Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
NEUROLOGY / CLINICAL RESEARCH
Diagnostic value of cerebrospinal fluid chemokine ligand 13 (CXCL13) levels for viral and autoimmune encephalitis
1
Department of Immunopathology of Infectious and Parasitic Diseases, Medical University of Warsaw, Poland
2
Department of Microbiology, Medical University of Warsaw, Poland
3
Department of Adults Infectious Diseases, Medical University of Warsaw, Poland
4
Department of Neurology, Medical University of Warsaw, Poland
Submission date: 2024-09-24
Final revision date: 2025-04-29
Acceptance date: 2025-07-02
Online publication date: 2025-08-23
Publication date: 2026-04-30
Corresponding author
Karol Perlejewski
Department of Immunopathology of Infectious and Parasitic Diseases Medical University of Warsaw Pawinskiego 3c 02-106 Warsaw, Poland
Department of Immunopathology of Infectious and Parasitic Diseases Medical University of Warsaw Pawinskiego 3c 02-106 Warsaw, Poland
Arch Med Sci 2026;22(2):841-847
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Chemokine ligand 13 (CXCL13) has been reported to be a valuable diagnostic biomarker in Lyme neuroborreliosis (LNB). However, its utility in the diagnostics of viral and autoimmune (AE) encephalitis still remains unclear.
Material and methods:
We measured CXCL13 concentrations in cerebrospinal fluid (CSF) samples collected from 21 patients with viral encephalitis (17 cases of herpes simplex viral (HSV) and 4 of enteroviral (EV) encephalitis) and 6 patients with AE (5 subjects with antibodies anti-NMDAR and 1 with anti-GABA) and compared them to those found in patients with LNB (7 subjects) and multiple sclerosis (8 cases) as well as ten control subjects without neuroinflammation.
Results:
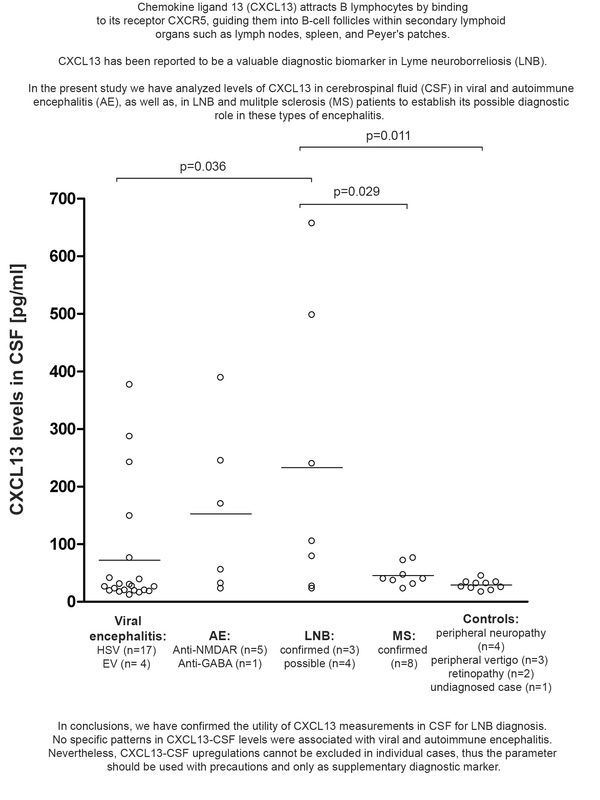
Patients with neuroinflammation had a mean level of CXCL13-CSF of 105 pg/ml compared to 29 pg/ml in controls. The highest mean level of CXCL13 in CSF was detected in LNB patients (233 pg/ml), and the lowest in controls (29 pg/ml). Significant upregulation of CXCL13-CSF levels in LNB patients was observed in comparison to viral encephalitis and MS patients as well as controls. A positive correlation between elevated chemokine levels and cell count in CSF was found in all patients (r = 0.6496; p < 0.0001), as well as in the LNB group when tested alone (r = 0.8428; p = 0.0173). A positive correlation with CSF protein levels was observed in all patients (r = 0.7216; p < 0.0001), and separately in LNB (r = 0.8573; p = 0.0137) and AE patients (r = 0.8885; p = 0.0180).
Conclusions:
The findings support the utility of CXCL13 measurements in CSF for LNB diagnosis. No specific patterns in CXCL13-CSF levels were associated with viral or autoimmune encephalitis.
