Introduction
Upper gastrointestinal endoscopy is a well-established, highly effective diagnostic and therapeutic procedure [1]. With the aging of the general population, the frequency of this procedure in the elderly is increasing rapidly [2, 3]. Although it is a non-traumatic invasive procedure, it often results in significant discomfort in the majority of patients, requiring sedation during the thorough examination [4]. Currently, sedation during upper gastrointestinal endoscopy is mainly obtained by using either propofol or a benzodiazepine, sometimes in combination with an analgesic [5, 6]. Propofol is regarded as a classical sedative, due to its rapid recovery profile [7]. However, propofol has many side effects, the most important of which are respiratory depression and hypotension [8]. The principal disadvantages of benzodiazepines for sedation are that they do not provide analgesia and have long half-lives [9]. Even midazolam, the shortest-acting of the benzodiazepines, has a half-life of approximately 1.8 to 6.4 h [10]. In addition, midazolam has an active metabolite, which has a profound contribution to its sedative profile, especially for a longer and unpredictable recovery from sedation [11].
Remimazolam is a new benzodiazepine class of sedative drugs equivalent to those of drugs such as midazolam but with a shorter terminal half-life [12]. It is designed to undergo rapid hydrolysis in the body by ubiquitous tissue esterases to its pharmacologically inactive carboxylic acid metabolite [13]. Because of its rapid and predictable onset and recovery and organ-independent metabolism, remimazolam appears to have potential advantages when used as an intravenous sedative agent in maintaining suitable sedation during upper gastrointestinal endoscopy [14, 15]. Remimazolam tosilate (HR7056) is a toluene sulfonate compound developed on the basis of remimazolam and was approved for marketing by the Chinese National Medical Products Administration for sedation during routine gastroscopy in December 2019. Currently, there are no reports about the safety and efficacy of HR7056 in sedation during upper gastrointestinal endoscopy. In this study, we aimed to assess both the safety and efficacy of different doses of HR7056 in maintaining suitable sedation during upper gastrointestinal endoscopy and the reversibility of HR7056’s sedative effects with flumazenil.
Material and methods
Study design
This study was divided into 2 parts: dose-exploration and flumazenil reversal. The objective of the dose-exploration part of the study was to assess the safety and efficacy of maintaining suitable sedation levels with HR7056 during upper gastrointestinal endoscopy. The objective of the flumazenil reversal part of the study was to assess the reversibility of HR7056’s sedative effects with flumazenil. This multicenter, randomized, single-blind, dose-exploration, positive controlled parallel, phase II trial enrolled 156 patients from November 2016 to March 2017 and involved nine centers in China. This trial was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice Guidelines and approved by the ethics committee institutional review board of each participating center. Written informed consent was obtained from all patients.
Patients
The main inclusion criteria were as follows: 1) patients aged 18–80 years; 2) patients undergoing routine upper gastrointestinal endoscopy; 3) patients with only American Society of Anesthesiologists (ASA) grade I or II; 4) patients with body mass index (BMI) 18–30 kg/m2; 5) patients with gastroscopy time less than 30 min.
The exclusion criteria were as follows: 1) patients suspected of upper gastrointestinal bleeding; 2) patients with severe respiratory diseases (obstructive sleep apnea syndrome, acute respiratory infection, asthma, etc.), acute heart failure or unstable angina, anemia, thrombocytopenia, abnormal liver function or abnormal kidney function; 3) patients who had a known sensitivity to benzodiazepines, opiates, naloxone, flumazenil, or a contraindication to receiving these medications and their components; 4) pregnant or lactating women; 5) patients requiring complicated endoscopic diagnosis and treatment techniques (such as pancreaticobiliary angiography, ultrasound endoscopy, etc.) or planning to undergo tracheal intubation; 6) patients who had participated in drug clinical trials as subjects in the past 3 months; 7) patients suspected of having gastroduodenal outflow obstruction accompanied by retention of contents; 8) patients diagnosed with respiratory tract management difficulties (modified Mallampati score was IV); 9) patients who had a drug or alcohol addiction two years prior to the study; 10) patients with sitting systolic blood pressure ≤ 90 mm Hg in the screening period; 11) hypertension patients with unsatisfactory blood pressure control treated with antihypertensive drugs (sitting systolic blood pressure ≥ 160 mm Hg, and/or diastolic blood pressure ≥ 100 mm Hg); 12) patients with a history of recent use of narcotics, analgesics, anesthetics and benzodiazepine drugs and a history of cerebral disease or mental disorder.
