Introduction
Prostate cancer (PCa) is one of the most common malignant tumours in the male genitourinary system worldwide [1]. In China, PCa is the seventh most commonly diagnosed cancer in men, reflecting that approximately 60,300 cases are newly diagnosed, and about 26,600 deaths occurred in 2015 [2]. At the earlier stage of PCa patients with castration-sensitive, androgen deprivation therapy is the preferred treatment strategy, while castration-resistance inevitably occurs in advanced PCa patients accompanied by cancer metastasis, which is associated with poor prognosis [3]. More importantly, several oncogenic pathway alterations, including androgen receptor, epidermal growth factor receptor, mitogen-activated protein kinase/phosphoinositide 3-kinase, and signal transducer, and activator of transcription 3, may be reasonable for PCa with castration resistance [4].
Cyclin-dependent kinase 8 (CDK8), as a primary transcriptional member of the CDK family, oversees diverse biological functions by phosphorylation of transcription factors, which can regulate gene expression [5]. The overriding concern in the function of CDK8 has conferred an oncogene status in various cancer types, including colorectal cancer (CRC), breast cancer, melanoma, acute myeloid leukaemia, and prostate cancer [6–10]. In fact, CDK8 is overexpressed in a large fraction of CRC and breast cancer tissues [6, 11]. Compared with normal tissues, CDK8 protein expression has no obvious change in primary prostate carcinoma samples but is dramatically elevated in metastatic and castration-resistant PCa tissues [10]. The knockdown of CDK8 or treatment with CDK8 inhibitor has the ability to repress migration and invasion and induce apoptosis and cell cycle arrest in PCa cells [10, 12].
On the other hand, several studies have shown that inhibition of CDK8 by microRNAs (miRs), which are representative of the class of single-stranded non-coding RNAs, consisting of approximately 22 nucleotides and play crucial roles in post-transcriptional gene silencing mechanism by binding with 3’-UTRs of targeted genes [13, 14], inhibits proliferation and induces apoptosis in glioma, papillary thyroid carcinomas, and laryngeal squamous cell carcinoma [15–17]. However, few reports focus on the interaction between miRs and CDK8 in the initiation and progression of PCa.
In our study, we found that CDK8 was a direct target of miR-216a-5p. Subsequently, the expression levels of CDK8 and miR-216a-5p were investigated in PCa tissues and cell lines. We also evaluated the prognostic significance of CDK8 and miR-216a-5p of PCa patients. Furthermore, we explored the post-transcriptional regulatory mechanism of miR-216a-5p in PCa cell proliferation and apoptosis via suppression of CDK8 signalling pathway.
Material and methods
Patient recruitment and specimen collection
A total of 22 benign prostate samples and 55 pairs of PCa tissues and adjacent non-tumourous tissues were collected from 22 patients with benign prostate hyperplasia and 55 patients with PCa, who had undergone surgery in Taizhou Hospital Affiliated to Wenzhou Medical University (Linhai, China). The Ethics Committee of the Taizhou Hospital Affiliated to Wenzhou Medical University (Linhai, China) approved our study.
Cell culture and transfection
PC3 and DU145 cells were purchased from ATCC and cultured in DMEM (Invitrogen) as described previously [18]. Short hairpin RNA (shRNA) was designed to specifically target CDK8. Sh-CDK8 and sh-Con were synthesised by GenePharma (Shanghai, China). CDK8 overexpressed plasmids were structured and transfected as described previously [19]. The sequence of miR-216a-5p mimics and miR-Con were 5’-UAAUCUCAGCUGGCAACUGUGA-3’ and 5’-UCUGCCUGAUCGUAGCGGAUCG-3’, respectively, which were synthesised by Guangzhou RiboBio Co., Ltd. (Guangzhou, China) and transfected into PC3 and DU145 cells using Lipofectamine® 2000 (Invitrogen) for 48 h at 37˚C.
Migration and invasion assays
The transwell migration and invasion assay were carried out as described previously [20]. Transwell plates (8 µm pore size) without Matrigel were used for Transwell migration assay, and Transwell plates (8-µm pore size) with Matrigel were performed for Transwell invasion assay.
