Introduction
Breast cancer (BC) is the foremost malignancy affecting women globally, constituting a significant public health challenge. The World Health Organization (WHO) reported 2.3 million newly diagnosed cases and 670,000 deaths due to BC in 2022, emphasizing its substantial impact on women’s health worldwide [1]. Although cancers during pregnancy are relatively rare, BC is the most common neoplasm affecting pregnant women, with an incidence estimated at 1 in 3,000 to 1 in 1,000 pregnancies, corresponding to approximately 2,000 to 4,000 cases annually in Europe alone [2–4].
Breast cancer diagnosed during pregnancy (PrBC) is a distinct clinical entity, identified either during pregnancy or within 1 year postpartum, and should be differentiated from postpartum breast cancer (PPBC). PPBC may manifest up to 5 to 10 years after delivery, and is characterized by its unique biological and molecular profiles. Meanwhile, the term “pregnancy-associated breast cancer” (PABC) is currently considered inaccurate and inconsistent, as it has been variably applied to refer to BC diagnosed solely during pregnancy or including those cases identified within 6 months to 1 year following childbirth [3].
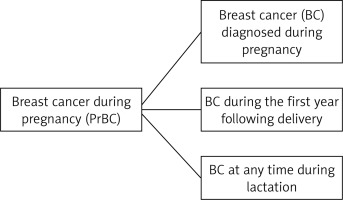
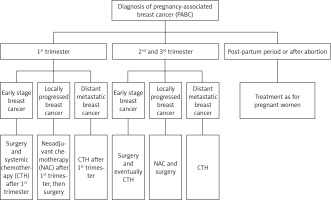
The occurrence of PrBC represents a distinct clinical entity, which can be categorized into three groups: breast cancer diagnosed during pregnancy, within the first year postpartum, or during lactation [5] (Figure 1). Notably, PrBC accounts for approximately 3% of all BC cases, with an incidence ranging from 1 in 3,000 to 1 in 10,000 pregnancies [6, 7]. Understanding the epidemiology and prognostic factors associated with PrBC is crucial for optimizing management strategies and improving patient outcomes. The management of this group of patients is of particular importance, as the data presented are based on clinical experience rather than on clinical trials. Therefore, this narrative review aimed to outline the epidemiological and molecular background, followed by presenting available therapeutic options.
Epidemiology and prognostic factors
BC typically affects women over the age of 50. However, it can also occur in young women, particularly during pregnancy. It is estimated that BC occurs in approximately 1 in 3,000 pregnancies, representing a relatively rare yet clinically significant condition [8]. The PrBC incidence rate varies between 0.2% and 2.6% among all BC diagnoses, while 35% to 55% of BC cases in pregnant women under 45 years of age occur within 5 to 10 years after pregnancy [6]. The risk is highest in the first year after birth, then gradually decreases. The greatest increase in the risk of developing BC is observed in women who have their first pregnancy after the age of 35. In this group, the risk is even higher than in non-childbearing women. The increase in PrBC observed in developing countries is due to women postponing motherhood until later in life. Studies indicate that the age of motherhood has been increasing in developed countries since the 1970s, and, more recently, similar trends have also been observed in several developing countries. The upward trend in BC incidence and the postponement of childbearing have increased the number of cases [9, 10]. Data from European cohorts indicate that the average age of onset for PrBC is 34, with diagnoses typically occurring at an average gestational age of 21 weeks [11]. Increased risk of death was found in women aged 40–44 years and diagnosed in the second year postpartum [11, 12]. In this population, the risk of tumorigenesis is mainly associated with a positive family history (50%) and BRCA1/BRCA2 germline gene mutations (30%) [7]. Additionally, during the first weeks of pregnancy, the BRCA1 gene is highly expressed to balance the state of proliferation and differentiation in mature follicles [7, 12]. Therefore, it is essential to consider the potential risk factor of the signaling of estrogen (ER) and progesterone (PR) hormones, as they play a crucial role in regulating the activity of stem cells in the mammary gland. These cells are responsible for significant transformations that take place during the menstrual cycle, pregnancy, and lactation [13, 14]. Meanwhile, lactation reduces the risk of BC by 4.3% for every 12 months of breastfeeding, including among BRCA1 mutation carriers. The mechanisms of reduced risk associated with pregnancy are related to changes in RNA processing and cellular differentiation. However, the protective effect of pregnancy depends on the age at which a woman gives birth to her first child. A young age at first pregnancy (< 25 years) reduces the risk of BC in postmenopausal women by 35% compared with non-breastfeeding women. If the first birth occurred between the ages of 25 and 29, women had an 11% increased risk compared with women who gave birth before the age of 20. If the first birth occurred at age 30 or older, the risk of BC increased by 24%. During pregnancy, the mammary gland undergoes various morphological changes under the influence of PR, prolactin, insulin-like growth factor 1 (IGF-1), glucocorticoids, and other hormones, culminating in the progressive formation of follicular structures capable of synthesizing and secreting milk [15–18]. Following the cessation of lactation, the mammary gland undergoes significant changes, characterized by the accumulation of milk in mammary epithelial cells, which may subsequently undergo apoptosis [15, 19].
