Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
ATHEROSCLEROSIS / RESEARCH PAPER
Beneficial Effects of Antioxidants in Regulating NET Generation Within the Course of Atherosclerosis
1
Medical Analysis Laboratory ALAB laboratory Sp. z o.o., T. Kwiatkowskiego 19, 06-102 Pułtusk,, Poland
2
Department of Immunology, Medical University of Bialystok, J. Waszyngtona 15A, 15-269 Bialystok,, Poland
3
Department of Regenerative Medicine and Immune Regulation, Medical University of Bialystok, J. Waszyngtona 13, 15-269, Bialystok,, Poland
4
Regional Centre for Transfusion Medicine, Bialystok, M. Skłodowskiej-Curie 23, 15-950, Bialystok,, Poland
5
Department of Biology and Pathology of Human Reproduction, Institute of Animal Reproduction and Food Research, Polish Academy of Sciences, Olsztyn, M. Skłodowskiej-Curie 24a, 15-276, Bialystok,, Poland
6
Department of Reproduction and Gynecological Endocrinology, Medical University of Bialystok, M. Skłodowskiej-Curie 24 A, 15-276 Bialystok,, Poland
Submission date: 2025-07-15
Final revision date: 2025-10-01
Acceptance date: 2025-10-29
Publication date: 2025-11-28
Corresponding author
Dorota Dąbrowska-Zagroba
Medical Analysis Laboratory ALAB laboratory Sp. z o.o., T. Kwiatkowskiego 19, 06-102 Pułtusk,, Poland
Medical Analysis Laboratory ALAB laboratory Sp. z o.o., T. Kwiatkowskiego 19, 06-102 Pułtusk,, Poland
Arch Med Sci 2026;22(3)
KEYWORDS
TOPICS
ABSTRACT
Introduction:
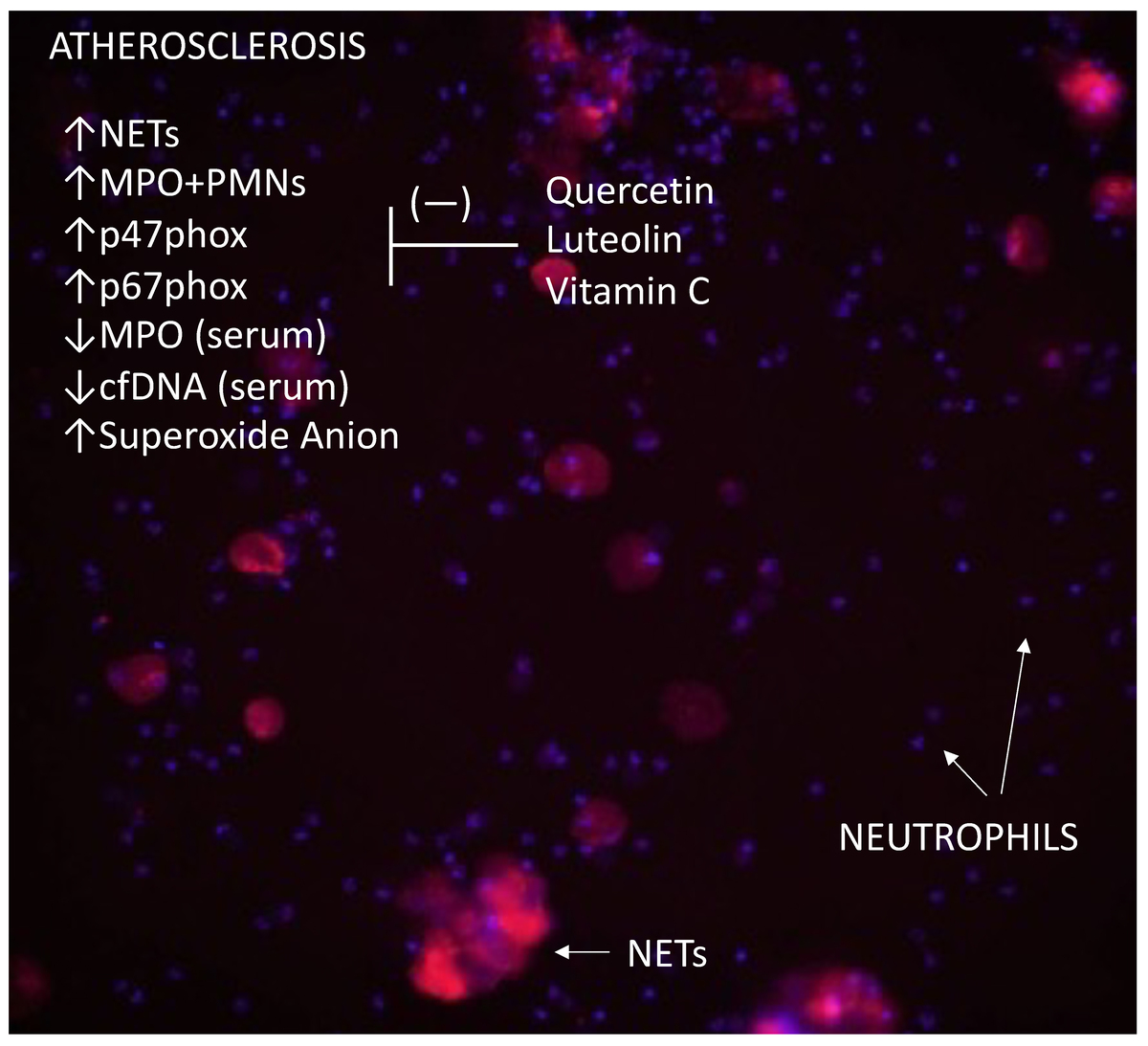
There are numerous publications confirming the involvement of neutrophils in the pathomechanism of atherosclerosis. The aim of this study was to determine the effects of flavonoids (quercetin and luteolin) and vitamin C on neutrophil extracellular trap (NET) formation and regulation of NET markers, including circulating free DNA (cfDNA) and myeloperoxidase (MPO). Additionally, we assessed the expression of NADPH oxidase complex subunits and superoxide anion generation in neutrophils.
