The association between solid cancers and elevated level of total plasma vitamin B12 (B12) has been demonstrated [1, 2] and remains after adjustment for other causes of elevated B12 [3]. However, the design of previous studies did not allow them to clearly determine whether solid cancers were the cause of the B12 elevation or vice versa. The B12 elevation could be related to cancer through the tumor mass or by means of the granulocytic immune response [4–6]. However, several authors consider that the B12 elevation could favor the onset of cancer, due to the role of vitamin B12 in cell proliferation [7, 8]. This hypothesis is contradictory to the short-term association observed in cohort studies [1, 2]. The change of B12 during the treatment of solid cancers may help explain the direction of this causal relation. Indeed, a decrease of B12 after curative cancer treatment would bring an argument for asserting that the cancer induced the B12 elevation, directly or indirectly.
In the present study, we compared the change of plasma B12 after curative versus supportive treatments for solid cancer in patients with initially elevated B12 levels that were related to solid cancers.
Methods
Ethics
The bioethical committee of Angers University Hospital approved this study (n°2019/105) and waived the need for patient consent for this observational study.
Study population
We included patients aged 18 years and over who had been admitted to Angers University Hospital between January 2007 and May 2015. Patients were required to have undergone two B12 measurements at two different times (T1 and T2), at least 7 days apart.
Patients were included in cases of both i) an elevated level of B12 at T1 defined as ≥ 1000 ng/l [3], and ii) a solid cancer diagnosed between T1 and T2. Patients with an active solid cancer already known before T1 or diagnosed after T2 were not included. T1 needed to be performed in the preceding 3 months before the solid cancer diagnosis, and T2 within the next 6 months after the cancer diagnosis. In cases where there were more than two B12 measurements in the period of interest, T2 was considered to be the measurement furthest from T1 in the 6 months following the cancer diagnosis.
We excluded patients presenting other elevated B12-related diseases previously known or diagnosed during the follow-up: acute liver disease (elevation of transaminases to more than 2 times normal) or chronic liver disease (dysmorphic ultrasound appearance, persistent signs of hepatocellular insufficiency, histology suggestive of cirrhosis), severe chronic renal failure (modification of diet in renal disease (MDRD) creatinine clearance ≤ 30 ml/min/1.73 m²), autoimmune or inflammatory disease, and myeloid blood malignancy [3–5]. Patients with pernicious anemia or B12 supplementation were also excluded. Assays performed in intensive care and maternity units were excluded because of the metabolic changes observed in these patients [9, 10].
Total plasma vitamin B12 assay
B12 measurement was centralized in the biochemistry laboratory of Angers University Hospital. Plasma vitamin B12 was identified using competitive immunoassays with direct chemiluminescence on the ADVIA Centaur system (Siemens Healthcare Diagnostics Inc. Tarrytown, NY 10591-5097 USA). The normal reference range was 200–999 ng/l and the coefficient of variation was 1.3–4.1%.
Composition of groups
Patients receiving a curative treatment for solid cancer (chemotherapy, radiotherapy, hormonotherapy and/or surgery) constituted the Curative care group, regardless of the efficacy of their treatment. Patients receiving only supportive care, analgesics or other symptomatic treatments represented the Supportive care group. Patients receiving only minor palliative surgery, symptomatic radiotherapy, or a systemic corticosteroid therapy were excluded because of their potential minor curative effects.
Statistical analysis
The quantitative data were presented as medians and quartiles and compared using the t-test, as the variables demonstrated a normal distribution according to the Kolmogorov-Smirnov test. The qualitative data were presented as absolute values and percentages and compared using Fisher’s exact test. To summarize the evolution of plasma B12 over time, the best fitting model was selected according to the least squares method. Changes in B12 over time were studied using linear modeling (straight line model) after checking for the normality of the distribution of residuals (graphically and with the Kolmogorov-Smirnov test) and the linear distribution with Runs test. Slopes were compared with the F test. The type I error was set at 5%.
The analyses were carried out using GraphPad Prism v6.01 software (GraphPad Software, Inc., La Jolla, CA 92037 USA).
Results
During the study period, 896 patients had at least two B12 measurements with B12 ≥ 1000 ng/l at T1, including 448 patients without any previously known solid cancer or other elevated-B12-related cause. A cancer was diagnosed between T1 and T2 in 39/448 patients. After excluding patients with other elevated-B12-related diseases diagnosed during follow-up (n = 5, all had chronic liver disease), and patients out of delays for T1 or T2 (n = 15), we included 19 patients for analyses.