Tumor biology
The classification of PrBC is the same as the general classification of BC and includes luminal A, luminal B (HER2-negative), luminal B (HER2-positive), and HER2-positive (non-luminal) and triple negative BC [20]. Staging includes prognostic information related to tumor biology, including tumor grade, estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2), and Ki-67. PrBC is similar to the phenotypes most commonly observed among young BC patients [1, 11]. It is more likely to have a high histologic grade and low or absent ER and/or PR receptors [21, 22]. A cohort study indicated that stage 2 BC is most commonly diagnosed in pregnant women. In the study, half of the subjects exhibited positive ER or PR receptors. PrBC is rarely found in the luminal A subtype. PrBC exhibits more aggressive biological behavior, with higher luminal B-like, HER2-positive, and triple-negative subtypes compared to non-pregnant women [22]. The risk of malignancy, particularly concerning basal BC subtypes such as triple-negative and BRCA1-positive tumors, may be elevated even at a young age and in instances where breastfeeding is absent [8, 23]. Conversely, breastfeeding confers a protective effect against BC incidence, with an inverse relationship observed between breastfeeding duration and cancer risk [1, 7]. Notably, while a protective effect against BC is evident in patients harboring BRCA1 mutations, such a benefit is not observed in BRCA2 mutation carriers [8]. Awareness of BRCA1/BRCA2 mutation status is important for planning the type of surgery and potential adjuvant systemic therapy [21].
Symptoms of breast cancer in pregnant women
BC symptoms among pregnant and non-pregnant patients are similar. They include a palpable or visible lump in the breast, change in the size, shape, outline, and tone of the skin of the breast, engorgement of the nipple (occurring suddenly, i.e. developing within, for example, a few weeks), retraction of the breast skin and occurrence of skin lesions or changes in the skin of the nipple, and swelling of the upper limb [21–23]. A particularly characteristic onset of PrBC is enlarged axillary/subclavian lymph nodes. However, hormonal changes during pregnancy can cause swelling, hypertrophy, nipple discharge, and increased breast tissue density, which may lead to misinterpretation of these symptoms and result in a delayed diagnosis [1, 23, 24].
Diagnosis
Delayed diagnosis of PrBC is a common issue, adversely impacting the prognosis [25, 26]. Due to the increase in breast size and density, the diagnosis may be delayed by up to 13 months [7]. Most patients typically present with a lump that is detected during breast self-examination. It is recommended that any palpable mass that persists for more than 2 weeks should be carefully diagnosed, initially with a comprehensive physical examination focusing on breast and regional lymph node status assessment [7, 25].
Next, due to the lack of ionizing radiation, high sensitivity and non-invasive nature, ultrasound is the preferred imaging modality [7, 27]. According to the National Comprehensive Cancer Network (NCCN) guidelines, CT and nuclear medicine scans are contraindicated during pregnancy. However, if indicated, a chest X-ray (with abdominal guarding), and abdominal ultrasound to assess for liver metastases should be performed, while MRI of the spine without contrast could be considered if bone metastases are suspected. Ultrasonography remains the first-choice diagnostic modality among pregnant women [28]. It can differentiate between benign and malignant lesions and identify metastases to the axillary nodes with greater sensitivity than mammography [27, 28]. The most commonly described ultrasound image in PrBC is an irregularly shaped mass with ill-defined margins and predominantly hypoechoic characteristics. During pregnancy, the parenchyma of the breast is characterized by an enlarged non-fatty fibroglandular component with little diffuse hypoechogenicity. In contrast, during lactation, the parenchyma shows diffuse hyperechogenicity, a prominent ductal system, and increased vascularization [27, 28].