Material and methods:
Whole blood samples were collected from patients with atherosclerosis. Neutrophils were stimulated with lipopolysaccharide (LPS), and the effects of flavonoids and vitamin C on NET release, NADPH oxidase subunit expression (p47phox, p67phox, Rac1), and superoxide production were evaluated.
Results:
Atherosclerotic patients showed increased NETosis, with elevated NET release, MPO, and cfDNA levels. Expression of NADPH oxidase subunits p47phox and p67phox, as well as superoxide anion generation, were significantly higher compared to controls. LPS further enhanced these effects. Treatment with quercetin, luteolin, and vitamin C reduced NET formation, NADPH oxidase expression, and superoxide production in stimulated neutrophils.
Conclusions:
Flavonoids and vitamin C modulate NET generation by affecting NADPH oxidase activity and reactive oxygen species production, suggesting potential therapeutic use in atherosclerosis. Further and more detailed analysis could allow for the potential application of these compounds as adjunctive therapy in the treatment of atherosclerosis.
There are numerous publications confirming the involvement of neutrophils in the pathomechanism of atherosclerosis. The aim of this study was to determine the effects of flavonoids (quercetin and luteolin) and vitamin C on neutrophil extracellular trap (NET) formation and regulation of NET markers, including circulating free DNA (cfDNA) and myeloperoxidase (MPO). Additionally, we assessed the expression of NADPH oxidase complex subunits and superoxide anion generation in neutrophils.
Material and methods:
Whole blood samples were collected from patients with atherosclerosis. Neutrophils were stimulated with lipopolysaccharide (LPS), and the effects of flavonoids and vitamin C on NET release, NADPH oxidase subunit expression (p47phox, p67phox, Rac1), and superoxide production were evaluated.