Intervention
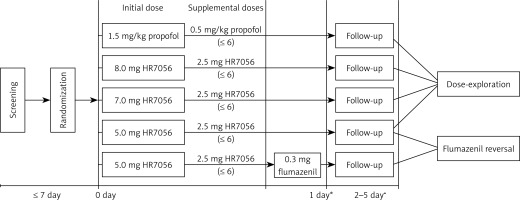
All eligible patients were randomly assigned to five groups in a ratio of 1 : 1 : 1 : 1 : 1 by the random number created by a computer-generated coding system. In China, midazolam was rarely used in painless gastroscopy, and the mainstream first-line drug was propofol. Therefore, propofol was used as the comparator in this study. Patients were scheduled to receive either HR7056 (Jiangsu Hengrui Medicine Co., Ltd, Jiangsu, China) or propofol (AstraZeneca, Zug, Switzerland) intravenously during upper gastrointestinal endoscopy for sedation induction and maintenance (Figure 1). The single intravenous bolus injection time was about 1 min. The initial dose of propofol was 1.5 mg/kg (group A), and the initial dose of HR7056 was 8.0 mg (group B), 7.0 mg (group C) or 5.0 mg (group D). When adequate sedation (defined as a Modified Observer’s Assessment of Alertness/Sedation (MOAA/S) score of ≤ 3) was achieved, gastroscopy was performed. If adequate sedation to allow the start of the procedure could not be achieved with the initial dose of HR7056 or propofol, up to a maximum of 2 supplemental doses of 2.5 mg (HR7056) or 0.5 mg/kg (propofol) were administered as IV boluses over about 15 s, not less than 2 min apart. Once the procedure was underway, supplemental doses of 2.5 mg (HR7056) or 0.5 mg/kg (propofol) were administered at the investigator’s discretion, at least 2 min apart (not to exceed a cumulative total of 6 supplemental boluses) to sustain a MOAA/S ≤ 4. In the 5.0 mg HR7056 plus flumazenil group (group E), intravenous injection of 0.3 mg of flumazenil was administered immediately after upper gastrointestinal endoscopy. From the subject’s sedation induction to fully alert, the investigator needs to monitor indicators such as MOAA/S score, heart rate, blood pressure, blood oxygen saturation, and respiratory rate. All drugs were prepared by an anesthesiologist who was blinded to this study. An investigator who was blinded to the group assignment recorded and assessed all observed parameters.
Clinical assessment
For the dose-exploration part of the study, the primary efficacy endpoint was the successful sedation rate. It was defined as the proportion of subjects who experienced successful sedation during upper gastrointestinal endoscopy. Successful sedation was defined as follows: 1) completion of the procedure of upper gastrointestinal endoscopy; 2) MOAA/S ≤ 4 for 3 consecutive measurements per minute; 3) no manual or mechanical ventilation; 4) no requirement for rescue sedative medication (some other drugs, such as midazolam).
Secondary efficacy endpoints included the following: 1) sedative recovery time. It was defined as the time between discontinuation of HR7056 or propofol to the first of 3 consecutive MOAA/S scores of 5; 2) rate of hypotension as measured by the proportion of subjects who experienced hypotension (the lowest systolic blood pressure < 100 mm Hg or a decrease of more than 20% of baseline) during upper gastrointestinal endoscopy; 3) rate of hypoxemia as measured by the proportion of subjects who experienced hypoxemia (oxygen saturation < 90%) during upper gastrointestinal endoscopy; 4) rate of respiratory depression as measured by the proportion of subjects who experienced respiratory depression (respiratory rate < 10 times/min) during upper gastrointestinal endoscopy; 5) pain on injection. When the initial dose of HR7056 or propofol was given for about 10 s, the researchers asked the subjects about the pain on injection. The pain degree was evaluated according to patients’ statement (0 points, no pain; 1 point, mild pain; 2 points, moderate pain; 3 points, severe pain). In order to eliminate the effect of rescue sedative, all the secondary efficacy endpoints were analyzed in the sedative successful subjects with the exception of the pain on injection.