In the case of a doubtful sonographic mass, a biopsy is advised. Histopathological examination of a core-needle biopsy (CNB) provides valuable information on nuclear grade, hormone receptor status, HER2 expression, and Ki-67 expression [20, 29]. Although CNB is considered the gold standard for the histological evaluation of abnormal mammographic and ultrasonographic breast masses, European Society for Medical Oncology (ESMO) guidelines advocate for fine-needle aspiration biopsy (FNAB) among pregnant women [30]. Additionally, FNAB is a common, rapid and minimally invasive method for regional lymph node assessment, including during pregnancy and lactation. Due to the overlapping sonographic features of benign and suspicious metastatic lymph nodes, ultrasound-guided FNAB provides more accurate results and is considered a highly specific technique for detecting axillary metastases in BC [23]. A complementary method to ultrasonography is mammography, which helps to detect microcalcifications. The NCCN guidelines state that shielded breast mammography can be safely performed among pregnant women, and full mammography is recommended as part of the locoregional staging to exclude multifocal or bilateral disease [8, 25, 31]. Meanwhile, the MRI contrast agent gadolinium has been restricted during pregnancy [7, 29]. Exposure of the fetus to gadolinium contrast increases the risk of rheumatological, inflammatory, or cutaneous disorders, as well as stillbirth or neonatal death [25, 27]. Breast MRI is recommended immediately after delivery or termination of pregnancy to assess local progression. The challenge is to differentiate between hypervascularization caused by cancer and hypervascularization caused by glandular hypertrophy during pregnancy and breastfeeding [31]. In accordance with the ESMO guidelines, performing MRI in pregnant women is allowed when diffusion-weighted imaging (DWI) is employed. The sensitivity and specificity of the examination for the lesion and regionally located lymph nodes range from 72% to 97% and 54% to 91%, respectively [32]. Furthermore, the examination enables the assessment of multifocality and involvement of the second breast [28]. The use of non-enhanced diffusion MRI is also becoming increasingly recognized in the diagnosis of PrBC for the purpose of systemic staging in pregnant patients, especially as contrast-enhanced MRI was demonstrated to be safe for lactating women. The doses of contrast excreted into breast milk are minimal, and the risk of complications, such as direct toxicity or allergic reactions, is very low. It is recommended, however, to refrain from breastfeeding for 12–24 h after administration of gadolinium contrast [28, 33].
Regarding scintigraphy, the method is contraindicated for pregnant women; also, pregnant women should not be in the company of patients who have undergone the examination [7]. MRI DWI outperforms bone scintigraphy and has shown high accuracy in diagnosing bone, liver, and peritoneal metastases among pregnant patients.
Treatment
Systemic therapy
Pregnancy is not a contraindication to systemic treatment. According to the NCCN, the considerations and selection of optimal local and systemic therapy are similar to those recommended for BC not associated with pregnancy. Coordination is recommended between the oncology and obstetrics teams to plan the optimal timing of systemic therapy administration during pregnancy. Chemotherapy as part of primary BC treatment is indicated in most young patients with cancer, depending on stage and tumor biology. The majority of BCs in young women are non-luminal A type, and for the majority of non-luminal A tumors, a neoadjuvant approach is recommended. Furthermore, such an approach enables monitoring of the response to systemic therapy and can help guide optimal post-neoadjuvant treatment [1, 33]. It is also the treatment of choice in stage IV of the disease. Notably, the general treatment regimen remains similar among both pregnant and non-pregnant patients [1]. However, systemic therapy should only be initiated in the second trimester of pregnancy due to the potential impact on fetal development. During the period of organogenesis (3–12 gestational weeks), the risk of congenital malformations and fetal loss due to chemotherapy exposure may be high. Retrospective data have shown a 14% to 20% rate of major fetal malformations when chemotherapy was administered during the first trimester of pregnancy [10]. Therefore, the benefit to the fetus of delaying treatment until the second trimester should be weighed against the risk to the mother [34, 35]. The systemic treatment must be completed or terminated before the 34th week of pregnancy, as chemotherapy increases the risk of maternal and neonatal myelosuppression during delivery. Moreover, treatment continuation after 34 gestational weeks is associated with an increased risk of preterm contractions and spontaneous labor [24, 29]. Therefore, chemotherapy after 34 gestational weeks is contradicted and, if indicated, should be administered 3 weeks after the last administration of chemotherapy.
