Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
ONCOLOGY / BASIC RESEARCH
The relationship between RSUME and VHL/HIF-1α signalling pathway in renal cell carcinoma
1
Departments of Urology, The Affiliated Hospital of Zunyi Medical University, Zunyi, Guizhou, China
Submission date: 2020-03-17
Final revision date: 2020-07-03
Acceptance date: 2020-07-16
Online publication date: 2020-08-11
Publication date: 2026-04-30
Corresponding author
Guobiao Liang
Department of Urology Affiliated Hospital of Zunyi Medical University 149 Dalian Road Zunyi, Guizhou 563000 China Phone: +86 18786916696 Fax: +86 21-64085875
Department of Urology Affiliated Hospital of Zunyi Medical University 149 Dalian Road Zunyi, Guizhou 563000 China Phone: +86 18786916696 Fax: +86 21-64085875
Arch Med Sci 2026;22(2):1064-1073
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Renal cell carcinoma (RCC) is characterised by loss of the von Hippel-Lindau tumour suppressor gene (VHL), and the functional tumourigenic consequences of this loss have been used to develop therapies for advanced renal cell carcinoma, such as targeting activation of the HIF pathway. Human RWD containing sumoylation enhancer (RSUME) has been previously reported to play a promoting role in pituitary tumours. However, the exact role of RSUME in RCC remains unclear. The present study aimed to investigate the expression levels of VHL, HIF-1α, and RSUME genes and proteins in human RCC tissues, and their correlations.
Material and methods:
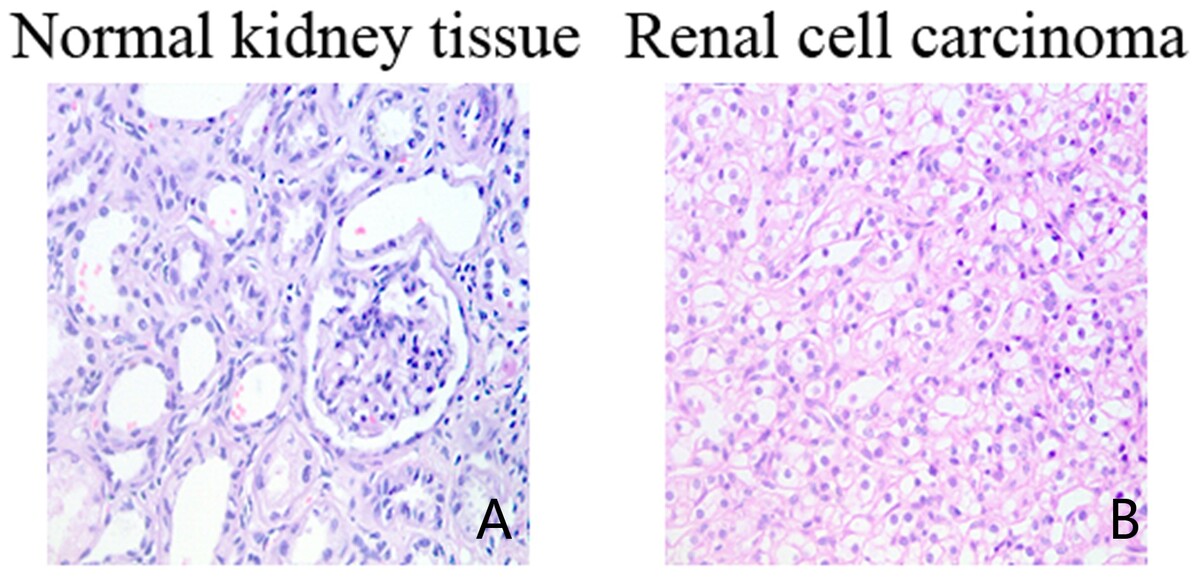
Immunohistochemistry and real-time PCR were applied to detect the expression levels of VHL, HIF-1α, and RSUME in normal renal tissue (control group) and RCC (experimental group).
Results:
The results revealed that RSUME and HIF-1α were significantly upregulated in RCC tissues compared with normal renal tissues (p < 0.05), while the expression level of VHL was significantly lower than that in normal renal tissues (p < 0.05). Correlation analysis results showed that the expression of VHL was negatively correlated with HIF-1α and RSUME, respectively (p < 0.05). However, a positive correlation between RSUME and HIF-1α expression was observed (p < 0.05).
Conclusions:
These results suggested that upregulated expression of RSUME may be involved in the formation and progression of renal cell carcinoma via interaction with VHL/HIF-1α signalling pathway, and thus RSUME may be a novel potential therapeutic target for renal cell carcinoma.
Renal cell carcinoma (RCC) is characterised by loss of the von Hippel-Lindau tumour suppressor gene (VHL), and the functional tumourigenic consequences of this loss have been used to develop therapies for advanced renal cell carcinoma, such as targeting activation of the HIF pathway. Human RWD containing sumoylation enhancer (RSUME) has been previously reported to play a promoting role in pituitary tumours. However, the exact role of RSUME in RCC remains unclear. The present study aimed to investigate the expression levels of VHL, HIF-1α, and RSUME genes and proteins in human RCC tissues, and their correlations.