For the flumazenil reversal part of the study, the primary efficacy endpoint was the sedative recovery time. It was defined as the time between discontinuation of HR7056 to the first of 3 consecutive MOAA/S scores of 5.
Safety assessments
Safety of treatment regimens was assessed by the changes of vital signs, physical examinations and laboratory data, and the incidence of adverse events (AEs). Assessment of vital signs (including heart rate, blood pressure, respiratory rate, etc.) and physical examination were performed at 2–5 days after the operation. In addition, blood routine, urine routine, blood biochemistry, and electrocardiogram were performed. Any AEs were recorded during the trial period.
Statistical analysis
This study was a phase II clinical study. According to the Chinese National Medical Registration Management Regulations, the sample size of a phase II clinical study should be more than 100. There was no need to estimate based on statistical assumptions, but it was expected to provide sufficient data to support our research purposes. Therefore, the sample size was 150 cases (30 subjects per group).
Efficacy analysis was based on the intention-to-treat (ITT) population, defined as all randomized patients who received at least one dose of medication and for whom evaluable data of therapeutic effectiveness were available. All efficacy analyses were performed for patients in the full analysis set (FAS). Safety assessment was analyzed in the safety set (SS), who had received at least 1 dose of study drug of medication and at least 1 assessment of safety data.
All statistical analyses were performed using the software SAS 9.4 (SAS Institute, Cary, NC, USA). Quantitative data were expressed as means ± standard deviations (SD). Qualitative data were expressed as number and frequency. For the primary efficacy endpoint, the χ2 test was used to perform the between-group comparisons. For secondary efficacy endpoints, Fisher’s exact test, the Kruskal-Wallis test or oneway analysis of variance (ANOVA) was used to perform the between-group comparisons. Multiple comparison was performed by Fisher’s exact test or the Wilcoxon rank-sum test. All statistical tests were two-sided, with significance set at p < 0.05 along with 95% CI.
Results
Baseline characteristics
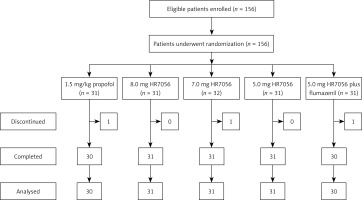
Of the 156 eligible patients enrolled in this study, all patients were randomly assigned to receive propofol, HR7056 (8.0 mg, 7.0 mg or 5.0 mg), or 5.0 mg HR7056 plus flumazenil (Figure 2). A total of 153 patients were included in FAS and SS analysis (group A: n = 30; group B: n = 31; group C: n = 31; group D: n = 31; group E: n = 30). The reasons for non-inclusion in FAS and SS analysis were non-use of flumazenil (n = 1) and withdrawal of informed consent (n = 2). Baseline characteristics of patients are shown in Table I. The five treatment groups were generally well balanced for age, gender, ASA grade, weight, height, BMI, vital signs (including heart rate, blood pressure, respiratory rate, etc.) and disease history (all p > 0.05).
Table I
Baseline characteristics of the patients
[i] Group A – propofol, group B – 8.0 mg remimazolam tosilate (HR7056); group C – 7.0 mg remimazolam tosilate (HR7056), group D – 5.0 mg remimazolam tosilate (HR7056); group E – 5.0 mg remimazolam tosilate (HR7056) plus flumazenil, BMI – body mass index, SD – standard deviations, ASA – American Society of Anesthesiologists, IQR – interquartile range.
Dose-exploration outcomes
In this part of the study, we assessed the feasibility of maintaining suitable sedation levels with HR7056 during upper gastrointestinal endoscopy. The supplemental doses of HR7056 and propofol for sedation induction and maintenance are shown in Table II. No difference was found in total doses between groups B, C and D (p = 0.461). The primary efficacy endpoint of the study was the successful sedation rate (Table III). The successful sedation rate was 87.10%, 100.00%, and 83.87% in group B, group C, and group D, respectively, compared with a sedation success rate of 96.67% in group A. There was no significant difference between the four treatment groups (p = 0.057). Procedure failures were all caused by the requirement for a rescue sedative.