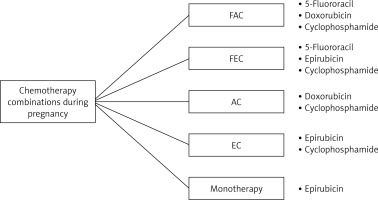
Chemotherapy doses for pregnant patients should not differ from those for non-pregnant patients, and should be calculated based on actual body surface area [36]. During chemotherapy, careful and continuous monitoring of maternal blood pressure and fetal monitoring are important, as the treatment may be associated with increased risk of preterm birth, intrauterine growth restriction, and hypertensive disorders of pregnancy [33]. It is crucial to underscore that although breastfeeding confers substantial advantages for newborns, it is contraindicated during systemic treatment including chemotherapy, targeted therapy, and immunotherapy, due to the potential excretion of the drug in breast milk [37, 38]. The first-line agents according to ESMO guidelines are doublet and triplet chemotherapy regimens including 5-fluorouracil, cyclophosphamide, or doxorubicin, administered in a 3-week regimen, whereas epirubicin monotherapy is given weekly [22, 28, 39, 40]. These agents are characterized by low risk of short- and long-term fetal cardiotoxicity, teratogenic risk, or neurocognitive impairment [22, 28]. Taxanes are indicated in neoadjuvant treatment and in advanced HER2-positive metastatic BC cases of pregnant patients who are not candidates for anthracycline-containing regimens. Preferably, weekly paclitaxel should be administered, as it has been demonstrated to prolong overall and disease-free survival [41, 42]. Anthracycline-based chemotherapy can be used with minimal risk to the fetus [43]. According to ESMO guidelines, neo- and adjuvant regimens based on anthracyclines and taxanes can be administered as in the non-pregnant setting [44].
Available systemic therapy during pregnancy is depicted in Figure 2.
In the case of pregnant patients with HER2+ BC, it is imperative that anti-HER2 therapy is postponed until the point of delivery, due to the potential risk of fetal toxicity [45]. The ErbB2/neu receptor is involved in fetal organogenesis; it may cause thrombocytopenia by blocking receptors in the fetal kidneys [25]. It is also contraindicated to administer endocrine therapy, as complications may arise in the form of neonatal ambiguous genitalia and oculoauriculovertebral dysplasia [46]. The ESMO indicates that immunotherapy, including immune checkpoint inhibitors (ICIs) such as anti-programmed cell death protein 1 (PD-1) and anti-programmed death ligand 1 (PD-L1), should be avoided during pregnancy and postponed until delivery. The interactions between PD-1 and PD-L1 have been demonstrated to play a pivotal role in maternal immunotolerance to paternal fetal alloantigen. Additionally, PD-L1 acts as an inhibitor of maternal immune responses against the fetus, and disrupting this pathway could lead to increased risk of fetal rejection. Therefore, it is advised to delay immunotherapy until after delivery, when anti-PD-1 can be included in pre-surgical treatment [44, 47]. For patients with advanced luminal BC, CDK-4 therapy is recommended. However, since CDK4/6 inhibitors can pass through the placental barrier in various animal models, their administration should be delayed after childbirth [48].
Regarding febrile neutropenia prevention during chemotherapy, granulocyte colony-stimulating factors (G-CSFs) may be considered [49]. Although the administration of G-CFS during pregnancy has increased, the indications for their use remain unclear. However, a multi-institutional analysis of G-CFS usage among pregnant women did not show unfavorable outcomes for mother and fetus, with preterm births being mostly iatrogenic [50]. With higher rates of chemotherapy administration among pregnant women and the need to facilitate dose-dense regimens, the use of G-CSFs is expected to increase systematically.
Perioperative care and surgery
Surgical treatment for PrBC is safe at any gestational stage, as currently used anesthetics are not teratogenic [51]. The timing of surgery should be individualized based on patient and tumor characteristics, gestational age, and preference. All surgical procedures should be planned and coordinated by a multidisciplinary team (MDT), including an obstetrician to provide fetal monitoring both pre- and postoperatively [52]. However, elective procedures are recommended to be postponed until after delivery [33]. According to American Congress of Obstetricians and Gynecologists (ACOG) guidelines, fetal heart rate (FHR) monitoring can facilitate maternal positioning and managing cardiopulmonary function [53]. For viable fetuses, FHR should be assessed with Doppler ultrasound before and after the surgery. Intraoperative electronic fetal monitoring may be employed if resources allow and if the procedure can be safely interrupted for emergency cesarean delivery if indicated.