Results:
Atherosclerotic patients showed increased NETosis, with elevated NET release, MPO, and cfDNA levels. Expression of NADPH oxidase subunits p47phox and p67phox, as well as superoxide anion generation, were significantly higher compared to controls. LPS further enhanced these effects. Treatment with quercetin, luteolin, and vitamin C reduced NET formation, NADPH oxidase expression, and superoxide production in stimulated neutrophils.
Conclusions:
Flavonoids and vitamin C modulate NET generation by affecting NADPH oxidase activity and reactive oxygen species production, suggesting potential therapeutic use in atherosclerosis. Further and more detailed analysis could allow for the potential application of these compounds as adjunctive therapy in the treatment of atherosclerosis.
REFERENCES (56)
2.
Netala VR, Teertam SK, Li H, Zhang Z. A Comprehensive Review of Cardiovascular Disease Management: Cardiac Biomarkers, Imaging Modalities, Pharmacotherapy, Surgical Interventions, and Herbal Remedies. Cells. 2024;13(17):1471. doi:10.3390/cells13171471.
3.
Ozdemir E, Stavileci B, Ozdemir B, et al. The association between growth differentiation factor 15 and presence and severity of coronary atherosclerosis. Adv Med Sci. 2024;69(1):56-60. doi:10.1016/j.advms.2024.02.003.
4.
Ionita MG, van den Borne P, Catanzariti LM, et al. High neutrophil numbers in human carotid atherosclerotic plaques are associated with characteristics of rupture-prone lesions. Arterioscler Thromb Vasc Biol. 2010;30(9):1842-1848. doi:10.1161/ATVBAHA.110.209296.
5.
Baetta R, Corsini A. Role of polymorphonuclear neutrophils in atherosclerosis: current state and future perspectives. Atherosclerosis. 2010;210(1):1-13. doi:10.1016/j.atherosclerosis.2009.10.028.
6.
Brinkmann V, Reichard U, Goosmann C, et al. Neutrophil extracellular traps kill bacteria. Science. 2004;303(5663):1532-1535. doi:10.1126/science.1092385.
7.
Dwivedi N, Neeli I, Schall N, et al. Deimination of linker histones links neutrophil extracellular trap release with autoantibodies in systemic autoimmunity. FASEB J Off Publ Fed Am Soc Exp Biol. 2014;28(7):2840-2851. doi:10.1096/fj.13-247254.
8.
Urban CF, Ermert D, Schmid M, et al. Neutrophil extracellular traps contain calprotectin, a cytosolic protein complex involved in host defense against Candida albicans. PLoS Pathog. 2009;5(10):e1000639. doi:10.1371/journal.ppat.1000639.
9.
Jaillon S, Peri G, Delneste Y, et al. The humoral pattern recognition receptor PTX3 is stored in neutrophil granules and localizes in extracellular traps. J Exp Med. 2007;204(4):793-804. doi:10.1084/jem.20061301.
10.
Wartha F, Beiter K, Normark S, Henriques-Normark B. Neutrophil extracellular traps: casting the NET over pathogenesis. Curr Opin Microbiol. 2007;10(1):52-56. doi:10.1016/j.mib.2006.12.005.
11.
Urban CF, Reichard U, Brinkmann V, Zychlinsky A. Neutrophil extracellular traps capture and kill Candida albicans yeast and hyphal forms. Cell Microbiol. 2006;8(4):668-676. doi:10.1111/j.1462-5822.2005.00659.x.
12.
Fuchs TA, Abed U, Goosmann C, et al. Novel cell death program leads to neutrophil extracellular traps. J Cell Biol. 2007;176(2):231-241. doi:10.1083/jcb.200606027.
13.
Hakkim A, Fuchs TA, Martinez NE, et al. Activation of the Raf-MEK-ERK pathway is required for neutrophil extracellular trap formation. Nat Chem Biol. 2011;7(2):75-77. doi:10.1038/nchembio.496.