RT-qPCR
Total RNA was extracted using the miRNeasy Mini Kit (Qiagen). TaqMan® RT kit (Applied Biosystems; Thermo Fisher Scientific, Inc.) and TaqMan® MicroRNA assay (Applied Biosystems; Thermo Fisher Scientific, Inc.) were used to perform RT-qPCR of miR-216a-5p. U6 small nuclear RNA was used as an endogenous control. The primers were synthesised by Sangon Biotech (Shanghai, China) as follows: forward primer 5’-UAAUCUCAGCUGGCAACUGUGA-3’ and reverse primer 5’-ACAGUUGCCAGCUGAGAUUAUU-3’ for miR-216a-5p; forward primer 5’-CGCTTCGGCAGCACATATACTAA-3’ and reverse primer 5’-TATGGAACGCTTCACGAATTTGC-3’ for U6.
Moloney murine leukaemia virus reverse transcriptase (Invitrogen) and TaqMan® Universal PCR Master Mix (Thermo Fisher Scientific, Inc.) were used to perform RT-qPCR of CDK8 using the Applied Biosystems 7300 Real-Time PCR system. The primers were synthesised by Sangon Biotech (Shanghai, China) as follows: forward primer 5’-GCCGGTTGTCAAATCCCTTAC-3’ and reverse primer 5’-TGTGACTGCTGTCTTGATTCCCT-3’ for CDK8; forward primer 5’-GCACCGTCAAGCTGAGAAC-3’ and reverse primer 5’-TGGTGAAGACGCCAGTGGA-3’ for GAPDH.
Immunohistochemical (IHC) staining and western blotting
IHC staining and result analysis were performed as previously described [21]. CDK8 primary antibody was purchased from Santa Cruz Biotechnology Inc., (dilution: 1 : 500; Dallas, TX, USA). The IHC staining was visualised using a microscope (Leica DM 2500). Total protein extraction, western blotting, and results analysis were conducted as described previously [21]. CDK8 (dilution: 1 : 500), β-catenin (dilution: 1 : 500), cyclin D1 (dilution: 1 : 2000), c-Myc (dilution: 1 : 1000), p-GSK-3β (dilution: 1 : 1000), and GSK-3β (dilution: 1 : 2000) primary antibody was purchased from Santa Cruz Biotechnology, Inc., Dallas, TX, USA.
Cell proliferation, colony formation assay, and apoptosis
Cell proliferation was measured using the MTT Cell Proliferation/Viability Assay kit (R&D Systems, Inc., Minneapolis, MN, USA) or colony formation assays according to the manufacturers’ protocol. Cell apoptosis was monitored using an Annexin V (FITC)/PI apoptosis detection kit (Carlsbad, Calif, USA). Apoptotic cell proportion was analysed by flow cytometry (FACScan, BD Biosciences, San Jose, CA, USA) and calculated by CELL Quest 3.0 software (BD Biosciences).
Luciferase reporter assay
CDK8 3’-UTR for both wild-type (WT) and mutant-type (Mut) were amplified and inserted into the pMIR-REPORT vector (Ambion; Thermo Fisher Scientific, Inc.). And then, miR-Con or miR-216a-5p mimics was co-transfected with WT or Mut 3’-UTR of CDK8 into PC3 and DU145 cells. The luciferase activity was detected using a luciferase reporter assay kit (Promega, Madison, WI, USA).
Statistical analysis
Data were presented as mean ± standard deviation. Statistical analysis was performed using IBM SPSS Statistics Version 19.0 (SPSS Inc., Chicago, IL, USA) and GraphPad Prism Version 7.0 (GraphPad Software, Inc., La Jolla, CA, USA). Student’s t-test, nonparametric Mann-Whitney U, or Kruskal-Wallis test was used to analyse two-group differences. Inter-group differences were analysed by one-way analysis of variance, followed by a post hoc Tukey test for multiple comparisons. Overall survival (OS) and disease-free survival (DFS) was calculated using the Kaplan-Meier method with the log-rank test applied for comparison. Spearman’s rank analysis was used to identify the correlation between the expression levels of miR-216a-5p and CDK8 in PCa tissues. Pearson χ2 tests were used to evaluate differences between the clinical characteristics and miR-216a-5p expression levels in PCa patients. P-value less than 0.05 indicated a significant statistical difference.
