Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
ONCOLOGY / RESEARCH PAPER
Evaluating the Causal Relationship Between Carnitine-Related Metabolites and Breast Cancer: A Mendelian Randomization and Transcriptomic Analysis
1
The First Affiliated Hospital of Guangxi Medical University, China
2
The Sixth Affiliated Hospital of Guangxi Medical University, The First People's Hospital of Yulin, China
These authors had equal contribution to this work
Submission date: 2025-10-16
Final revision date: 2025-12-18
Acceptance date: 2025-12-29
Online publication date: 2026-04-03
Publication date: 2026-04-12
Corresponding author
Leifeng Liang
The Sixth Affiliated Hospital of Guangxi Medical University, The First People's Hospital of Yulin, China
The Sixth Affiliated Hospital of Guangxi Medical University, The First People's Hospital of Yulin, China
Arch Med Sci 2026;22(3)
KEYWORDS
female malignant tumoursfatty acid metabolic intermediatesgenetic causal inferencetumour microenvironment regulation
TOPICS
ABSTRACT
Introduction:
Breast cancer (BC) is the most common malignancy among women worldwide. Although observational studies have linked carnitine-related metabolites (CRMs) to BC, causal inference has been limited by confounding and reverse causality. This study utilized Mendelian randomization (MR) analysis to investigate the potential causal link between CRMs and BC.
Material and methods:
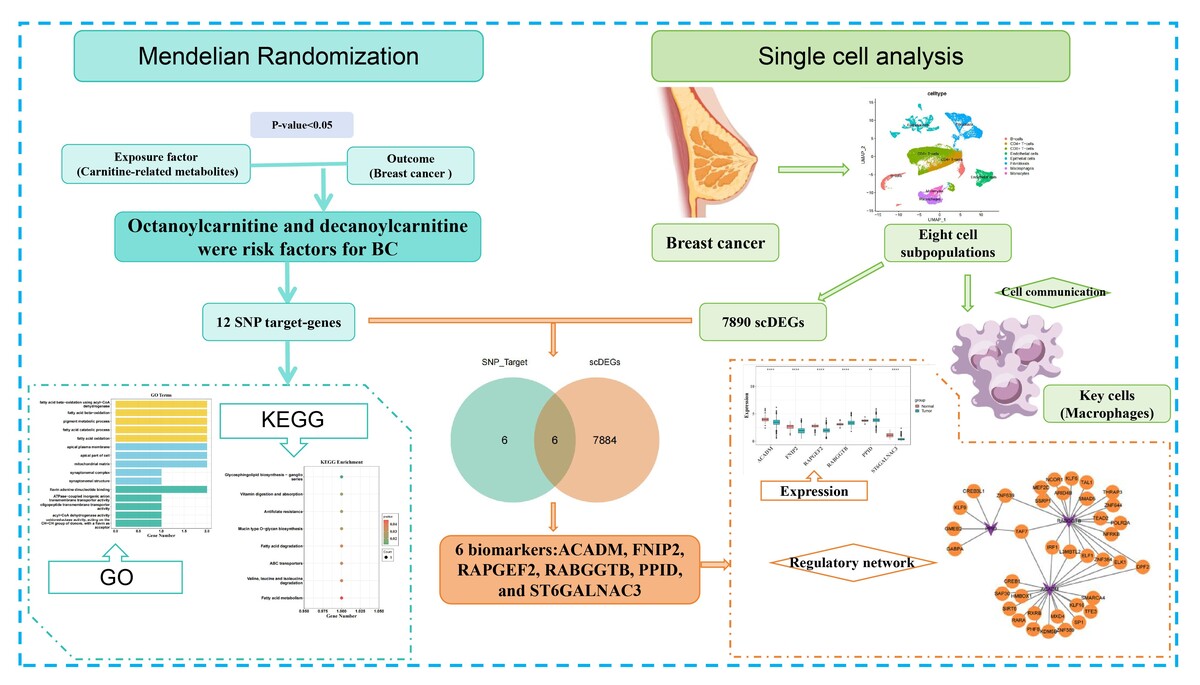
MR analysis was conducted using the Cancer Genome Atlas-BC and CRM-related datasets to explore the causal relationship between CRMs and BC. Variants were screened based on criteria encompassing genome-wide significance (p < 5×10⁻⁸), independence (r² < 0.001, kb=10000), and sufficient strength (F-statistic > 10). Various MR techniques, including Inverse-Variance Weighted, MR-Egger, simple mode, weighted mode, and weighted median approaches, were applied to assess these causal associations. Single-cell RNA sequencing (scRNA-seq) was employed to investigate the expression and biological functions of biomarkers linked to metabolites.
Results:
The MR analysis revealed a significant causal relationship between elevated levels of octanoylcarnitine and decanoylcarnitine and an increased risk of BC. Functional enrichment analysis identified 12 candidate genes associated with these metabolites, which are involved in fatty acid β-oxidation (FAO) pathways. ScRNA-seq analysis revealed eight distinct cell subpopulations, with macrophages exhibiting the highest intercellular communication. Six biomarkers were identified as potential contributors to BC development: ACADM, FNIP2, RAPGEF2, RABGGTB, PPID, and ST6GALNAC3.
Conclusions:
This study identifies octanoylcarnitine and decanoylcarnitine as significant causal risk factors for BC, linked to dysregulated FAO. Integration of single-cell transcriptomics revealed six CRM-associated biomarkers and their dynamic expression within tumor microenvironments. These findings provide novel candidates for BC diagnosis and personalized therapy, warranting further validation.
Breast cancer (BC) is the most common malignancy among women worldwide. Although observational studies have linked carnitine-related metabolites (CRMs) to BC, causal inference has been limited by confounding and reverse causality. This study utilized Mendelian randomization (MR) analysis to investigate the potential causal link between CRMs and BC.
Material and methods:
MR analysis was conducted using the Cancer Genome Atlas-BC and CRM-related datasets to explore the causal relationship between CRMs and BC. Variants were screened based on criteria encompassing genome-wide significance (p < 5×10⁻⁸), independence (r² < 0.001, kb=10000), and sufficient strength (F-statistic > 10). Various MR techniques, including Inverse-Variance Weighted, MR-Egger, simple mode, weighted mode, and weighted median approaches, were applied to assess these causal associations. Single-cell RNA sequencing (scRNA-seq) was employed to investigate the expression and biological functions of biomarkers linked to metabolites.
Results:
The MR analysis revealed a significant causal relationship between elevated levels of octanoylcarnitine and decanoylcarnitine and an increased risk of BC. Functional enrichment analysis identified 12 candidate genes associated with these metabolites, which are involved in fatty acid β-oxidation (FAO) pathways. ScRNA-seq analysis revealed eight distinct cell subpopulations, with macrophages exhibiting the highest intercellular communication. Six biomarkers were identified as potential contributors to BC development: ACADM, FNIP2, RAPGEF2, RABGGTB, PPID, and ST6GALNAC3.
Conclusions:
This study identifies octanoylcarnitine and decanoylcarnitine as significant causal risk factors for BC, linked to dysregulated FAO. Integration of single-cell transcriptomics revealed six CRM-associated biomarkers and their dynamic expression within tumor microenvironments. These findings provide novel candidates for BC diagnosis and personalized therapy, warranting further validation.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.