M, Kalafatakis F, Koutelekos I, Kokoularis D. Fatigue in heart failure outpatients: levels, associated factors, and the impact on quality of life. Arch Med Sci Atheroscler Dis 2019; 4: e103-12.
34.
Akira S, Hirano T, Taga T, Kishimoto T. Biology of multifunctional cytokines: IL 6 and related molecules (IL 1 and TNF). FASEB J 1990; 4: 2860-7.
35.
Nakajima K, Yamanaka Y, Nakae K, et al. A central role for Stat3 in IL-6-induced regulation of growth and differentiation in M1 leukemia cells. EMBO J 1996; 15: 3651-8.
36.
Foa R, Massaia M, Cardona S, et al. Production of tumor necrosis factor-alpha by B-cell chronic lymphocytic leukemia cells: a possible regulatory role of TNF in the progression of the disease. Blood 1990; 76: 393-400.
37.
Sethi G, Sung B, Aggarwal BB. TNF: a master switch for inflammation to cancer. Front Biosci 2008; 13: 5094-107.
38.
Digel W, Stefanic M, Schoniger W, et al. Tumor necrosis factor induces proliferation of neoplastic B cells from chronic lymphocytic leukemia. Blood 1989; 73: 1242-6.
39.
Xing Z, Gauldie J, Cox G, et al. IL-6 is an antiinflammatory cytokine required for controlling local or systemic acute inflammatory responses. J Clin Invest 1998; 101: 311-20.
40.
Pels E. Concentration of tumour necrosis factor-alpha in saliva of patients with acute lymphoblastic leukaemia in relation to oral mucositis during chemotherapy. Chemo Open Access 2015; 4: 2.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.