Results
Association of CDK8 expression with clinical PCa disease stage and prognosis
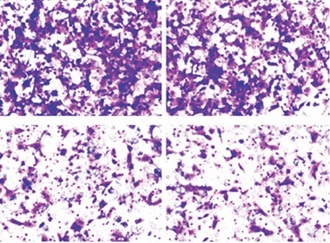
Compared with benign prostate samples, CDK8 mRNA expression was significantly increased in PCa samples (Figure 1 A). In addition, we validated that higher CDK8 mRNA expression was associated with distant metastasis (Figure 1 B), and higher pathological tumour stage (Figure 1 C) and Gleason score (Figure 1 D) of PCa. IHC staining also showed that CDK8 protein expression was upregulated in PCa samples as compared to benign prostate samples (Figure 1 E). Kaplan-Meier survival analysis revealed that PCa patients with high protein expression of CDK8 displayed significantly poor OS and DFS in our cohort (Figure 1 F).
Figure 1
Association of CDK8 expression with clinical PCa disease stage and prognosis. CDK8 mRNA expression in benign prostate samples and PCa tissues is measured using RT-qPCR (A). An increase in CDK8 mRNA expression is associated with distant metastasis (B), higher pathological tumour stage (C) and Gleason score (D) IHC staining is performed in benign prostate samples and PCa tissues to assess CDK8 protein expression (E). Kaplan-Meier survival analysis with the log-rank test applied for evaluation of the association between CDK8 protein expression and OS or DFS (F) *P < 0.05.
Knockdown of CDK8 inhibits proliferation and facilitates apoptosis
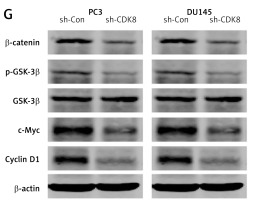
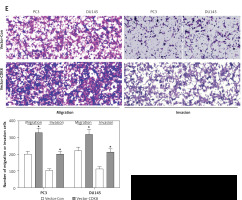
We performed the loss-of-function analysis in PC3 and DU145 cells by transfection with CDK8-specific shRNA, which effectively repressed CDK8 mRNA expression in vitro (Figure 2 A). CDK8 loss of function led to a significant decrease in cell proliferation, as measured by MTT assay (Figure 2 B) and colony formation assay (Figure 2 C). We also found that an increase in apoptotic cell proportion was observed in PC3 and DU145 cells after transfection with sh-CDK8 (Figures 2 D and E). After transfection with sh-CDK8 into PC3 and DU145 cells, cell migration and invasion were significantly blocked compared with those in the sh-Con group (Figure 2 F). Previous studies suggest that CDK8 as an oncogene participates in carcinogenesis via regulation of canonical Wnt/β-catenin pathway [6, 22]. We discovered that the protein expression of Wnt/β-catenin pathway components, including β-catenin, p-GSK-3β, c-Myc, and Cyclin D1, was dramatically reduced in PC3 and DU145 cells after transfection with sh-CDK8 (Figure 2 G).
Figure 2
The knockdown of CDK8 inhibits proliferation and facilitates apoptosis. After transfection with sh-Con or sh-CDK8 into PC3 and DU145 cells, CDK8 mRNA expression is measured using RT-qPCR (A); cell proliferation is detected using MTT assay (B) and colony formation assay (C); cell apoptosis is determined by Annexin V (FITC) double staining with flow cytometry (D and E) Migration and invasion of PC3 and DU145 cells are analysed using transwell assays (F) Protein expression of Wnt/β-catenin pathway components, including β-catenin, p-GSK-3β, GSK-3β, c-Myc, and Cyclin D1, is detected using western blotting (G) *P < 0.05.
CDK8 overexpression promotes proliferation and migration
We performed gain-of-function analysis in PC3 and DU145 cells by transfection with CDK8-overexpressed plasmids. CDK8 mRNA and protein expression were significantly elevated in PC3 and DU145 cells after transfection with CDK8-overexpressed plasmids compared with the control group (Figures 3 A and B). MTT assay (Figure 3 C) and colony formation assay (Figure 3 D) revealed that CDK8 overexpression had the ability to promote the proliferation of PCa cells. Furthermore, migration and invasion of PCa cells were measured by transwell assays, and the results demonstrated that CDK8 overexpression accelerated cell migration and invasion (Figure 3 E).
Figure 3
CDK8 overexpression promotes proliferation and migration. After transfection with vector- Con or vector-CDK8 into PC3 and DU145 cells, CDK8 mRNA and protein expression are measured using RT-qPCR and western blotting (A, B); cell proliferation detected using MTT assay (C) and colony formation assay (D) Migration and invasion of PC3 and DU145 cells analysed using transwell assays (E)
*P < 0.05.