It is recommended to conduct surgical procedures in facilities that provide access to neonatology and pediatric services [54]. Emergency procedures should be performed without delay to ensure optimal maternal and fetal outcomes. The principles include the maintenance of uteroplacental perfusion by preventing maternal hypoxemia, hypotension, hyper- and hypocapnia, temperature extremes and stress. When feasible, regional anesthesia is preferred due to its favorable outcomes for both mother and fetus [55]. Pregnant women should be evaluated for the risk of venous thromboembolism, with an appropriate perioperative prophylaxis. Additionally, the use of antenatal corticosteroids should be considered for fetal lung maturation in cases of preterm birth risk. Obtaining informed consent is critical and must explicitly cover risks such as preterm birth, fetal death, and the potential need for emergency cesarean section [54].
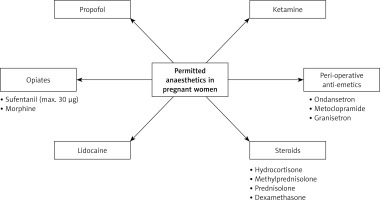
After 20 gestational weeks, pregnant women should be placed in the left lateral decubitus position to avoid inferior vena cava compression and prevent hypoxia, hypotension, hypoglycemia, fever, pain, infection, or thrombosis [56, 57]. Therefore, continuous cardiotocographic monitoring during the perioperative period is mandatory for early recognition of fetal distress signs [58]. Approved anesthetics among PrBC patients are depicted in Figure 3.
Propofol (2,6 diisopropylphenol) is employed in induction and maintenance phases of general anesthesia. It facilitates rapid and mild induction of anesthesia, although it lacks analgesic properties. Depending on the dose administered, it reduces cardiac output and blood pressure. The primary advantage of propofol is the capacity for rapid awakening. In pregnant patients, it is a low-molecular-weight lipophilic molecule that rapidly crosses the placenta. In studies, it was found to be rapidly cleared from the neonatal circulation. Furthermore, the concentration in breast milk was low [59]. Ketamine is usually used as an induction anesthetic and causes unconsciousness within 30–60 s after an intravenous induction dose, which can last 15–20 min [60]. Due to its rapid onset of action, anesthesia, and amnesia, it is a valuable agent in obstetric patients [59]. It rapidly crosses the placenta and reaches a maximum concentration in the fetus approximately 1.5 to 2 min after administration. Antiemetic and anti-nausea drugs used during pregnancy include ondansetron, metoclopramide, and granisetron [54]. Ondansetron is a procainamide derivative that acts as a cholinergic receptor agonist peripherally and a dopaminergic receptor antagonist centrally. It increases lower esophageal sphincter tone, has antiemetic effects, and reduces gastric volume by increasing gastric peristalsis [60]. The administration of metoclopramide during pregnancy is deemed to be safe and does not appear to be associated with an increased risk of teratogenic effects, preterm birth, low birth weight, or perinatal death. Metoclopramide is more effective than monotherapy with prochlorperazine or promethazine in relieving vomiting and achieving subjective improvement. Compared with ondansetron, similar improvements have been shown for nausea but less for vomiting [61]. Due to adverse effects on the fetus, ondansetron must not be used in the first trimester of pregnancy.
Exposure of the fetus to glucocorticoids may result in several adverse effects, which may become apparent both during pregnancy (intrauterine growth retardation, increased risk of preterm birth, increased risk of cleft palate) and later in life (hypertension, increased hypothalamic arterial hypertension, increased activity of the hypothalamic-pituitary-adrenal system, behavioral disorders) [55, 56]. It is recommended that prednisone, prednisolone, and methylprednisolone be used as the drugs of choice in the treatment of pregnant women, as they are minimally transported to the fetus [62]. Local anesthetics administered during labor readily cross the placenta and can, therefore, cause toxicity symptoms in both mother and child. The toxicity of these drugs depends on the method and technique of anesthesia and the dose used. Lidocaine use is only permitted in exceptional cases due to the risk of cardiac dysfunction in the fetus (bradycardia, atrioventricular block, and ventricular tachycardia). The use of amide-derived drugs for periventricular block during labor has been associated with bradycardia in approximately 30% of fetuses. Continuous monitoring of the fetal heart rate is therefore advisable. Excessive concentrations of lidocaine in the mother’s blood may cause a reduction in fetal blood pressure. In its other forms, lidocaine can usually be used in pregnant women after weighing the benefit-to-risk ratio of its administration. Following topical administration, the dose of the drug that penetrates into breast milk is low, thus allowing for the drug to be used during breastfeeding with caution.
