Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
PUBLIC HEALTH / RESEARCH PAPER
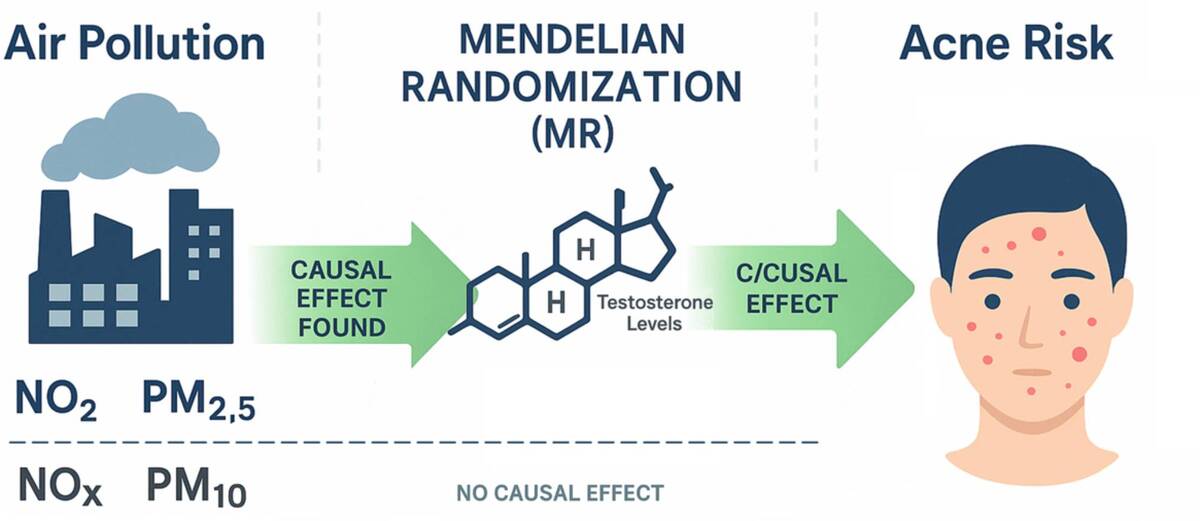
Causal Relationships Among Air Pollution, Testosterone Levels, and Acne: A Two-Sample Mendelian Randomization Study
1
The First Hospital of Hebei Medical University, China
2
Hebei Academy of Chinese Medicine Sciences, China
Submission date: 2025-05-26
Final revision date: 2025-07-28
Acceptance date: 2025-09-05
Online publication date: 2025-11-28
KEYWORDS
single nucleotide polymorphismair pollutionacnetestosterone levelstwo‐sample Mendelian randomization
TOPICS
ABSTRACT
Introduction:
This study aimed to investigate the causal relationships between air pollution, testosterone levels, and acne using a Mendelian randomization (MR) approach. Air pollution exposure, including nitrogen oxides (Nitrogen oxides air pollution, NOAP), particulate matter (PM2.5 absorbance, nitrogen dioxide (Nitrogen dioxide air pollution, NDAP), PM2.5, and PM10), was analyzed in relation to total testosterone (TTL) and bioavailable testosterone levels (BTL). Given the potential role of environmental and hormonal factors in acne pathogenesis, this study sought to clarify their causal contributions.
Material and methods:
This two-sample Mendelian randomization (MR) study was performed by applying genome-wide association study (GWAS) summary statistics. Multiple MR methods, including inverse variance weighted (IVW), MR-Egger, weighted median (WM), and weighted mode, were applied to assess causality. Sensitivity analyses, such as MR-Egger regression, WM, and MR-PRESSO, were conducted to evaluate pleiotropy and heterogeneity.
Results:
Genetically predicted NDAP and PM2.5 exposure exhibited significant causal effects on both TTL and BTL, which in turn were associated with acne development. However, NOAP, PM2.5-10, and PM10 showed no causal links with acne. Sensitivity analyses confirmed the robustness of the IVW results, with no substantial evidence of pleiotropy or heterogeneity.
Conclusions:
This MR study provides evidence supporting a causal role of NDAP and PM2.5 in influencing testosterone levels and acne risk. These findings underscore the importance of environmental factors, particularly air pollution, in modulating hormonal pathways involved in acne. Future research should explore mechanisms underlying these associations,while public health strategies should consider reducing air pollution exposure as part of acne prevention and management approaches.
This study aimed to investigate the causal relationships between air pollution, testosterone levels, and acne using a Mendelian randomization (MR) approach. Air pollution exposure, including nitrogen oxides (Nitrogen oxides air pollution, NOAP), particulate matter (PM2.5 absorbance, nitrogen dioxide (Nitrogen dioxide air pollution, NDAP), PM2.5, and PM10), was analyzed in relation to total testosterone (TTL) and bioavailable testosterone levels (BTL). Given the potential role of environmental and hormonal factors in acne pathogenesis, this study sought to clarify their causal contributions.