14.
McCormick A, Heesemann L, Wagener J, et al. NETs formed by human neutrophils inhibit growth of the pathogenic mold Aspergillus fumigatus. Microbes Infect. 2010;12(12-13):928-936. doi:10.1016/j.micinf.2010.06.009.
15.
Abi Abdallah DS, Lin C, Ball CJ, King MR, Duhamel GE, Denkers EY. Toxoplasma gondii triggers release of human and mouse neutrophil extracellular traps. Infect Immun. 2012;80(2):768-777. doi:10.1128/IAI.05730-11.
16.
Guimarães-Costa AB, Nascimento MTC, Froment GS, et al. Leishmania amazonensis promastigotes induce and are killed by neutrophil extracellular traps. Proc Natl Acad Sci U S A. 2009;106(16):6748-6753. doi:10.1073/pnas.0900226106.
17.
Saitoh T, Komano J, Saitoh Y, et al. Neutrophil extracellular traps mediate a host defense response to human immunodeficiency virus-1. Cell Host Microbe. 2012;12(1):109-116. doi:10.1016/j.chom.2012.05.015.
18.
Narasaraju T, Yang E, Samy RP, et al. Excessive neutrophils and neutrophil extracellular traps contribute to acute lung injury of influenza pneumonitis. Am J Pathol. 2011;179(1):199-210. doi:10.1016/j.ajpath.2011.03.013.
19.
Kaplan MJ, Radic M. Neutrophil extracellular traps: double-edged swords of innate immunity. J Immunol Baltim Md 1950. 2012;189(6):2689-2695. doi:10.4049/jimmunol.1201719.
20.
Shi D, Huang J, Wu J. Down-regulation of SHP2 promotes neutrophil autophagy and inhibits neutrophil extracellular trap formation to alleviate asthma through the ERK5 pathway. Cent Eur J Immunol. 2024;49(1):252-272. doi:10.5114/ceji.2024.143691.
21.
Dąbrowska D, Jabłońska E, Garley M, Sawicka-Powierza J, Nowak K. The Phenomenon of Neutrophil Extracellular Traps in Vascular Diseases. Arch Immunol Ther Exp (Warsz). 2018;66(4):273-281. doi:10.1007/s00005-018-0505-y.
22.
Knight JS, Luo W, O’Dell AA, et al. Peptidylarginine deiminase inhibition reduces vascular damage and modulates innate immune responses in murine models of atherosclerosis. Circ Res. 2014;114(6):947-956. doi:10.1161/CIRCRESAHA.114.303312.
23.
Bhuyan KC, Bhuyan DK. Superoxide dismutase of the eye Relative functions of superoxide dismutase and catalase in protecting the ocular lens from oxidative damage. Biochim Biophys Acta BBA - Gen Subj. 1978;542(1):28-38. doi:10.1016/0304-4165(78)90229-5.
24.
Manda A, Pruchniak MP, Araźna M, Demkow UA. Neutrophil extracellular traps in physiology and pathology. Cent-Eur J Immunol. 2014;39(1):116-121. doi:10.5114/ceji.2014.42136.
25.
Megens RTA, Vijayan S, Lievens D, et al. Presence of luminal neutrophil extracellular traps in atherosclerosis. Thromb Haemost. 2012;107(3):597-598. doi:10.1160/TH11-09-0650.
26.
Garley M, Omeljaniuk WJ, Motkowski R, et al. Immunoaging – the effect of age on serum levels of NET biomarkers in men: a pilot study. Int J Occup Med Environ Health. 2023;36(3):333-348. doi:10.13075/ijomeh.1896.02125.
27.
Hazell LJ, Stocker R. Oxidation of low-density lipoprotein with hypochlorite causes transformation of the lipoprotein into a high-uptake form for macrophages. Biochem J. 1993;290 ( Pt 1)(Pt 1):165-172. doi:10.1042/bj2900165.
28.
