Introduction
Traumatic brain injury (TBI) is mainly caused by external physical insults to the brain, which may lead to alterations in consciousness, of the mental or physical state of the patient [1]. TBI remains one of the principal causes of deaths and disabilities with almost 10 million victims worldwide each year [2, 3]. Nearly 2.5 million Americans suffer from the tragic consequences of TBI. These patients live with impairment in sensory, motor, behavioral, or cognitive functions. The incidence rate for moderate and severe TBI in children has not improved in the last 10 years, with disappointing outcomes for those with severe injures [4–6].
The World Health Organization warns that along with the human loss, TBI is one of the top financial burdens on health-providing platforms [2, 7]. The damage that occurs during the primary impact is referred to as the “primary injury”, whereas the damage secondary to the initial insult via cellular, physiological, and biochemical events is referred to as “secondary injury” [8]. Brain edema followed by increased intracranial pressure (ICP) is the characteristic of many neurosurgical diseases. An extreme ICP is believed to be the main cause of death in such patients. The drug of choice to decrease an acute ICP is mannitol or hypertonic saline [9, 10].
The current treatment options for TBI, such as hyperbaric oxygenation, rehabilitation and brain stimulation are only of supportive nature; therefore, it is necessary to seek an absolute therapeutic option [11, 12]. Considering the complex pathomechanism of TBI, a treatment that could maintain or restore the function of injured neurons would be the best approach. Progenitor cells are of great importance in this regard due to their plasticity, migration, and self-renewal capacity [13, 14]. Recently cellular therapy has gained a particular interest in various diseases, such as cerebral palsy, TBI, stroke, spinal cord injury, and autism [15–17].
Several types of cells, such as bone marrow derived stem cells, neural stem cells (NSCs), embryonic cells, pluripotent cells and umbilical cord blood cells, have improved TBI in different animal models [18–21]. The way transplanted cells help to repair TBI might be via replacing the damaged cells through proliferation and differentiation, or by secreting trophic factors to cause endogenous repair [22]. As cell transplantation for TBI is not only studied in preclinical models, but also tried in various clinical trials, we decided to perform a meta-analysis on the effects of cellular therapy for TBI in adult patients.
Material and methods
This meta-analysis is compliant with PRISMA 2020, and follows a previously described protocol [23].
Search strategy
A systemic search was conducted on PubMed, China National Knowledge Infrastructure (CNKI), Cochrane Library, Embase, Wan Fang Data and Google Scholar using the search terms “traumatic brain injury”, “cellular therapy”, and “clinical trial” for articles published in English language prior to May 2024. Two researchers independently examined the titles and abstracts of all searched records and excluded those that did not meet eligibility criteria.
Inclusion criteria
The studies were included if they met the following criteria: (1) the main focus of the study was on cellular therapy for TBI; (2) original, controlled clinical trial research article; (3) adult patients aged 18 years or over; (4) full text article available.
Exclusion criteria
The studies were excluded if they met one of the following criteria: (1) patients aged under 18 years; (2) no full text accessible; (3) no control available; (4) case report; (5) letter to editor; (6) preclinical study; (7) review article; (8) study with no quantitative data; (9) meeting abstract; (10) book chapter; (11) low-quality study.
Quality assessment
In order to assess the quality of a study, the Newcastle–Ottawa Scale (NOS) was used. In case-control trials, the NOS covers three areas – selection, exposure, and comparability – while in a cohort study it covers selection, outcome, and comparability [24]. A numbered item in exposure, outcome, or selection categories can be maximally awarded with one star, whereas a maximum of two stars can be given to a numbered item in a comparability category [25]. A study can maximally receive 9 stars, and a study was considered as high quality with 6+ stars, moderate quality with 4–5 stars, and low quality with less than 4 stars [26].
Data extraction
The following data were extracted from all included articles: first authors’ names, publication year, country of research, type of study, (number, sex and age) of participants, type of cells used, and post-treatment follow-up. The extracted data were entered into a predesigned data collection sheet, and then tabulated onto a spreadsheet (Table I). Moreover, outcomes such as Fugl-Meyer Motor Scale (FMMS), Disability Rating Scale (DRS), and overall improvement of the patients were extracted and analyzed using RevMan (Ver.5.4.1). FMMS is a broadly accepted scale used in clinical practice to measure motor deficit of the affected limb(s) in conditions such as stroke or TBI. It has a maximum score of 100, with a score of zero indicating hemiplegia, and 100 representing a normal individual [27, 28]. The DRS measures general functional changes in TBI patients. Its scores range from zero to 29, with zero representing no disability and 29 designating a profound vegetative state [27, 29].
Table I
Summarized characteristics of studies included in meta-analysis
Statistical analysis
The data were analyzed using RevMan (Ver.5.4.1) software. Heterogeneity among the studies was tested, and a p-value < 0.05 or I2 > 50% was considered to demonstrate significant heterogeneity [10]. Dichotomous data such as in “overall improvement” were expressed as odds ratio (OR) with 95% confidence interval (CI). Continuous data such as in FMMS and DRS were expressed as mean difference (MD) with 95% CI. A p-value less than 0.05 was considered to be statistically significant.