CDK8 is a direct target of miR-216a-5p
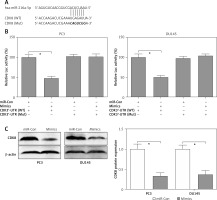
Using the online tool TargetScan, a conserved complementary pairing sequence in the 3’-UTR of CDK8 could bind with miR-216a-5p, as shown in Figure 4 A. Therefore, we hypothesised that CDK8 might be regulated by miR-216a-5p via the post-transcriptional regulatory mechanism. Dual-luciferase reporter assay was performed to elucidate the mechanism underlying miR-216a-5p-mediated suppression of CDK8. The luciferase activity was reduced by approximately 50% in PC3 and DU145 cells containing the 3’-UTR of CDK8 WT and miR-216a-5p mimics. However, the luciferase activity had not been affected by miR-216a-5p in PC3 and DU145 cells containing the 3’-UTR of CDK8 Mut (Figure 4 B). We also indicated that miR-216a-5p mimics decreased the protein expression levels of CDK8 in PC3 and DU145 cells (Figure 4 C). These findings showed that CDK8 could be regulated by miR-216a-5p via post-transcriptional repression.
Figure 4
CDK8 is a direct target of miR-216a-5p. Using the online tool TargetScan, a conserved complementary pairing sequence between the 3’-UTR of CDK8 and miR-216a-5p is predicted (A). The luciferase activity is detected in PC3 and DU145 cells containing the 3’-UTR of CDK8 WT or Mut co-transfection with miR-Con or miR-216a-5p mimics (B). After transfection with miR-Con or miR-216a-5p mimics into PC3 and DU145 cells, protein expression of CDK8 is detected using western blotting (C)
*P < 0.05.
CDK8 negatively regulates the function of miR-216a-5p in PCa cells
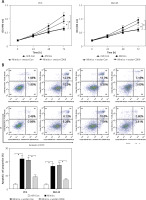
First, the roles of miR-216a-5p on proliferation and apoptosis of PCa cells were investigated. Overexpression of miR-216a-5p inhibited proliferation (Figure 5 A) and promoted apoptosis (Figure 5 B) of PCa cells, as well as downregulated the protein expression of β-catenin (Figure 5 C). Moreover, a rescued experiment was performed to discuss the interaction between miR-216a-5p and CDK8. Overexpression of CDK8 neutralised the antineoplastic activities of miR-216a-5p in PCa cells (Figures 5 A and B). In addition, miR-216a-5p-modulated downregulation of β-catenin protein expression was reversed by overexpressed CDK8 in PC3 and DU145 cells (Figure 5 C).
Figure 5
CDK8 negatively regulates the function of miR-216a-5p in PCa cells. After co-transfection with miR-216a-5p mimics and CDK8 overexpressed plasmids into PC3 and DU145 cells, cell proliferation is detected using MTT assay (A); cell apoptosis is determined by Annexinv-FITC double staining with flow cytometry (B) Cont. Protein expression of β-catenin is detected using western blotting (C) *P < 0.05.
miR-216a-5p is downregulated and is an independent prognostic indicator of PCa
RT-qPCR was performed to detect miR-216a-5p expression in 22 benign prostate samples and 55 PCa samples, and the results showed that miR-216a-5p was significantly lower in PCa samples than in benign prostate samples (Figure 6 A), and miR-216a-5p was decreased in 85.5% of PCa samples (Figure 6 B). We also found a significant negative correlation between CDK8 protein expression and miR-216a-5p expression levels in PCa samples (Figure 6 C).
Figure 6
miR-216a-5p is down-regulated in PCa. miR-216a-5p expression in benign prostate samples and PCa tissues are measured using RT-qPCR (A); miR-216a-5p is decreased in 85.5% PCa samples (B). Spearman’s rank analysis is used to identify the correlation between miR-216a-5p and CDK8 protein expression in PCa tissues (C)
*P < 0.05.

