Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
UROLOGY / RESEARCH PAPER
NBPF1 acts as a tumor suppressor in prostate cancer by regulating the PI3K/AKT pathway
1
Department of Urology, Guangdong Provincial People's Hospital (Guangdong Academy of Medical Sciences), Southern Medical University, Guangzhou, 510080, China, China
2
Shantou University Medical College, Shantou, 515041, China, China
3
Department of Urology, Guangdong Cardiovascular Institute, Guangdong Provincial People's Hospital, Guangdong Academy of Medical Sciences, Guangzhou, 510080, China, China
4
Department of Immunology, School of Basic Medical Sciences, Southern Medical University, Guangzhou, 510515, China, China
5
Department of Urology, Northern Jiangsu People's Hospital Affiliated Yangzhou University, Yangzhou, 225001, China, China
These authors had equal contribution to this work
Submission date: 2025-09-08
Final revision date: 2025-11-18
Acceptance date: 2025-11-19
Online publication date: 2026-04-03
Publication date: 2026-04-12
Corresponding author
Yuming Yu
Department of Urology, Guangdong Provincial People's Hospital (Guangdong Academy of Medical Sciences), Southern Medical University, Guangzhou, 510080, China, China
Department of Urology, Guangdong Provincial People's Hospital (Guangdong Academy of Medical Sciences), Southern Medical University, Guangzhou, 510080, China, China
Arch Med Sci 2026;22(3)
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Prostate cancer (PCa) is a leading malignancy in men, yet the roles of many candidate regulators remain unclear. Neuroblastoma breakpoint family member 1 (NBPF1) has been implicated as a tumor suppressor in other cancers; however, its function and clinical relevance in PCa remain undefined.
Material and methods:
NBPF1 expression was examined in tissue microarrays (TMAs) comprising 77 PCa tumors and 73 normal prostate tissues using immunohistochemistry, and its correlation with clinicopathological features and outcomes was assessed. NBPF1 gain- and loss-of-function models were established in PCa cell lines (LNCaP, DU145, PC3, 22RV1) and compared with RWPE-1 cells. Proliferation (CCK-8, EdU, colony formation), migration/invasion (wound healing, Transwell), and protein expression (qRT-PCR, Western blot) were assessed. In vivo oncogenic effects were evaluated using PC3 xenografts (N=5 per group). RNA-seq of NBPF1-silenced PC3 cells, and KEGG analysis, was used to identify downstream pathways.
Results:
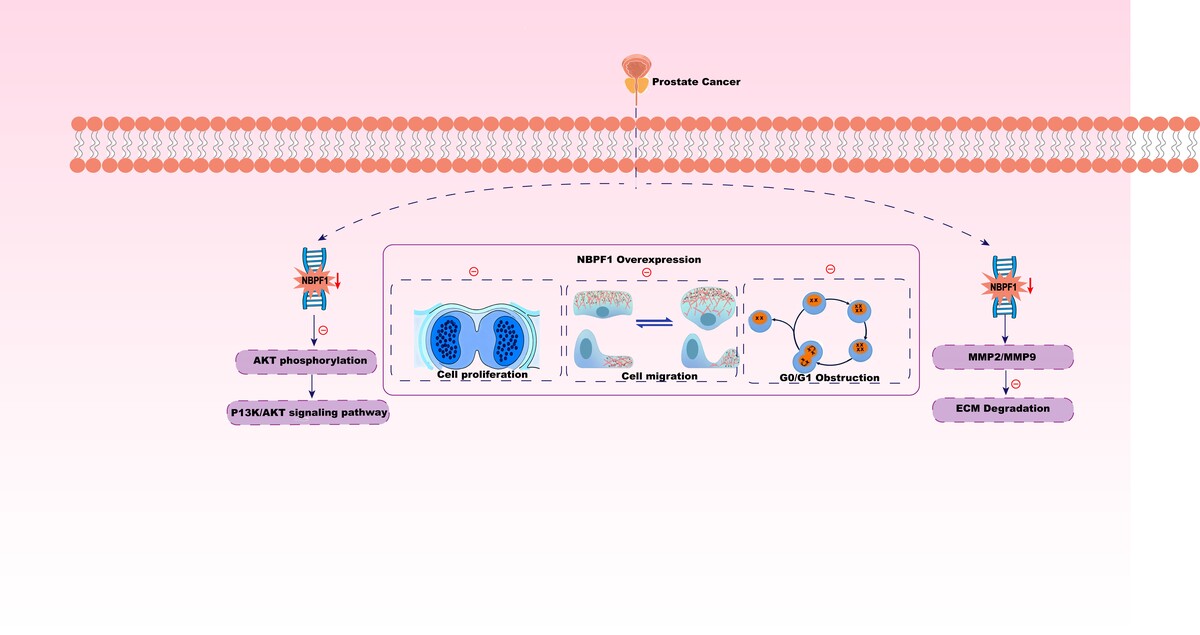
NBPF1 was markedly downregulated in PCa compared with normal prostate, and low expression was associated with adverse features (e.g., higher Gleason grade) and poorer prognosis. NBPF1 overexpression suppressed proliferation, migration, and invasion, whereas NBPF1 knockdown had the opposite effects. In mice, NBPF1 loss accelerated tumor growth. Transcriptomic profiling implicated the PI3K/AKT signaling pathway as a key downstream pathway; concordantly, NBPF1 loss increased p-AKT and elevated MMP2/MMP9 expression, while NBPF1 overexpression reduced pathway activation and protease expression.
Conclusions:
NBPF1 functions as a tumor suppressor in PCa, inhibiting progression at least partly through modulation of the PI3K/AKT pathway. NBPF1 may serve as a prognostic biomarker and a potential therapeutic target in prostate cancer.
Prostate cancer (PCa) is a leading malignancy in men, yet the roles of many candidate regulators remain unclear. Neuroblastoma breakpoint family member 1 (NBPF1) has been implicated as a tumor suppressor in other cancers; however, its function and clinical relevance in PCa remain undefined.
Material and methods:
NBPF1 expression was examined in tissue microarrays (TMAs) comprising 77 PCa tumors and 73 normal prostate tissues using immunohistochemistry, and its correlation with clinicopathological features and outcomes was assessed. NBPF1 gain- and loss-of-function models were established in PCa cell lines (LNCaP, DU145, PC3, 22RV1) and compared with RWPE-1 cells. Proliferation (CCK-8, EdU, colony formation), migration/invasion (wound healing, Transwell), and protein expression (qRT-PCR, Western blot) were assessed. In vivo oncogenic effects were evaluated using PC3 xenografts (N=5 per group). RNA-seq of NBPF1-silenced PC3 cells, and KEGG analysis, was used to identify downstream pathways.
Results:
NBPF1 was markedly downregulated in PCa compared with normal prostate, and low expression was associated with adverse features (e.g., higher Gleason grade) and poorer prognosis. NBPF1 overexpression suppressed proliferation, migration, and invasion, whereas NBPF1 knockdown had the opposite effects. In mice, NBPF1 loss accelerated tumor growth. Transcriptomic profiling implicated the PI3K/AKT signaling pathway as a key downstream pathway; concordantly, NBPF1 loss increased p-AKT and elevated MMP2/MMP9 expression, while NBPF1 overexpression reduced pathway activation and protease expression.
Conclusions:
NBPF1 functions as a tumor suppressor in PCa, inhibiting progression at least partly through modulation of the PI3K/AKT pathway. NBPF1 may serve as a prognostic biomarker and a potential therapeutic target in prostate cancer.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.