Chemokine ligand 13 (CXCL13) has been reported to be a valuable diagnostic biomarker in Lyme neuroborreliosis (LNB). However, its utility in the diagnostics of viral and autoimmune (AE) encephalitis still remains unclear.
Material and methods:
We measured CXCL13 concentrations in cerebrospinal fluid (CSF) samples collected from 21 patients with viral encephalitis (17 cases of herpes simplex viral (HSV) and 4 of enteroviral (EV) encephalitis) and 6 patients with AE (5 subjects with antibodies anti-NMDAR and 1 with anti-GABA) and compared them to those found in patients with LNB (7 subjects) and multiple sclerosis (8 cases) as well as ten control subjects without neuroinflammation.
Results:
Patients with neuroinflammation had a mean level of CXCL13-CSF of 105 pg/ml compared to 29 pg/ml in controls. The highest mean level of CXCL13 in CSF was detected in LNB patients (233 pg/ml), and the lowest in controls (29 pg/ml). Significant upregulation of CXCL13-CSF levels in LNB patients was observed in comparison to viral encephalitis and MS patients as well as controls. A positive correlation between elevated chemokine levels and cell count in CSF was found in all patients (r = 0.6496; p < 0.0001), as well as in the LNB group when tested alone (r = 0.8428; p = 0.0173). A positive correlation with CSF protein levels was observed in all patients (r = 0.7216; p < 0.0001), and separately in LNB (r = 0.8573; p = 0.0137) and AE patients (r = 0.8885; p = 0.0180).
Conclusions:
The findings support the utility of CXCL13 measurements in CSF for LNB diagnosis. No specific patterns in CXCL13-CSF levels were associated with viral or autoimmune encephalitis.
REFERENCES (52)
1.
Feng G, Zhou L, Li F et al. Predictors of outcome in clinically diagnosed viral encephalitis patients: a 5-year prospective study. Biomed Res Int 2020; 2020: 2832418.
2.
Du FZ, Zhang X, Zheng XL et al. Cerebrospinal fluid CXCL13 concentration for diagnosis of neurosyphilis: a systematic review and meta-analysis. BMJ Open 2024; 14: e078527.
3.
Cellucci T, Van Mater H, Graus F, et al. Clinical approach to the diagnosis of autoimmune encephalitis in the pediatric patient. Neurol Neuroimmunol Neuroinflamm 2020; 7: e663.
4.
Alam AM, Easton A, Nicholson TR, et al. Encephalitis: diagnosis, management and recent advances in the field of encephalitides. Postgrad Med J 2023; 99: 815-25.
5.
Glaser CA, Honarmand S, Anderson LJ, et al. Beyond viruses: clinical profiles and etiologies associated with encephalitis. Clin Infect Dis 2006; 43: 1565-77.
6.
Perlejewski K, Radkowski M, Pawelczyk A, et al. Enteroviral central nervous system infections in patients with Lyme neuroborreliosis. Ticks Tick Borne Dis 2023; 14: 102253.
7.
Granerod J, Ambrose HE, Davies NW, et al. Causes of encephalitis and differences in their clinical presentations in England: a multicentre, population-based prospective study. Lancet Infect Dis 2010; 10: 835-44.
8.
Popiel M, Perlejewski K, Bednarska A, et al. Viral etiologies in adult patients with encephalitis in Poland: a prospective single center study. PLoS One 2017; 12: e0178481.
9.
Dutra LA, Abrantes F, Toso FF, et al. Autoimmune encephalitis: a review of diagnosis and treatment. Arq Neuropsiquiatr 2018; 76: 41-9.
10.
Huber AK, Irani DN. Targeting CXCL13 during neuroinflammation. Adv Neuroimmune Biol 2015; 6: 1-8.
11.
Lintner H, Hochgatterer-Rechberger P, Pischinger B, et al. Sensitivity and specificity of cerebrospinal fluid CXCL13 for diagnosing Lyme neuroborreliosis – a study on 1410 patients and review of the literature. J Neurol Sci 2020; 414: 116843.
12.
Barstad B, Tveitnes D, Dalen I, et al. The B-lymphocyte chemokine CXCL13 in the cerebrospinal fluid of children with Lyme neuroborreliosis: associations with clinical and laboratory variables. Infect Dis (Lond) 2019; 51: 856-63.
13.
