Introduction
Medical and technological progress has allowed for longer life expectancy. By 2030, there is a probability of more than 50% that female life expectancy will break the 90 years barrier [1]. Longer life span implies an increasing incidence of chronic medical conditions such as cancer and chronic kidney disease. Renal replacement therapy, such as hemodialysis, has allowed a significant number of patients suffering from the most severe form of chronic kidney disease to live an acceptable life span. The risk for the development of malignancy in patients suffering from end-stage renal disease differs among different cancers; previous studies have reported an increased risk for developing urinary tract cancer, liver cancer, and breast cancer [2–4]. However, some authors have found a higher incidence of lung cancer [5], while others found no difference [6–8] or even a lower incidence of lung cancer in patients receiving dialysis [2, 9]. The literature regarding the clinical outcome of lung cancer with chronic kidney disease under renal replacement therapy (hemodialysis) is scarce [4, 10]. Possible reasons include the hesitant medical attitude toward these patients given their shortened life expectancy, the increased mortality related to cardiovascular conditions, and the problematic drug dosage adjustment or simply therapeutic abstention. Population-based survival analysis has demonstrated contradictory results for lung cancer with end-stage renal disease patients (WITH-ESRD) compared to lung cancer without end-stage renal disease patients (WITHOUT-ESRD) [4, 10].
This study aims to determine the clinical outcome of lung cancer with end-stage renal disease through a matched-control study.
Material and methods
The institutional review board of Chang Gung Memorial Hospital has approved this retrospective study. Using the Chang Gung Memorial Hospital Cancer Registry and Death registration (CGRD), all adult patients (older than 18 years) diagnosed with end-stage renal disease (International Classification of Diseases (ICD-9) code 585) and coexisting diagnosis of lung cancer (International Classification of Diseases (ICD-9) code 162) from January 2001 to December 2014 were included in this study. Lung cancer patients with coexisting end-stage renal disease were matched 1 : 4 to patients with lung cancer without end-stage renal disease based on age, gender, cancer histology, cancer stage, and treatment type. CGRD is a high-quality cancer registry and provides information regarding specific demographics, disease staging, histology type, and primary treatment details. The 7th edition of the TNM staging system for lung cancer served for lung cancer staging [11]. We excluded cases with an incomplete medical record or the absence of a pathology report reconfirmed by our in-house pathologist. Overall survival was determined from the day of pathology confirmation until the last follow-up or at the end of 2015, whatever comes first. Patients lost to follow-up were contacted by telephone by the cancer center case manager, and those not reachable by telephone were presumed dead if withdrawn from the National Health Insurance. The National Health Insurance offers universal coverage to more than 99% of the Taiwanese population. Those patients excluded are usually from death, missing premium payments > 6 months, emigration or change of nationality. The Health Promotion Administration, Ministry of Health and Welfare, Taiwan, releases an annual death report of all cancer cases registered back to each cancer center for the status update.
Statistical analysis
We presented continuous data as the median plus standard deviation and categorical data as percentages. We used Pearson’s χ2 test to compare categorical variables and the Mann-Whitney U test for continuous variables. We categorized continuous variables into a categorical variable using median values as the cut-off point. We divided the clinical stage into stage I–IIIA versus stage IIIB–IV, and treatment modality into supportive treatment, surgical treatment (excluding diagnostic/staging procedure) and medical treatment group. We used the Kaplan-Meier method for survival analysis (overall survival), and the difference in survival was calculated using the log-rank test. Cox proportional hazard analysis was used to estimate the level of significance and the relative risks with 95% confidence interval. A p-value of less than 0.05 was considered significant. All the analysis was conducted using SAS statistical software (version 9.4; SAS Institute, Cary, NC, USA).