Zhang R, Brennan ML, Fu X, et al. Association between myeloperoxidase levels and risk of coronary artery disease. JAMA. 2001;286(17):2136-2142. doi:10.1001/jama.286.17.2136.
29.
Brennan ML, Penn MS, Van Lente F, et al. Prognostic value of myeloperoxidase in patients with chest pain. N Engl J Med. 2003;349(17):1595-1604. doi:10.1056/NEJMoa035003.
30.
Parker H, Dragunow M, Hampton MB, Kettle AJ, Winterbourn CC. Requirements for NADPH oxidase and myeloperoxidase in neutrophil extracellular trap formation differ depending on the stimulus. J Leukoc Biol. 2012;92(4):841-849. doi:10.1189/jlb.1211601.
31.
Palmer LJ, Cooper PR, Ling MR, Wright HJ, Huissoon A, Chapple ILC. Hypochlorous acid regulates neutrophil extracellular trap release in humans. Clin Exp Immunol. 2012;167(2):261-268. doi:10.1111/j.1365-2249.2011.04518.x.
32.
Metzler KD, Fuchs TA, Nauseef WM, et al. Myeloperoxidase is required for neutrophil extracellular trap formation: implications for innate immunity. Blood. 2011;117(3):953-959. doi:10.1182/blood-2010-06-290171.
33.
Jylhävä J, Lehtimäki T, Jula A, et al. Circulating cell-free DNA is associated with cardiometabolic risk factors: the Health 2000 Survey. Atherosclerosis. 2014;233(1):268-271. doi:10.1016/j.atherosclerosis.2013.12.022.
34.
Gould TJ, Lysov Z, Liaw PC. Extracellular DNA and histones: double-edged swords in immunothrombosis. J Thromb Haemost JTH. 2015;13 Suppl 1:S82-91. doi:10.1111/jth.12977.
35.
Borissoff JI, Joosen IA, Versteylen MO, et al. Elevated levels of circulating DNA and chromatin are independently associated with severe coronary atherosclerosis and a prothrombotic state. Arterioscler Thromb Vasc Biol. 2013;33(8):2032-2040. doi:10.1161/ATVBAHA.113.301627.
36.
Fuchs TA, Brill A, Duerschmied D, et al. Extracellular DNA traps promote thrombosis. Proc Natl Acad Sci U S A. 2010;107(36):15880-15885. doi:10.1073/pnas.1005743107.
37.
Gupta AK, Joshi MB, Philippova M, et al. Activated endothelial cells induce neutrophil extracellular traps and are susceptible to NETosis-mediated cell death. FEBS Lett. 2010;584(14):3193-3197. doi:10.1016/j.febslet.2010.06.006.
38.
Warnatsch A, Ioannou M, Wang Q, Papayannopoulos V. Inflammation. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science. 2015;349(6245):316-320. doi:10.1126/science.aaa8064.
39.
Döring Y, Manthey HD, Drechsler M, et al. Auto-antigenic protein-DNA complexes stimulate plasmacytoid dendritic cells to promote atherosclerosis. Circulation. 2012;125(13):1673-1683. doi:10.1161/CIRCULATIONAHA.111.046755.
40.
Bianchi M, Hakkim A, Brinkmann V, et al. Restoration of NET formation by gene therapy in CGD controls aspergillosis. Blood. 2009;114(13):2619-2622. doi:10.1182/blood-2009-05-221606.
41.
Lim MBH, Kuiper JWP, Katchky A, Goldberg H, Glogauer M. Rac2 is required for the formation of neutrophil extracellular traps. J Leukoc Biol. 2011;90(4):771-776. doi:10.1189/jlb.1010549.
42.
Röhm M, Grimm MJ, D’Auria AC, Almyroudis NG, Segal BH, Urban CF. NADPH oxidase promotes neutrophil extracellular trap formation in pulmonary aspergillosis. Infect Immun. 2014;82(5):1766-1777. doi:10.1128/IAI.00096-14.