Material and methods:
This two-sample Mendelian randomization (MR) study was performed by applying genome-wide association study (GWAS) summary statistics. Multiple MR methods, including inverse variance weighted (IVW), MR-Egger, weighted median (WM), and weighted mode, were applied to assess causality. Sensitivity analyses, such as MR-Egger regression, WM, and MR-PRESSO, were conducted to evaluate pleiotropy and heterogeneity.
Results:
Genetically predicted NDAP and PM2.5 exposure exhibited significant causal effects on both TTL and BTL, which in turn were associated with acne development. However, NOAP, PM2.5-10, and PM10 showed no causal links with acne. Sensitivity analyses confirmed the robustness of the IVW results, with no substantial evidence of pleiotropy or heterogeneity.
Conclusions:
This MR study provides evidence supporting a causal role of NDAP and PM2.5 in influencing testosterone levels and acne risk. These findings underscore the importance of environmental factors, particularly air pollution, in modulating hormonal pathways involved in acne. Future research should explore mechanisms underlying these associations,while public health strategies should consider reducing air pollution exposure as part of acne prevention and management approaches.
REFERENCES (50)
1.
Chen B, Zheng Y, Liang Y. Analysis of Potential Genes and Pathways Involved in the Pathogenesis of Acne by Bioinformatics. Biomed Res Int 2019; 2019: 3739086.
2.
Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol 2014; 134: 1527-34.
3.
Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2163-96.
4.
Bernardis E, Shou H, Barbieri JS, et al. Development and Initial Validation of a Multidimensional Acne Global Grading System Integrating Primary Lesions and Secondary Changes. JAMA Dermatol 2020; 156: 296-302.
5.
Sutaria AH, Masood S, Saleh HM, Schlessinger J. Acne Vulgaris. StatPearls. Treasure Island (FL): StatPearls Publishing.
7.
Heng AHS, Chew FT. Systematic review of the epidemiology of acne vulgaris. Sci Rep 2020; 10: 5754.
8.
Kim KE, Cho D, Park HJ. Air pollution and skin diseases: Adverse effects of airborne particulate matter on various skin diseases. Life Sci 2016; 152: 126-34.
9.
Ramos F, Trilles S, Muñoz A, Huerta J. Promoting Pollution-Free Routes in Smart Cities Using Air Quality Sensor Networks. Sensors (Basel) 2018; 18.
10.
Piscitelli P, Valenzano B, Rizzo E, et al. Air Pollution and Estimated Health Costs Related to Road Transportations of Goods in Italy: A First Healthcare Burden Assessment. Int J Environ Res Public Health 2019; 16.
11.
Kelishadi R, Poursafa P. Air pollution and non-respiratory health hazards for children. Arch Med Sci 2010; 6: 483-95.
12.
Morgenstern V, Zutavern A, Cyrys J, et al. Atopic diseases, allergic sensitization, and exposure to traffic-related air pollution in children. Am J Respir Crit Care Med 2008; 177: 1331-7.
13.
Dijkhoff IM, Drasler B, Karakocak BB, et al. Impact of airborne particulate matter on skin: a systematic review from epidemiology to in vitro studies. Part Fibre Toxicol 2020; 17: 35.
14.
Ferrara F, Prieux R, Woodby B, Valacchi G. Inflammasome Activation in Pollution-Induced Skin Conditions. Plast Reconstr Surg 2021; 147: 15s-24s.
15.
Fussell JC, Kelly FJ. Oxidative contribution of air pollution to extrinsic skin ageing. Free Radic Biol Med 2020; 151: 111-22.
16.
Valacchi G, Sticozzi C, Pecorelli A, Cervellati F, Cervellati C, Maioli E. Cutaneous responses to environmental stressors. Ann N Y Acad Sci 2012; 1271: 75-81.
17.
Leccia MT, Auffret N, Poli F, Claudel JP, Corvec S, Dreno B. Topical acne treatments in Europe and the issue of antimicrobial resistance. J Eur Acad Dermatol Venereol 2015; 29: 1485-92.
18.
Chilicka K, Dzieńdziora-Urbińska I, Szyguła R, Asanova B, Nowicka D. Microbiome and Probiotics in Acne Vulgaris-A Narrative Review. Life (Basel) 2022; 12.
19.
Chilicka K, Rogowska AM, Szyguła R, Dzieńdziora-Urbińska I, Taradaj J. A comparison of the effectiveness of azelaic and pyruvic acid peels in the treatment of female adult acne: a randomized controlled trial. Sci Rep 2020; 10: 12612.
20.
Zhang XD, Sun J, Zheng XM, et al. Plin4 exacerbates cadmium-decreased testosterone level via inducing ferroptosis in testicular Leydig cells. Redox Biol 2024; 76: 103312.
21.
Greenland S. An introduction to instrumental variables for epidemiologists. Int J Epidemiol 2018; 47: 358.
22.
Ference BA, Holmes MV, Smith GD. Using Mendelian Randomization to Improve the Design of Randomized Trials. Cold Spring Harb Perspect Med 2021; 11.
23.
Jiang R, Qu Q, Wang Z, Luo F, Mou S. Association between air pollution and bone mineral density: a Mendelian randomization study. Arch Med Sci 2024; 20: 1334-8.