Material and methods:
Immunohistochemistry and real-time PCR were applied to detect the expression levels of VHL, HIF-1α, and RSUME in normal renal tissue (control group) and RCC (experimental group).
Results:
The results revealed that RSUME and HIF-1α were significantly upregulated in RCC tissues compared with normal renal tissues (p < 0.05), while the expression level of VHL was significantly lower than that in normal renal tissues (p < 0.05). Correlation analysis results showed that the expression of VHL was negatively correlated with HIF-1α and RSUME, respectively (p < 0.05). However, a positive correlation between RSUME and HIF-1α expression was observed (p < 0.05).
Conclusions:
These results suggested that upregulated expression of RSUME may be involved in the formation and progression of renal cell carcinoma via interaction with VHL/HIF-1α signalling pathway, and thus RSUME may be a novel potential therapeutic target for renal cell carcinoma.
REFERENCES (21)
1.
Kondo K, Yao M, Yoshida M, et al. Comprehensive mutational analysis of the VHL gene in sporadic renal cell carcinoma: relationship to clinicopathological parameters. Genes Chromosomes Cancer 2002; 34: 58-68.
2.
Vortmeyer AO, Choo D, Pack S, Oldfield E, Zhuang Z. VHL gene inactivation in an endolymphatic sac tumor associated with von Hippel-Lindau disease. Neurology 2000; 55: 460.
3.
Baldewijns MM, van Vlodrop IJ, Vermeulen PB, Soetekouw PM, van Engeland M, de Bruine AP. VHL and HIF signalling in renal cell carcinogenesis. J Pathol 2010; 221: 125-38.
4.
Pavlović I, Pejić S, Radojević-Škodrić S, et al. The effect of antioxidant status on overall survival in renal cell carcinoma. Arch Med Sci 2020; 16: 94-101.
5.
van der Veldt AA, Haanen JB, van den Eertwegh AJ, Boven E. Targeted therapy for renal cell cancer: current perspectives. Discov Med 2010; 10: 394-405.
6.
Banyra O, Tarchynets M, Shulyak A. Renal cell carcinoma: how to hit the targets? Cent European J Urol 2013; 66: 394-404.
7.
Carbia-Nagashima A, Gerez J, Perez-Castro C, et al. RSUME, a small RWD-containing protein, enhances SUMO conjugation and stabilizes HIF-1alpha during hypoxia. Cell 2007; 131: 309-23.
8.
Shan B, Gerez J, Haedo M, et al. RSUME is implicated in HIF-1-induced VEGF-A production in pituitary tumour cells. Endocr Relat Cancer 2011; 19: 13-27.
9.
Chen X, Kuang W, Huang H, et al. Knockdown of RWD domain containing 3 inhibits the malignant phenotypes of glioblastoma cells via inhibition of phosphoinositide 3-kinase/protein kinase B signaling. Exp Ther Med 2018; 16: 384-93.
10.
Livak KJ, Schmittgen TDJ. Analysis of relative gene expression data using real-time quantitative PCR and the 2−DDCT method. Methods 2001; 25: 402-8.
11.
Sato Y, Yoshizato T, Shiraishi Y, et al. Integrated molecular analysis of clear-cell renal cell carcinoma. Nat Genet 2013; 45: 860-7.
12.
The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 2013; 499: 43.
13.
Hughson MD, He Z, Liu S, Coleman J, Shingleton WB. Expression of HIF-1 and ubiquitin in conventional renal cell carcinoma: relationship to mutations of the von Hippel-Lindau tumor suppressor gene. Cancer Genetics Cytogenet 2003; 143: 145-53.
14.
Maxwell PH, Pugh CW, Ratcliffe PJ. The pVHL-hIF-1 system. A key mediator of oxygen homeostasis. Adv Exp Med Biol 2001; 502: 365-76.
15.
Fuertes M, Gerez J, Haedo M, et al. Cytokines and genes in pituitary tumorigenesis: RSUME role in cell biology. Front Hormone Res 2010; 38: 1-6.
16.
Melchior F. SUMO: nonclassical ubiquitin. Ann Rev Cell Developm Biol 2000; 16: 591-626.
17.
Vertegaal AC, Ogg SC, Jaffray E, et al. A proteomic study of SUMO-2 target proteins. J Biol Chem 2004; 279: 33791-8.
18.
Fowkes RC, Vlotides G. Hypoxia-induced VEGF production ‘RSUMEs’ in pituitary adenomas. Endocrine Relat Cancer 2012; 19: C1-5.
19.
Gerez J, Fuertes M, Tedesco L, et al. In silico structural and functional characterization of the RSUME splice variants. PLoS One 2013; 8: e57795.
20.
Chan JY, Tsai CY, Wu CH, et al. Sumoylation of hypoxia-inducible factor-1alpha ameliorates failure of brain stem cardiovascular regulation in experimental brain death. PLoS One 2011; 6: e17375.
21.
Bonora M, Wieckowski MR, Chinopoulos C, et al. Molecular mechanisms of cell death: central implication of ATP synthase in mitochondrial permeability transition. Oncogene 2015; 34: 1475.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.