We analysed the correlation between clinicopathological variables and miR-216a-5p expression in PCa patients. As shown in Table I, a low expression level of miR-216a-5p was significantly correlated with poor pathological tumour stage and distant metastasis, while the expression level of miR-216a-5p was not associated with age, preoperative PSA level, tumour size, and Gleason score. Kaplan-Meier analysis and log-rank tests revealed that age, preoperative PSA level, tumour size, and Gleason score could not be used as prognostic indicators of PCa (Figure 7). However, poor pathological tumour stage and distant metastasis were significantly correlated with a poor OS of PCa. Interestingly, PCa patients with low expression level of miR-216a-5p showed a worse OS and DFS than those of PCa patients with high miR-216a-5p expression (Figure 7). COX univariate and multivariate analyses revealed that miR-216a-5p and distant metastasis were independent risk factors for predicting the prognosis of PCa patients (Table II).
Table I
Correlation between clinicopathological variables and miR-216a-5p expression in PCa
Table II
Univariate and multivariate regression analysis of PCa patients for overall survival
Discussion
CDK8 as an oncogene has been reported in various cancers, especially CRC [6, 22], and it modulates over-activation of Wnt/β-catenin signalling pathway, which is one of the earliest events of tumourigenesis and progression [23]. Emerging evidence suggests that CDK8 may represent a promising target for cancer therapeutics via development of novel CDK8 inhibitors [23, 24]. But the current situation is not so optimistic for pharmacological inhibition of CDK8 in cancer therapy, reflecting that CDK8 gene knockdown may have some advantages for inhibiting cancer cell proliferation [25]. In our study, knockdown of CDK8 by shRNA was able to repress proliferation and induce apoptosis in PC3 and DU145 cells and reduce the protein expression of Wnt/β-catenin pathway components, including β-catenin, p-GSK-3β, c-Myc, and Cyclin D1. These results indicate that CDK8 may act as an oncogenic gene in PCa, as shown by the induction of cell proliferation and anti-apoptotic activity.
An increase in CDK8 expression is identified in castration-resistant prostate cancer tissues, and CDK8 knockdown by small interfering RNA inhibits migration in PC3 cells [10]. In our cohort, the upregulation of CDK8 was observed in PCa samples and associated with poor OS and DFS. In vitro experiments showed that overexpression of CDK8 promoted proliferation and migration in PC3 and DU145 cells. These findings suggest that overexpression of CDK8 may contribute to oncogenesis in the prostate.
In an attempt to elaborate on the molecular mechanism underlying inhibition of CDK8 expression in the progression of PCa, we investigated whether repression of CDK8 expression could be regulated by miRNAs by post-transcriptional gene silencing mechanism. Using the bioinformatics algorithm and experimental measurements, CDK8 was validated as a direct target of miR-216a-5p. miR-216a-5p is a member of the miR-216 cluster and locates on human chromosome 2p16.1 [26]. Reduction of miR-216a-5p is frequently reported in various cancer specimens, such as cervical cancer, pancreatic ductal adenocarcinoma, and uveal melanoma [26–28]. Ectopic expression of miR-216a-5p leads to retarded cancer cell aggressiveness, suggesting that miR-216a-5p serves as anti-tumour miRNA in the progression of tumourigenesis [26–28]. Yang et al. revealed that the expression levels of miR-216a-5p were obviously decreased in DU145 and PC3 cells compared with human normal prostate epithelial cell line RWPE1, while the antineoplastic activity of miR-216a-5p was not really explored in this report [29]. In our work, we found that overexpression of miR-216a-5p played an anti-proliferative and pro-apoptotic role in DU145 and PC3 cells. Transfection with miR-216a-5p mimics into DU145 and PC3 cells led to the repression of CDK8 protein expression. Mechanically, CDK8 was a direct target of miR-216a-5p, and overexpression of CDK8 reduced the antineoplastic activities of miR-216a-5p in vitro.
At present, there is no relevant evaluation report about whether miR-216a-5p functions as a valuable prognostic predictor in cancers. Our experimental analysis determined that miR-216a-5p was downregulated in PCa specimens, and that low miR-216a-5p expression in PCa patients was closely correlated with poor OS and DFS. Cox regression model analysis suggested that miR-216a-5p could serve as an independent prognostic indicator for predicting the OS of PCa.
Taken together, miR-216a-5p/CDK8/β-catenin signalling cascade modulates the aggression of PCa. miR-216a-5p as a promising therapeutic target and prognostic marker may certainly boost and facilitate the rapid development of therapeutic strategies for the treatment of PCa.