Results
Characteristics and selection of individual studies
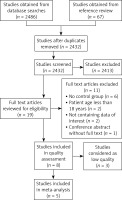
Initially, 2553 articles were identified through database searches and reference review. The records were checked for duplicates, and 2432 articles were left after repetitive articles were removed. Screening the titles and abstracts of the remaining articles, 2413 articles were removed, 19 articles were selected to be relevant, and their full texts were accessed. The 19 full text articles were evaluated for eligibility; 6 articles had no control group, the participants of 2 articles were less than 18 years old, 2 articles contained no extractable data, 1 article was a conference abstract, and 3 articles were of low literature quality. After applying the eligibility criteria, 5 controlled clinical trials (Table II) were finally included in the meta-analysis (Figure 1). The included articles contained 367 patients, with a sample size in the range of 24 to 166.
Overall improvement of patients
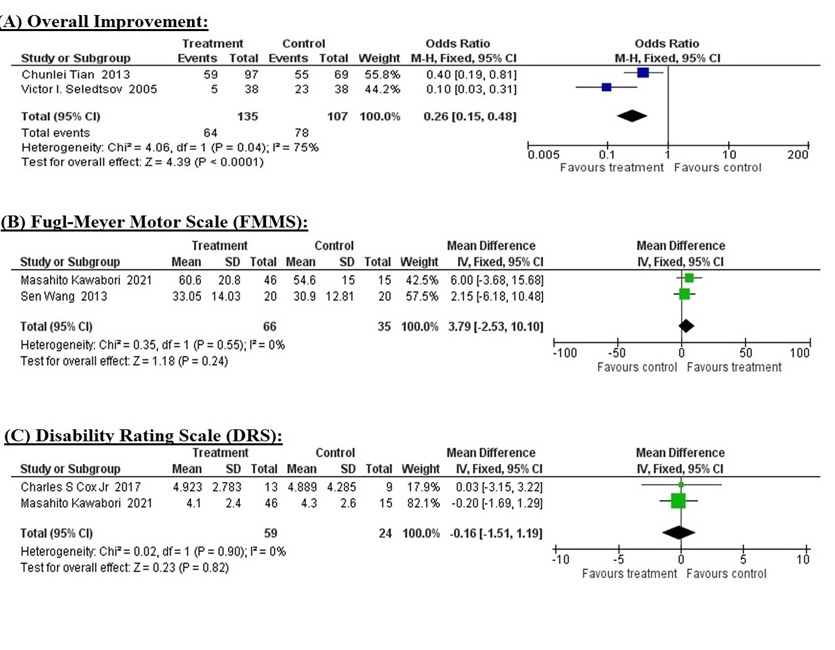
Two studies [30, 31] provided data on overall improvement of the patients after cellular therapy comparing treatment and control groups. The fixed effect model was chosen based on statistically significant heterogeneity (I2 = 75%, p = 0.04) among the studies. The pooled mean difference (MD) of overall improvement in treatment groups versus control groups was 0.26 (95% CI: 0.15 to 0.48, p = 0.0001) which indicates that the treatment significantly improves overall condition of the patients (Figure 2 A).
Figure 2
Forest and funnel plots of all studies included in meta-analysis. Forest plots of overall improvement (A), Fugl-Meyer Motor Scale (B), and Disability Rating Scale (C) comparing treatment versus control groups. Funnel plots for observing any possible publication bias in overall improvement (D), Fugl-Meyer Motor Scale (E), and Disability Rating Scale (F)

Fugl-Meyer Motor Scale
FMMS related information was obtained from two studies [27, 32], comparing treatment and control groups. Heterogeneity (I2 = 0%, p = 0.55) across the studies was not significant based on the fixed effect model. The pooled MD of FMMS in the two groups was 3.79 (95% CI: –2.53 to 10.10, p = 0.24). Although the data show that the treatment’s effect on improving motor activity is not statistically significant, it may still have clinical significance (Figure 2 B).
Disability Rating Scale
DRS was reported by two studies [27, 33], comparing treatment and control groups. There was no heterogeneity among the studies (I2 = 0%, p = 0.90) with the fixed effect model. The pooled MD was –0.16 (95% CI: –1.51 to 1.19, p = 0.82) showing that cellular therapy does not significantly improve disability in TBI patients (Figure 2 C).
Risk of bias
To estimate the risk of publication bias, funnel plots were obtained and visually assessed for all studies included in the meta-analysis. Based on the symmetric distribution of the studies’ effect sizes in the funnel plots, it was concluded that no publication bias was present (Figures 2 D–F).