Sellebjerg F, Bornsen L, Khademi M, et al. Increased cerebrospinal fluid concentrations of the chemokine CXCL13 in active MS. Neurology 2009; 73: 2003-10.
14.
Fissolo N, Pappolla A, Rio J, et al. Serum levels of CXCL13 are associated with teriflunomide response in patients with multiple sclerosis. Neurol Neuroimmunol Neuroinflamm 2023; 10: e200050.
15.
Karaaslan Z, Kurtuncu M, Akcay HI, et al. CXCL13 levels indicate treatment responsiveness to fingolimod in MS patients. Eur Neurol 2022; 85: 69-71.
16.
Pan Z, Zhu T, Liu Y, Zhang N. Role of the CXCL13/CXCR5 axis in autoimmune diseases. Front Immunol 2022; 13: 850998.
17.
Da Z, Li L, Zhu J, et al. CXCL13 promotes proliferation of mesangial cells by combination with CXCR5 in SLE J Immunol Res 2016; 2016: 2063985.
18.
Zhao J, Ye X, Zhang Z. The predictive value of serum soluble ICAM-1 and CXCL13 in the therapeutic response to TNF inhibitor in rheumatoid arthritis patients who are refractory to csDMARDs. Clin Rheumatol 2020; 39: 2573-81.
19.
Nocturne G, Seror R, Fogel O, et al. CXCL13 and CCL11 serum levels and lymphoma and disease activity in primary Sjogren’s syndrome. Arthritis Rheumatol 2015; 67: 3226-33.
20.
Zhang M, Guo J, Li H, et al. Expression of immune molecules CD25 and CXCL13 correlated with clinical severity of myasthenia gravis. J Mol Neurosci 2013; 50: 317-23.
21.
Singh UP, Singh NP, Murphy EA, et al. Chemokine and cytokine levels in inflammatory bowel disease patients. Cytokine 2016; 77: 44-9.
22.
Rupprecht TA, Manz KM, Fingerle V, et al. Diagnostic value of cerebrospinal fluid CXCL13 for acute Lyme neuroborreliosis. a systematic review and meta-analysis. Clin Microbiol Infect 2018; 24: 1234-40.
23.
Dersch R, Hottenrott T, Senel M, et al. The chemokine CXCL13 is elevated in the cerebrospinal fluid of patients with neurosyphilis. Fluids Barriers CNS 2015; 12: 12.
24.
Marra CM, Tantalo LC, Sahi SK, et al. CXCL13 as a cerebrospinal fluid marker for neurosyphilis in HIV-infected patients with syphilis. Sex Trans Dis 2010; 37: 283-7.
25.
Machura P, Gorka E, Mlynarczyk-Bonikowska B, et al. [Novel multiplex real-time PCR assay for detection and differentiation of herpes simplex virus type 1 and 2 DNA]. Med Dosw Mikrobiol 2015; 67: 125-32.
26.
Les K, Przybylski M, Dzieciatkowski T, Mlynarczyk G. Detection of human enteroviruses with real-time PCR assay using TaqMan fluorescent probe. Med Dosw Mikrobiol 2010; 62: 245-53.
27.
Mygland A, Ljostad U, Fingerle V, et al. EFNS guidelines on the diagnosis and management of European Lyme neuroborreliosis. Eur J Neurol 2010; 17: 8-16, e11-14.
28.
Wodecka B. flaB gene as a molecular marker for distinct identification of Borrelia species in environmental samples by the PCR-restriction fragment length polymorphism method. Appl Environ Microbiol 2011; 77: 7088-92.
29.
Tamura K, Stecher G, Kumar S. MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 2021; 38: 3022-7.
30.
Perlejewski K, Pawelczyk A, Bukowska-Osko I, et al. Search for Viral infections in cerebrospinal fluid from patients with autoimmune encephalitis. Open Forum Infect Dis 2020; 7: ofaa468.
31.
Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 2018; 17: 162-73.
32.
Sivina M, Xiao L, Kim E, et al. CXCL13 plasma levels function as a biomarker for disease activity in patients with chronic lymphocytic leukemia. Leukemia 2021; 35: 1610-20.
33.
Legler DF, Loetscher M, Roos RS, et al. B cell-attracting chemokine 1, a human CXC chemokine expressed in lymphoid tissues, selectively attracts B lymphocytes via BLR1/CXCR5. J Exp Med 1998; 187: 655-60.
34.