Results
Patient selection
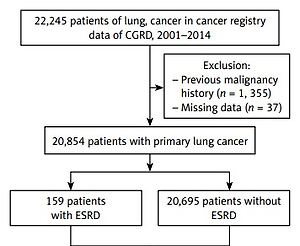
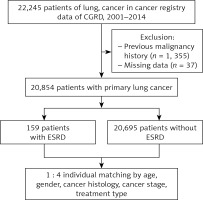
After the exclusion of 1355 patients with a previous second malignancy and 37 patients with incomplete clinical data, we included 20854 lung cancer patients from the cancer registry. There were 20695 WITHOUT-ESRD patients and 159 WITH-ESRD patients. WITHOUT-ESRD patients matched WITH-ESRD patients in a 1-to-4 ratio based on age, gender, cancer histology type, cancer stage, and treatment type. Figure 1 shows the matching process.
Survival outcome of patients with lung cancer without and with end-stage renal disease
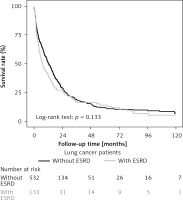
There was no statistically significant difference in baseline clinical characteristics after the matching process. Table I presents the clinical characteristics of the cohort. Survival analysis revealed no survival difference for WITH-ESRD and WITHOUT-ESRD patients (Figure 2). The median overall survival for WITH-ESRD patients was 7.36 months (95% CI: 5.19–11.10) compared to 12.25 months (95% CI: 10.81–14.65) for WITHOUT-ESRD patients, p = 0.133 (Table II). There was a marginal significant difference in survival by gender. The median survival for male WITH-ESRD patients was 6.31 months (95% CI: 3.71–8.51) compared to 11.56 months (95% CI: 8.77–14.19) for WITHOUT-ESRD patients, p = 0.059, while the difference was not significant for female patients. The median overall survival differed significantly for patients with advanced stage lung cancer and patients with medical treatment. For advanced stage lung cancer (stage IIIB/IV), the median overall survival for WITH-ESRD patients was 5.19 months (95% CI: 3.71–5.76) compared to 9.63 months (95% CI: 7.85–11.27) for WITHOUT-ESRD patients, p = 0.022. For the medical treatment group, the median overall survival for WITH-ESRD patients was 5.98 months (95% CI: 4.34–11.76) compared to 14.13 months (95% CI: 11.30–16.43) for WITHOUT-ESRD patients, p = 0.019. Table II presents the median survival according to different clinical characteristics.
Table I
Cohort and matched-cohort demographic characteristics of patients
Table II
Kaplan-Meier survival analysis for WITHOUT-ESRD and WITH-ESRD group
[i] The overall survival was not statistically significantly different between the WITHOUT-ESRD and WITH-ESRD group. Survival for male WITHOUT-ESRD patients is nearly double the survival for male WITH-ESRD patients (median survival of 11.56 months versus 6.31 months respectively, p = 0.059). There is a significant survival difference between WITH-ESRD and WITHOUT-ESRD groups for cancer stage IIIB–IV patients (median survival of 5.19 months compared to 9.63 months), p = 0.022. NSCLC – non-small cell lung cancer. SCLC – small cell lung cancer.
Figure 2
Survival curve for lung cancer WITH-ESRD and WITHOUT-ESRD. For WITH-ESRD patients, the median overall survival was 7.36 months (95% CI: 5.19–11.10) compared to 12.25 months (95% CI: 10.81–14.65) for patients WITHOUT-ESRD, p = 0.133
Cox regression analysis revealed that male gender and stage IIIB–IV were independent factors associated with a poor outcome for WITH-ESRD patients (Table III). Although patients with surgical treatment compared to those with medical treatment had an improvement of survival by 46% (HR = 0.54, 95% CI: 0.19–1.53, p = 0.243), the difference did not reach statistical significance.
Table III
Adjusted Cox proportional hazard analysis for WITH-ESRD group
[i] Female gender and cancer stage I–IIIA were associated with improved survival. Although patients receiving surgical treatment showed improved survival of 46% (HR = 0.54, 95% CI: 0.19–1.53), it did not reach statistical significance (p = 0.243). NSCLC – non-small cell lung cancer, SCLC – small cell lung cancer.