Opioids recommended for use among pregnant women include morphine and sufentanil, which provide an analgesic effect and in low doses may serve as an adjunct to other anesthetics [54, 59]. In terms of safety among pregnant women, they are categorized as C, but large doses of morphine are category D, according to the Food and Drug Administration (FDA). The misuse of opioids by pregnant women is associated with an increased risk of preterm birth, intrauterine growth restriction, and neonatal death.
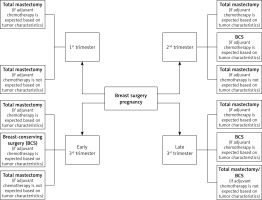
Mastectomy and breast-conserving surgery (BCS) are essential surgical procedures in the curative-intent treatment of BC, providing comparable benefit in terms of long-term survival [63]. The choice of surgery during pregnancy should follow the same guidelines as for non-pregnant women. BCS should be preferred and can be performed in the second and third trimesters of pregnancy, followed by postpartum radiotherapy and systemic treatment if necessary.
Mastectomy with immediate breast reconstruction (IBR) is currently one of the most popular reconstructive methods and should be considered for women diagnosed with BC during pregnancy. IBR eliminates the necessity for subsequent surgical intervention. However, to minimize the potential adverse effects on the fetus, delayed breast reconstruction (DBR) is preferred [58, 63] (Figure 4).
Axillary lymph node dissection supplements the surgery [28, 63, 64]. Nowadays, due to the increasing number of pathologically negative nodes, a significant number of patients benefit from axillary treatment de-escalation, which often can be limited to the sentinel lymph node biopsy (SLNB) [64, 65]. Of note, SLNB requires the injection of radioisotope-labeled colloids or blue dyes into the breast, which poses some difficulties for pregnant women [63]. Both isosulfan blue and methylene blue have been classified as category C by the FDA [38, 64]. Therefore, due to the risk of anaphylactic shock and fetal intestinal atresia, ESMO advises against the use of blue dye during pregnancy [66]. According to NCCN guidelines, it can be replaced with radioisotope-labelled sulfur colloid, which appears to be safe for SLNB in pregnancy. Recently, the SOUND trial presented results suggesting that among pregnant women with a small tumor in the breast, axillary surgery or even SLNB can be entirely omitted without compromising distant disease-free survival at 5 years [67, 68].
To facilitate tissue division and promote hemostasis during surgery, electrocautery with high frequency alternating current to generate heat in living tissue may be considered [69]. The procedure provides a fulguration, desiccation/coagulation, or vaporization/ablation effect while ensuring the absence of electrical shock. Devices can be classified into two main categories, monopolar and bipolar, based on their circuit design and return electrode. However, reports on the use of energy during laparoscopic procedures among pregnant women remain limited. Nevertheless, there is no evidence that electrocautery is harmful to the fetus or embryo or that there is an increased risk of energy-related complications with the use of any type of energy device. When monopolar energy is used, it is recommended that the return plate not be placed in such a way that the uterus is between the electrode and the plate [69–72].
Radiotherapy
In accordance with the ESMO guidelines, radiotherapy is not inherently contraindicated among PrBC patients [28]. The radiation dose to the fetus depends on the distance from the target volumes being irradiated and the radiotherapy parameters, including the recommended dose, size, location of the target volumes, and technical specifications. The cumulative fetal exposure to radiation must be calculated by a medical physicist, and the patient must be informed of the associated risks. Among pregnant women, the potential benefits and risks of radiotherapy, including the potential risk to the fetus, must be carefully considered [44]. Radiation may induce malformations during organogenesis, particularly in the central nervous system (e.g., microcephaly). However, recent cohort studies have shown that the differences in neurocognitive and psychosocial performance among children prenatally exposed to radiotherapy were not associated with radiation [73]. After the first trimester, the main effects are growth restriction, mental retardation, and infertility. Although postponing irradiation until after delivery is recommended, the critical threshold for teratogenic effects has been set at 0.1 Gy [66, 74]. The dose can be further reduced by using additional shielding in the pelvic region. For BC, the main indications for radiotherapy include postoperative breast/chest wall irradiation with or without lymph drainage, after BCS and after mastectomy, depending on risk factors [20, 28].