Table II
HR7056 and propofol dosing in the study arms
Table III
The successful sedation rate of each group
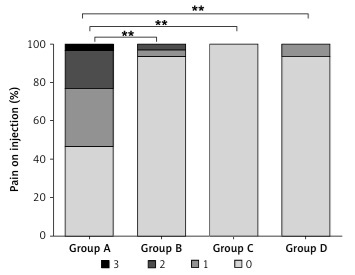
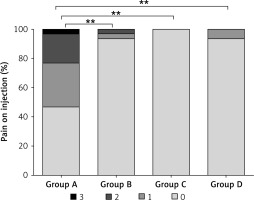
The results of the secondary efficacy endpoint are shown in Table IV and Figure 3. There was a significant difference between the four treatment groups (p = 0.004) in sedation recovery time. In addition, the sedative recovery time in group A was significantly higher than that in group D (8.03 ±3.77 min vs. 5.38 ±1.83 min, p < 0.05, Table IV). These results indicated that the initial dose of 5.0 mg of HR7056 was capable of inducing rapid sedation with a quick recovery profile in patients undergoing upper gastrointestinal endoscopy. No significant difference was observed in the rate of hypotension (p = 0.671) or the rate of respiratory depression (p = 0.171) between any of the treatment groups. The rate of hypoxemia in group A was significantly higher than that in all HR7056 dose groups (0.69% vs. 0.00% (groups B, C and D), all p < 0.05, Table IV). Similarly, the pain on injection in group A was significantly higher than that in all HR7056 dose groups (all p < 0.001, Figure 3).
Table IV
The secondary efficacy endpoint in the sedative successful subjects
| Parameter | Group A (n = 29) | Group B (n = 27) | Group C (n = 31) | Group D (n = 26) | p-value |
|---|---|---|---|---|---|
| Sedative recovery time, mean ± SD [min] | 8.03 ±3.77 | 5.78 ±2.81 | 6.29 ±2.48 | 5.38 ±1.83* | 0.004 |
| Rate of hypotension, n (%) | 6 (20.69) | 4 (14.81) | 3 (9.68) | 3 (11.54) | 0.671 |
| Rate of hypoxemia, n (%) | 6 (20.69) | 0 (0.00)* | 0 (0.00)* | 0 (0.00)* | 0.001 |
| Rate of respiratory depression, n (%) | 2 (6.90) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.171 |
Flumazenil reversal outcomes
In this part of the study, we assessed the reversibility of HR7056’s sedative effects with flumazenil. The results showed that there was no significant difference in the sedative recovery time between group D and group E (5.38 ±1.83 min vs. 4.97 ±1.57 min, p = 0.879), indicating that the initial dose of HR7056 5.0 mg could induce rapid sedation without the need for flumazenil reversal.
Safety analysis
A summary of adverse events is listed in Table V. Overall, AEs were reported in 12 (40.00%) patients in group A, 9 (29.03%) patients in group B, 5 (16.13%) patients in group C, 2 (6.45%) patients in group D and 2 (6.67%) patients in group E. There was no significant difference between groups D and E (p = 1.000). The rate of AEs in group A was significantly higher than that in groups C and D (all p < 0.05). The majority of AEs were mild adverse events and there were no serious AEs. No notable changes were observed in vital signs, physical examinations or laboratory data.
Table V
Summary of adverse events
| Parameter | Group A (n = 30) | Group B (n = 31) | Group C (n = 31) | Group D (n = 31) | Group E (n = 30) | p-value |
|---|---|---|---|---|---|---|
| Any adverse event, n (%) | 12 (40.00) | 9 (29.03) | 5 (16.13)* | 2 (6.45)* | 2 (6.67) | 0.003 |
| Mild adverse event, n (%) | 10 (33.33) | 8 (25.81) | 3 (9.68) | 2 (6.45) | 2 (6.67) | |
| Moderate adverse event, n (%) | 2 (6.67) | 1 (3.22) | 2 (6.45) | 0 (0.00) | 0 (0.00) |
Discussion
Currently, sedation during upper gastrointestinal endoscopy is mainly obtained by using either midazolam or propofol [5, 6, 16]. However, despite the documented sedation effectiveness of midazolam and propofol, each drug has its disadvantages [17]. HR7056 is a new benzodiazepine class of sedative drugs, which possesses a faster onset, a shorter duration of sedative action, and a more rapid recovery than currently available short-acting sedatives [13, 18]. Currently, there are no reports about the safety and efficacy of HR7056 in sedation during upper gastrointestinal endoscopy. In this study, we aimed to assess both the safety and efficacy of different doses of HR7056 in maintaining suitable sedation during upper gastrointestinal endoscopy and the reversibility of HR7056’s sedative effects with flumazenil.