9/19 patients received curative cancer treatment between T1 and T2 and constituted the Curative care group, while 10/19 patients received only supportive care and represented the Supportive care group (Table I).
Table I
Characteristics of patients
The Supportive and Curative care groups did not differ according to age (74.5 (67.5–83.3) and 68.0 (57.0–74.0) years respectively, p = 0.14), gender (5/10 and 5/9 men respectively, p > 0.99), or B12 level at T1 (1229 (1110–1427) and 1567 (1175–1872) ng/l respectively, p = 0.16).
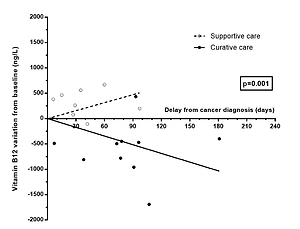
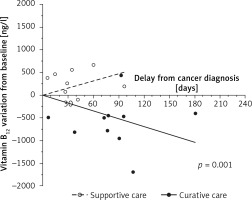
The change of B12 over time differed between groups (p = 0.001, Figure 1): B12 levels increased in the Supportive care group (+157.4 ng/l/month) and decreased in the Curative care group (–171.6 ng/l/month).
Discussion
Previous studies have demonstrated an association between solid cancers and elevated B12 level [1–3]. The short-term association observed in cohort studies suggested that the B12 elevation could be due to the cancer, but these studies failed to draw clear conclusions about the direction in which the two are causally connected. In this study, we demonstrated that B12 levels decreased during the curative treatment of solid cancers compared to supportive care. This represents an argument for considering that the elevation of B12 was directly or indirectly linked to the presence of the solid cancer.
Some authors hypothesized that elevated B12 is an underlying condition leading to cancer onset and/or progression [7, 8]. However, our results seemed in favor of the other hypothesis, which attributes an inducing role to the solid cancer, as this biological abnormality decreased in the first weeks following the initiation of a curative treatment [11]. Our results are in line with the study of Wakatsuki et al., who observed a decrease in B12 levels after surgical excision of gastric cancers. However, this study was restricted to surgical treatment in gastric cancer. Moreover, these results need to be interpreted with caution because gastric surgical procedures might have modified the absorption of vitamin B12 [12].
The decrease in B12 levels after curative treatment of cancer led to the hypothesis that the B12 level depended on the tumor mass or replication capacity. Both the higher frequency of elevated B12 levels in metastatic cancers [3] and the worst prognosis of cancers associated with elevated B12 [3, 6] supported this hypothesis. The mechanism of elevated B12 in cases of solid cancer is poorly understood. This may be related to the secretion of a mediator increasing the bioavailability of vitamin B12 or to the release of haptocorrins by the granulocytic cells involved in the anti-tumor response [13].
Our study has some limitations. First, the number of subjects is limited. However, the difference of evolution according to the type of treatment is such that our results were significant even with a limited population. Changes in B12 must be interpreted with the intra-subject variations of the plasma vitamin B12 measurements in mind, but this variation should be similar in both groups [14]. The low number of subjects prevents us from assessing the B12 change according to the efficacy of the curative treatments. Another limitation is the retrospective data collection resulting in heterogeneous delay between T1 and T2, whereas a prospective method would allow time points to be standardized. In order to obtain a homogeneous population, we restricted the period of interest of B12 measurements. As the B12 level may increase in the absence of curative care, we restricted the time from T1 to the cancer diagnosis to allow the B12 level at T1 to be approximately equal to the B12 level at the cancer diagnosis. We also restricted the time from the cancer diagnosis to T2 to limit the risk of relapses, which might modify the interpretation of B12 at T2. We only evaluated the routinely used global assay for vitamin B12, which corresponds to the sum of cobalamin and transcobalamins. However, some authors hypothesized that mainly transcobalamin I was elevated in the solid cancer condition [12]. Lastly, our results suggested that the B12 elevation might be secondary to cancers, but the study design did not allow us to assess a causal link.
In conclusion, the B12 level decreased during curative treatment in solid cancers associated with elevated B12 at the time of diagnosis. This represents an argument for considering this B12 elevation as secondary to solid cancers rather than an underlying condition that favors their onset or progression.