Discussion
Patients with cancer are usually older in general, with a higher prevalence of comorbidities, such as chronic kidney disease. The prevalence of chronic kidney disease is exceptionally high in the elderly, affecting > 40% of people over the age of 70 years [12]. According to the 2007–2010 National Health and Nutrition Examination Surveys, the prevalence of chronic kidney disease in the United States was 14% (48.3% at stage 3 and 4.7% at stage 4–5) [13]. The estimated prevalence of CKD in Taiwanese patients according to a recent report by Tsai et al. was 15.46% for CKD stage 1–5 and 9.06% for CKD stage 3–5 [14]. End-stage renal disease requiring hemodialysis is the most severe form of chronic kidney disease. The survival of ESRD patients has improved in the last few years, but has not kept pace with that seen in the general population [15]. Only 57% of hemodialysis patients remain alive at 3 years, according to the recent 2017 USRDS annual data report [16].
Taiwan presents with the highest incidence of treated ESRD in the world (476/million) [16]. High 5-year cumulative incidence of any cancer for ESRD patients (9.48%) suggests that patients with ESRD are at risk of developing cancer while receiving hemodialysis [3]. The interface of lung cancer in end-stage renal disease is contradictory in the literature. Population-based studies from Asia for patients receiving chronic dialysis have demonstrated that the risk for developing lung cancer is not increased or even lower [2, 8, 9, 17]; however, reports from western countries have found higher risk for developing lung cancer [3, 5]. A meta-analysis performed by Shang et al. [18] demonstrated no higher risk of developing lung cancer risk among patients receiving dialysis with a pooled standardized incidence ratio of 0.98 (95% CI: 0.77–1.24). Ethnic differences in cancer risk could be attributed to genetic differences [17]. In Singapore, the highest smoking prevalence is observed in Malay males, but they present with a lower age-standardized lung cancer rate compared to Chinese males [17]. The differences of deletion rate of the enzyme cytochrome P450 2A6 (CYP2A6), an enzyme involved in the metabolism of tobacco carcinogens, is high among Malays (35.2%) followed by Chinese (18.3%) and Indians (7.1%) [19]. Using real-world data from Poland, Brzozowska et al. confirmed the clinical efficacy of tyrosine-kinase inhibitors (TKIs), but the survival results are lower compared to clinical studies from Asian populations [20].
The US Preventive Services Task Force recommends annual low-dose computed tomography (LDCT) screening for persons aged 55 to 80 years with a 30 pack-year smoking history, either currently smoking or having quit within 15 years [21]. The usefulness of lung cancer screening with low dose computed tomography is well established; however, the high false positive rate, the cost, and the irradiation risk still need to be addressed [22]. Unfortunately, because of the reduced life for ESRD patients to the general population, preventive cancer screening is not recommended [23]. However, the frequent medical visits by ESRD patients, in theory, should have allowed for earlier detection of malignancies. The conventional dialysis schedule for a typical ESRD patient is usually three times per week. Despite this high medical visit rate, nearly 70% of patients in our cohort presented with advanced stage lung cancer upon diagnosis. Although the likelihood of non-localized stage lung cancer for a patient receiving hemodialysis at diagnosis was not significantly different from the general population [24], the high medical visit rate did not translate into earlier detection of cancer. The reason for the delay in diagnosis merits further studies.
The survival of lung cancer with end-stage renal disease patients has conflicting results in the literature. In this study, we found that the median overall survival for WITH-ESRD patients was 7.36 months (95% CI: 5.19–11.10) compared to 12.25 months (95% CI: 10.81–14.65) for WITHOUT-ESRD patients, p = 0.133. Our result concurs with Cheung et al. [10] where the overall survival of lung cancer patients with ESRD did not differ significantly from patients without ESRD. In contrast, in a report that included only ESRD patients without a general population for comparison, Chien et al. [4] found a decreased survival rate in lung cancer-ESRD patients with a 5-year survival rate of 8% for male lung cancer-ESRD patients and 4% for female lung cancer-ESRD patients (p = 0.422), which is much lower than previously reported 16.3% at 5 years for lung cancer in Taiwan [25].