24.
Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol 2016; 40: 304-14.
25.
Bycroft C, Freeman C, Petkova D, et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 2018; 562: 203-9.
26.
Ruth KS, Day FR, Tyrrell J, et al. Using human genetics to understand the disease impacts of testosterone in men and women. Nat Med 2020; 26: 252-8.
27.
Huang SJ, Li R, Xu S, Liu Y, Li SH, Duan SZ. Assessment of bidirectional relationships between circulating cytokines and periodontitis: Insights from a mendelian randomization analysis. Front Genet 2023; 14: 1124638.
28.
Wang S, Lai J, Wu R, et al. Grocott Methenamine Silver Staining Is the Optimal Approach to Histological Diagnosis of Pulmonary Cryptococcosis. Front Microbiol 2022; 13: 885511.
29.
Palmer TM, Lawlor DA, Harbord RM, et al. Using multiple genetic variants as instrumental variables for modifiable risk factors. Stat Methods Med Res 2012; 21: 223-42.
30.
Burgess S, Small DS, Thompson SG. A review of instrumental variable estimators for Mendelian randomization. Stat Methods Med Res 2017; 26: 2333-55.
31.
Wang X, Chen Z, Tian W, et al. Plasma homocysteine levels and risk of congestive heart failure or cardiomyopathy: A Mendelian randomization study. Front Cardiovasc Med 2023; 10: 1030257.
32.
Burgess S, Butterworth A, Thompson SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol 2013; 37: 658-65.
33.
Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 2015; 44: 512-25.
34.
Verbanck M, Chen CY, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet 2018; 50: 693-8.
35.
Hartwig FP, Davey Smith G, Bowden J. Robust inference in summary data Mendelian randomization via the zero modal pleiotropy assumption. Int J Epidemiol 2017; 46: 1985-98.
36.
Papadimitriou N, Dimou N, Tsilidis KK, et al. Physical activity and risks of breast and colorectal cancer: a Mendelian randomisation analysis. Nat Commun 2020; 11: 597.
37.
Liu W, Pan X, Vierkötter A, et al. A Time-Series Study of the Effect of Air Pollution on Outpatient Visits for Acne Vulgaris in Beijing. Skin Pharmacol Physiol 2018; 31: 107-13.
38.
Lefebvre MA, Pham DM, Boussouira B, Bernard D, Camus C, Nguyen QL. Evaluation of the impact of urban pollution on the quality of skin: a multicentre study in Mexico. Int J Cosmet Sci 2015; 37: 329-38.
39.
Lefebvre MA, Pham DM, Boussouira B, et al. Consequences of urban pollution upon skin status. A controlled study in Shanghai area. Int J Cosmet Sci 2016; 38: 217-23.
40.
Krutmann J, Moyal D, Liu W, et al. Pollution and acne: is there a link? Clin Cosmet Investig Dermatol 2017; 10: 199-204.
41.
Pham DM, Boussouira B, Moyal D, Nguyen QL. Oxidization of squalene, a human skin lipid: a new and reliable marker of environmental pollution studies. Int J Cosmet Sci 2015; 37: 357-65.
42.
Jin H, Lin Z, Pang T, et al. Effects and mechanisms of polycyclic aromatic hydrocarbons in inflammatory skin diseases. Sci Total Environ 2024; 925: 171492.
43.
Tsuji G, Takahara M, Uchi H, et al. An environmental contaminant, benzo(a)pyrene, induces oxidative stress-mediated interleukin-8 production in human keratinocytes via the aryl hydrocarbon receptor signaling pathway. J Dermatol Sci 2011; 62: 42-9.
44.
Housman E, Reynolds RV. Polycystic ovary syndrome: a review for dermatologists: Part I. Diagnosis and manifestations. J Am Acad Dermatol 2014; 71: 847.e1-.e10; quiz 57-8.
45.
Schmidt TH, Shinkai K. Evidence-based approach to cutaneous hyperandrogenism in women. J Am Acad Dermatol 2015; 73: 672-90.
46.
Oge LK, Broussard A, Marshall MD. Acne Vulgaris: Diagnosis and Treatment. Am Fam Physician 2019; 100: 475-84.
47.
Sato T, Shibata F, Koiwai T, Akimoto N. Different regulation of lipogenesis in sebocytes and subcutaneous preadipocytes in hamsters in vitro. Biochem Biophys Rep 2020; 22: 100761.
48.
Yunoki T, Miyakoshi A, Otsuka M, Hayashi A. Clinicopathological features of considerable reduction in androgen receptor expression in sebaceous gland carcinoma of the eyelid. Int Ophthalmol 2019; 39: 1703-8.
49.
Ju Q, Tao T, Hu T, Karadağ AS, Al-Khuzaei S, Chen W. Sex hormones and acne. Clin Dermatol 2017; 35: 130-7.
50.
da Cunha MG, Fonseca FL, Machado CD. Androgenic hormone profile of adult women with acne. Dermatology 2013; 226: 167-71.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.