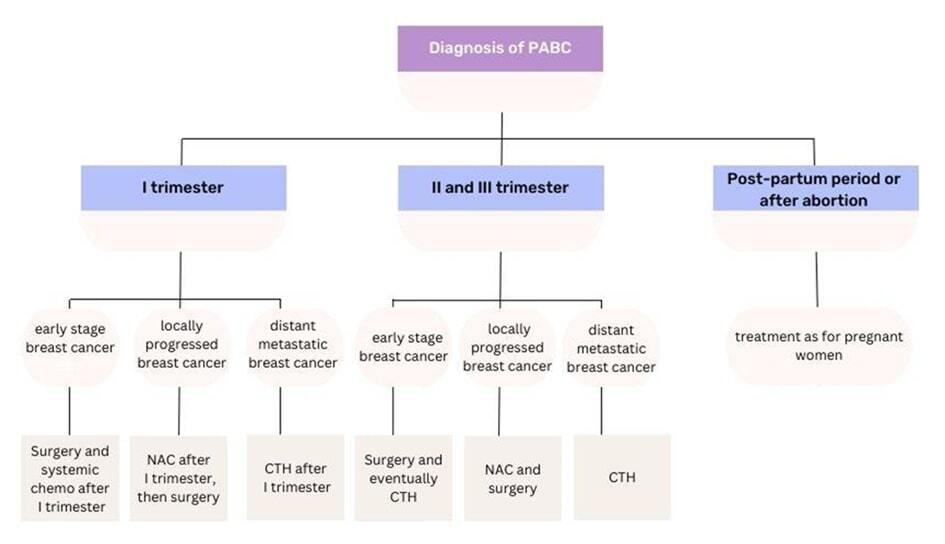
Importantly, the sensitivity of fetal tissues to radiation, and the risk of radiation-related toxicity, depend on gestational age. Therefore, the stage of pregnancy along with the parameters associated with radiotherapy determines the risk, and early involvement of the radiotherapy team may be beneficial. Furthermore, it is important to reiterate that radiotherapy makes BCS possible [44]. The therapeutic algorithm during pregnancy is depicted in Figure 5.
Treatment of breast cancer in postpartum women
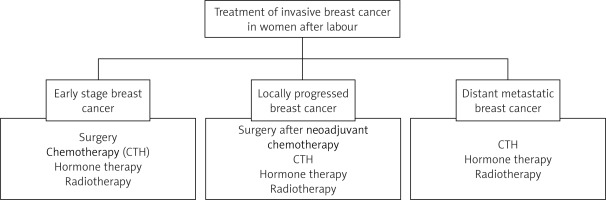
BC that develops within the first year after birth is classified as PrBC, whereas tumors that develop up to 5 years after birth are defined as PPBC [28]. Pregnancy and the postnatal period have been demonstrated to induce profound alterations in cell proliferation, survival, angiogenesis, and tissue remodeling. These processes have been shown to promote cancer progression significantly. Pregnancy and the postnatal period have been identified as distinct subgroups of PrBC, and cellular and molecular modifications specific to pregnancy or the postnatal period have been observed to exert differential effects on tumor progression [75, 76]. The therapeutic management of women after childbirth or abortion is essentially the same as for non-pregnant women. It follows recommendations that consider the biological subtype of the tumor, the clinical stage, the patient’s general condition, and concomitant diseases [75, 77]. The most significant difference in the treatment of PrBC compared with PPBC is the use of endocrine therapy, immunotherapy, and anti-HER2 therapy [76]. Tamoxifen can lead to fetal malformations and should therefore be delayed until after birth [78] (Figure 6).
Conclusions
Diagnostic workup and multimodal treatment of BC in pregnancy should be carefully planned by the multidisciplinary team, which includes an obstetrician. The management should follow the established recommendations for non-pregnant patients while considering the potential risks to the fetus. Curative-intent treatment should incorporate systemic therapy and – whenever safe and feasible – radiotherapy, to facilitate BCS.