Gender survival analysis of our cohort is in agreement with previous reports on gender survival difference in lung cancer, which revealed consistently improved survival for female patients [26–30]. In this study, the median overall survival for female patients with ESRD is 12.25 months (95% CI: 5.78–17.45) comparable to the 14.13 months (95% CI: 11.07–16.43) for female patients without ESRD, p = 0.996. However, a marginally significant median survival difference was noted for male patients without ESRD 11.56 months (95% CI: 8.77–14.19) compared to 6.31 months (3.71–8.51) for male patients with ESRD (p = 0.059).
Among different treatment modalities, surgical resection is currently the treatment that offers the best chance for long-term survival. Using a population-based cancer registry for lung cancer in Taiwan, Wang et al. [31] reported a surgical resection rate of 16.4%. In this report, the resection rate for WITH-ESRD patient was 14.3%, and it concurred with the previous report. The surgical outcome of lung resection for patients under dialysis reported in several small clinical series demonstrated acceptable surgical risk and long-term outcome [32–35]. In a series of 24 dialysis patients from Japan (lobectomies: 22, wedge resection: 1 and segmentectomy: 1), 54 months median survival was reported [32]. In this report, the median overall survival for surgically treated WITH-ESRD patients was 62 months (95% CI: 15.97–84.63) compared to 50.23 months (95% CI: 29.47–96.30) for WITHOUT-ESRD patients, p = 0.781.
In our cohort, 36.1% of WITH-ESRD patients received some form of chemotherapy with poor median survival. For the medical treatment group, the median survival for WITH-ESRD patients was 5.98 months (95% CI: 4.34–11.76) compared to 14.13 months (95% CI: 11.30–16.43) for WITHOUT-ESRD patients, p = 0.019. We found that the median survival for WITH-ESRD patients receiving medical treatment was very similar to the median survival of no treatment/supportive treatment for WITHOUT-ESRD patients (5.98 months vs. 6.51 months). The poor median survival for WITH-ESRD patients receiving medical treatment could be the effect of the toxicity at higher doses, and the lack of efficacy at sub-therapeutic doses. In the CANDY study [36], only 28% of the cancer patients with dialysis received anticancer drugs; among them, 44% of the treated patients developed iatrogenic toxicity. Since clinical trials usually exclude patients on dialysis, the clinical profile and safety of a standard regimen are uncertain as well as lacking evidence of a benefit.
Radiotherapy plays an essential role in the treatment of lung cancer, from the treatment of medically inoperable early-stage lung cancer to palliative treatment for metastatic disease. In cases of brain metastasis, the combination of tyrosine-kinase inhibitors plus radiotherapy has shown greater efficacy than radiotherapy plus chemotherapy for the management of lung cancer with brain metastasis [37, 38]. However, the timing and the sequence to start the treatment are not clear. Wang et al. [39] demonstrated that the intracranial progression-free survival (iPFS) differed significantly according to the timing of using radiotherapy in combination with tyrosine-kinase inhibitors. Patients receiving concurrent TKI-radiotherapy or TKI after first-line radiotherapy had an iPFS of 11.1 months and 11.3 months compared to 8.1 months for radiotherapy after TKI failure (p = 0.032). The timing and sequence of combined modality treatment deserve further evaluation.
There are several limitations of this study, such as the retrospective design, lack of smoking status, histology grading, molecular abnormalities, absence of treatment details (possible reduction in chemotherapy dose, toxicity events), and the completion rate of the treatment plan, which may have influenced the clinical outcome. This cohort of patients was composed entirely of Chinese descendants; extrapolation of the results to other ethnic groups needs caution.
In conclusion, in our limited experience, the survival for lung cancer with ESRD is not inferior to lung cancer patients without ESRD. For physically fit patients, surgical resection provides acceptable long-term survival. The reasons for poor survival in patients with medical treatment and the late-stage diagnosis in this group of patients with frequent medical visits merit further investigation.