In this study, we found that HR7056 has the attributes of a sedative drug, with success rates comparable with propofol. Pambianco et al. found that the sedative recovery time in the remimazolam 5.0/3.0 mg group (initial dose 5.0 mg, combined with top-up doses of up to 3.0 mg) was 13.3 ±7.21 min during colonoscopy [12]. These results were found to be higher than in our study. In our study, we found that the sedative recovery time in the 5.0 mg HR7056 group was 5.38 ±1.83 min. The possible reasons were that a supplementary dose of fentanyl was given before procedures were started. Moreover, the operating time was longer for colonoscopy than gastroscopy and the cumulative dose could be higher. In addition, the rate of hypoxemia and pain on injection in the propofol group were significantly higher than those in all HR7056 groups. These results were consistent with a phase III study, carried out to compare the efficacy of remimazolam with propofol in 375 subjects, which showed that a general anesthetic effect was observed in all the patients of each group [19]. However, the phase III study showed that the advantage of remimazolam over propofol was that there was no pain on injection.
A previous study showed that the remimazolam 5.0/3.0 mg dose (initial dose 5.0 mg, combined with top-up doses of up to 3.0 mg) group demonstrated the highest sedation success rate and at the same time the best safety profile [12]. In this study, we found that there was no significant difference in the successful sedation rate between any of the treatment groups. However, the sedative recovery time in the propofol group was significantly longer than that in the 5.0 mg HR7056 group. These results indicated that the HR7056 5.0/2.5 mg dose was capable of inducing rapid sedation with a quick recovery profile in patients undergoing upper gastrointestinal endoscopy. Our results were consistent with a phase III study [20], which showed that fentanyl 50–75 µg, followed by remimazolam at an initial dose of 5.0 mg and subsequent doses of 2.5 mg as needed, resulted in adequate sedation for colonoscopy.
Flumazenil is a competitive benzodiazepine receptor antagonist that is used to reverse or block the effects of benzodiazepines [21, 22]. Worthington et al. reported that sedation of remimazolam was rapidly reversible by flumazenil (1.0 min flumazenil vs. 10.5 min placebo) [23]. However, the flumazenil reversal part of the study showed that the sedative recovery time between group A and group E was similar, which did not reflect the advantage of flumazenil as a reversal agent. On the one hand, it may be that the short operation time of gastroscopy and the small stimulating effect on the subject resulted in the fact that the subject did not need to be sedated deeply, so the cumulative dose of the drug was generally not excessive. Therefore, whether or not the sedative reversal agent was used, the subjects could wake up quickly after the operation. On the other hand, it was limited by the sample size.
With regard to the safe assessment, we found that the rate of AEs in the propofol group was significantly higher than that in 5.0 mg and 7.0 mg HR7056 groups. The rates of respiratory depression and hypoxemia were 6.90% and 20.69% in the propofol group, compared with the rate of respiratory depression and hypoxemia of 0.00% in the HR7056 groups. Hypotension occurred in all groups, but the rate of hypotension in the propofol group (20.69%) was higher than that in the HR7056 groups. Similarly, some patients sedated with propofol may have suffered hypotension and oxygen desaturation during colonoscopy [23]. There were no serious AEs in any of the treatment groups. Overall, our study demonstrated the acceptable safety and tolerability for HR7056.
Although the results of this study were very encouraging, the principal limitations were the small sample size and lack of placebo control for the flumazenil reversal part. Further studies are needed in patients undergoing upper gastrointestinal endoscopy to refine the optimal dosing regimen before widespread clinical use.
In conclusion, HR7056 was comparable to propofol in safety and efficacy in maintaining sedation during upper gastrointestinal endoscopy. HR7056 in a 5.0/2.5 mg dose (initial dose 5.0 mg, combined with supplemental doses of 2.5 mg) was capable of inducing rapid sedation without the need for flumazenil reversal. This provides the basis for further development of this short-acting compound.