Discussion
Our meta-analysis included five clinical trials covering overall outcome, motor activity and disability improvement of adult TBI patients after cell therapy. Pathophysiological events during TBI fall into two categories: primary and secondary. The biomechanical or physical insults that lead to the immediate events are followed by a flow of events such as, production of free radicals, excitotoxicity, hypoperfusion, ischemia, disturbance to cerebrovascular autoregulation, intracranial hypertension and metabolic dysfunction [34]. The flow of events that release various biological factors causes cellular death, which results in local or global cerebral atrophy [35]. Thyroid cancer stem cells (TCSCs) are interesting biomarkers and possible targets for clinical intervention since they are essential to the pathophysiology, metastasis, and therapeutic response of thyroid cancer. Numerous studies conducted in the last few years have shown a strong correlation between cancer stem cells (CSCs) and the development and progression of tumors [36]. Research findings suggest that global injury occurs more commonly, which is mainly evidenced in the frontal lobe, hypothalamus, temporal lobe, basal ganglia, corpus callosum, fornices, hippocampus, and superior cerebral peduncles [37, 38]. Salidroside can reduce both the neurological impairment score and the infarct volume of the rat brain in the focal cerebral ischemia/reperfusion injury model in rats [39]. Injury to these structures initially causes mood disorders, psychiatric deficits, depression, and neurobehavioral alterations [40].
Various preclinical studies have shown the regenerative ability of stem cells in animal TBI models [41–45]. Several preclinical TBI models have demonstrated improvement in motor, behavioral and cognitive functions after neural, mesenchymal, or progenitor stem cell therapy. These effects are most likely caused by production of neurotrophic factors, improvement of angiogenesis and downregulation of astrogliosis [18, 46, 47]. Treating unilateral limbal stem cell deficits has shown promise with autologous limbal epithelium transplantation [48]. When existing treatment options and accepted medical standards are inadequate, transplantology is a branch of medicine that saves lives [49]. Ma et al. indicated that transplanted cells significantly decrease at the early stage of transplantation. The possible reason could be the post-traumatic inflammatory cascade in the recipient brain that affects the survival of the cells [50, 51]. Zhang et al. applied bone marrow derived autologous mesenchymal stem cells in 7 TBI patients via intracranial and intravenous route. They found that it was safe and the patients showed significant improvement in neurological functions [52]. Moreover, Cox et al. and Liao et al. also transplanted bone marrow mononuclear cells to severe TBI patients through the intravenous route, and they too reported the treatment to be safe and clinically significant [53, 54]. Histopathological damage and the CNS inflammatory response progressively resolve and return. Consequently, microglia may be one of the key targets of thermal stimulation-mediated central nervous system injury, and controlling their polarization by restricting M1 or encouraging M2 activation may develop into a viable therapeutic approach for disorders that produce heat-induced brain damage [55]. The administration of autologous bone marrow mononuclear cells to chronic TBI patients by Sharma et al. also improved the condition of the patients without any major side effect [1].
Studies have shown that NSC therapy improved the neurological functions in preclinical models of TBI [56–58]. Several potential mechanisms have been proposed for obtaining these effects, such as immunomodulation and restoring neuronal circuits [59], production of neurotrophic factors [60], secretion of specific neurotransmitters [61], and neuronal cell replacement [62]. Research on spinal cord injury (SCI) in animals has demonstrated that SCI causes two types of damage: mechanical damage; and secondary injury caused by neuronal apoptosis in the central nervous system (CNS), which causes the damage to spread. According to our research, the rs531564 polymorphism may cause down-regulation of miR-124, which in turn may enhance the production of BIM. This could lead to death in cells and prolong the time required for patients to recover following SCI [63]. Together, necrosis and apoptosis result in death of neurons and glia during TBI. Some preclinical studies show that NSC transplantation reduces apoptosis around the ischemic spots, resulting in functional improvement [64]. Osteoblast progenitors found in bone marrow stem cells (MSCs) in blood clots can result in the production of bone on scaffolds in the presence of growth stimuli [65].
It is also possible that these transplanted cells enhance endogenous repair responses such as improving synaptogenesis, neurogenesis, and angiogenesis [66–69]. Some researchers also propose that the secretion of specific trophic factors such as BDNF, NGF, GDNF, and VEGF by transplanted cells could be a possible mechanism for neuronal regeneration and repair [70, 71]. As the capacity of the brain is very limited to regenerate neurons, it is challenging to repair a damaged structure in the brain. At present, no treatment exists to treat diffuse axonal injury and to divert the cascade of pathological events that leads to cellular death [1]. Even in preclinical TBI models, transplantation of NSCs leaves several questions unanswered, such as the ideal time of therapy, effective route of administration, and optimal dose for the cells [72]. Cellular therapy demonstrated potential to repair cerebral damage via neuroprotective and neurorestorative mechanisms. It is believed that stem cells use their neurogenic ability to repair injured brain [41]. After all, stem cell therapy remains the only hope for the future of TBI patients.
In conclusion, this meta-analysis suggests that cellular therapy significantly improves (p = 0.0001) the overall condition of adult TBI patients. Moreover, the pooled data for the Fugl-Meyer Motor Scale (p = 0.24) and Disability Rating Scale (p = 0.82) show a non-statistically significant improvement, which is still of great clinical importance.