Carlsen HS, Baekkevold ES, Morton HC, et al. Monocyte-like and mature macrophages produce CXCL13 (B cell-attracting chemokine 1) in inflammatory lesions with lymphoid neogenesis. Blood 2004; 104: 3021-7.
35.
Harrer C, Otto F, Radlberger RF, et al. The CXCL13/CXCR5 immune axis in health and disease-implications for intrathecal B cell activities in neuroinflammation. Cells 2022; 11: 2649.
36.
Forster R, Mattis AE, Kremmer E, et al. A putative chemokine receptor, BLR1, directs B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen. Cell 1996; 87: 1037-47.
37.
Schmidt C, Plate A, Angele B, et al. A prospective study on the role of CXCL13 in Lyme neuroborreliosis. Neurology 2011; 76: 1051-8.
38.
Erhart DK, Klose V, Schaper T, et al. CXCL13 in cerebrospinal fluid: clinical value in a large cross-sectional study. Int J Mol Sci 2023; 25: 425.
39.
Remy MM, Schöbi N, Kottanattu L, et al. Cerebrospinal fluid CXCL13 as a diagnostic marker of neuroborreliosis in children: a retrospective case-control study. J Neuroinflammation 2017; 14: 173.
40.
Henningsson AJ, Gyllemark P, Lager M, et al. Evaluation of two assays for CXCL13 analysis in cerebrospinal fluid for laboratory diagnosis of Lyme neuroborreliosis. APMIS 2016; 124: 985-90.
41.
Wagner JN, Weis S, Kubasta C, et al. CXCL13 as a diagnostic marker of neuroborreliosis and other neuroinflammatory disorders in an unselected group of patients. J Neurol 2018; 265: 74-81.
42.
Pilz G, Steger R, Wipfler P, et al. Beyond LNB: real life data on occurrence and extent of CSF CXCL13 in neuroinflammatory diseases. J Neuroimmunol 2020; 338: 577087.
43.
Smiskova D, Dzupova O, Moravcova L, Picha D. Cerebrospinal fluid CXCL13 in non-borrelial central nervous system infections: contribution of CXCL13 to the differential diagnosis. Infect Dis (Lond) 2023; 55: 551-8.
44.
Hytönen J, Kortela E, Waris M, et al. CXCL13 and neopterin concentrations in cerebrospinal fluid of patients with Lyme neuroborreliosis and other diseases that cause neuroinflammation. J Neuroinflammation 2014; 11: 103.
45.
Leypoldt F, Hoftberger R, Titulaer MJ, et al. Investigations on CXCL13 in anti-N-methyl-D-aspartate receptor encephalitis: a potential biomarker of treatment response. JAMA Neurol 2015; 72: 180-6.
46.
Kothur K, Wienholt L, Mohammad SS, et al. Utility of CSF cytokine/chemokines as markers of active intrathecal inflammation: comparison of demyelinating, anti-NMDAR and enteroviral encephalitis. PLoS One 2016; 11:L e016156.
47.
Liba Z, Kayserova J, Elisak M, et al. Anti-N-methyl-D-aspartate receptor encephalitis: the clinical course in light of the chemokine and cytokine levels in cerebrospinal fluid. J Neuroinflammation 2016; 13: 55.
48.
Tilea B, Voidazan S, Balasa R, et al. CXCL13 levels are more increased in cerebrospinal fluid and plasma of patients with acute infectious than in non-infectious diseases of the central nervous system. Rev Rom Med Labor 2017; 25: 63-73.
49.
Lin YT, Yang X, Lv JW, et al. CXCL13 is a biomarker of anti-leucine-rich glioma-inactivated protein 1 encephalitis patients. Neuropsychiatr Dis Treat 2019; 15: 2909-15.
50.
Zajkowska J, Moniuszko-Malinowska A, Pancewicz SA, et al. Evaluation of CXCL10, CXCL11, CXCL12 and CXCL13 chemokines in serum and cerebrospinal fluid in patients with tick borne encephalitis (TBE). Adv Med Sci 2011; 56: 311-7.
51.
Pilz G, Wipfler P, Otto F, et al. Cerebrospinal fluid CXLC13 indicates disease course in neuroinfection: an observational study. J Neuroinflammation 2019; 16: 13.
52.
Bremell D, Mattsson N, Edsbagge M, et al. Cerebrospinal fluid CXCL13 in Lyme neuroborreliosis and asymptomatic HIV infection. Bmc Neurology 2013; 13: 2.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.