43.
Violi F, Basili S, Nigro C, Pignatelli P. Role of NADPH oxidase in atherosclerosis. Future Cardiol. 2009;5(1):83-92. doi:10.2217/14796678.5.1.83.
44.
Levy R, Schlaeffer F. Successful treatment of a patient with recurrent furunculosis by vitamin C: improvement of clinical course and of impaired neutrophil functions. Int J Dermatol. 1993;32(11):832-834. doi:10.1111/j.1365-4362.1993.tb02780.x.
45.
Vohra K, Khan AJ, Telang V, Rosenfeld W, Evans HE. Improvement of neutrophil migration by systemic vitamin C in neonates. J Perinatol Off J Calif Perinat Assoc. 1990;10(2):134-136.
46.
de la Fuente M, Ferrández MD, Burgos MS, Soler A, Prieto A, Miquel J. Immune function in aged women is improved by ingestion of vitamins C and E. Can J Physiol Pharmacol. 1998;76(4):373-380.
47.
Bozonet SM, Carr AC, Pullar JM, Vissers MCM. Enhanced human neutrophil vitamin C status, chemotaxis and oxidant generation following dietary supplementation with vitamin C-rich SunGold kiwifruit. Nutrients. 2015;7(4):2574-2588. doi:10.3390/nu7042574.
48.
Mohammed BM, Fisher BJ, Kraskauskas D, et al. Vitamin C: a novel regulator of neutrophil extracellular trap formation. Nutrients. 2013;5(8):3131-3151. doi:10.3390/nu5083131.
49.
Kirchner T, Hermann E, Möller S, et al. Flavonoids and 5-aminosalicylic acid inhibit the formation of neutrophil extracellular traps. Mediators Inflamm. 2013;2013:710239. doi:10.1155/2013/710239.
50.
Santos EOL, Kabeya LM, Figueiredo-Rinhel ASG, et al. Flavonols modulate the effector functions of healthy individuals’ immune complex-stimulated neutrophils: a therapeutic perspective for rheumatoid arthritis. Int Immunopharmacol. 2014;21(1):102-111. doi:10.1016/j.intimp.2014.04.014.
51.
Liu G, Wang W, Masuoka N, et al. Effect of three flavonoids isolated from Japanese Polygonum species on superoxide generation in human neutrophils. Planta Med. 2005;71(10):933-937. doi:10.1055/s-2005-871283.
52.
Lu HW, Sugahara K, Sagara Y, et al. Effect of three flavonoids, 5,7,3’,4’-tetrahydroxy-3-methoxy flavone, luteolin, and quercetin, on the stimulus-induced superoxide generation and tyrosyl phosphorylation of proteins in human neutrophil. Arch Biochem Biophys. 2001;393(1):73-77. doi:10.1006/abbi.2001.2457.
53.
Lu J, Feng X, Sun Q, et al. Effect of six flavonoid compounds from Ixeris sonchifolia on stimulus-induced superoxide generation and tyrosyl phosphorylation in human neutrophils. Clin Chim Acta Int J Clin Chem. 2002;316(1-2):95-99. doi:10.1016/s0009-8981(01)00725-2.
54.
Yang SC, Chen PJ, Chang SH, et al. Luteolin attenuates neutrophilic oxidative stress and inflammatory arthritis by inhibiting Raf1 activity. Biochem Pharmacol. 2018;154:384-396. doi:10.1016/j.bcp.2018.06.003.
55.
Pečivová J, Mačičková T, Sviteková K, Nosáľ R. Quercetin inhibits degranulation and superoxide generation in PMA stimulated neutrophils. Interdiscip Toxicol. 2012;5(2):81-83. doi:10.2478/v10102-012-0014-5.
56.
Ross JA, Kasum CM. Dietary flavonoids: bioavailability, metabolic effects, and safety. Annu Rev Nutr. 2002;22:19-34. doi:10.1146/annurev.nutr.22.111401.144957.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.