Various terms have been employed to describe persistence of symptoms after COVID-19 (C-19) based on the duration and insistence of symptoms, including “long COVID (L-C19)”, “post-COVID-19 syndrome (PCS)”, “post-COVID-19 manifestations”, “post-acute sequelae of SARS-CoV-2 infection (PASC)”, “persistent COVID-19 symptoms”, and “long haulers” [1–3]. The definition proposed by the World Health Organisation (WHO) states that the presence of clinical symptoms in infected individuals that persist for at least 3 months after disease onset, or the emergence of new symptoms lasting more than 2 months without any other explanation and not attributable to other existing pathologies, is defined as long COVID-19 (L-C19) [3].
Based on a conservative incidence rate of 10% among infected individuals and over 651 million documented COVID-19 cases worldwide, the prevalence of long COVID is estimated to be at least 65 million individuals globally. However, due to numerous undocumented cases the actual number is probably much higher [4]. The incidence of post-COVID complications varies according to different situations: among vaccinated cases its incidence is estimated at 10–12% [5, 6], and 10–30% among non-hospitalised cases and 50–70% among hospitalised cases [7, 8].
Long COVID affects individuals of all ages and severities of the acute phase of the disease. Even asymptomatic individuals and those with a mild course of COVID-19 can develop severe CV complications [9]. The factors contributing to these outcomes have not been extensively investigated to date. Risk factors for long COVID include female sex, age, and moderate to severe COVID-19 infection [10]. However, long COVID can manifest irrespective of the initial disease severity [9]. Additionally, long COVID is linked to diminished health-related quality of life in individuals with both moderate to severe and mild or asymptomatic COVID-19. On the other hand, it remains unclear which patients are at higher risk for long-COVID cardiovascular complications.
Therefore, our objective was to identify risk factors associated with the development of cardiovascular complications requiring hospitalisation, following COVID-19.
Methods
We enrolled all consecutive patients who were admitted to the Department of Cardiology at the Polish Mother’s Memorial Hospital – Research Institute (PMMHRI) due to cardiovascular symptoms following COVID-19 infection (meeting the criteria of long-COVID diagnosis) between October 2019 and December 2023. In all hospitalised patients, laboratory tests, 24-hour ECG monitoring, and echocardiography were performed. We collected data regarding post-COVID complications including thromboembolic complications, myocarditis, exacerbation of angina pectoris, new onset of atrial fibrillation, new onset of non-sustained ventricular tachycardia, new onset of supraventricular tachycardia (SVT), and decrease in ejection fraction. SARS-CoV2 infection was confirmed with real-life PCR testing. All data were retrospectively collected from medical records of our institution and were anonymized.
Statistical analysis
We performed statistical analyses using Stata/BE 18.0 software. The Shapiro-Wilk test was employed to evaluate the normality of the distribution. Results were expressed as mean ± standard deviation (SD) for continuous variables, or as median values, and as percentages for categorical variables. Group comparisons were conducted using Student’s t-test for normally distributed variables and the Mann-Whitney U test for non-normally distributed variables. Correlations were analysed using either the Spearman or Pearson correlation tests. The χ2 test, with Yates correction applied when necessary, was utilised to compare categorical variables. To explore the relationship between cardiovascular complications and risk factors, we conducted both univariate and multivariate linear regression analyses. Multivariable regression models were adjusted for gender and age. Cut-off values were estimated using Youden’s index. We considered p-values less than 0.05 as statistically significant for all calculations.
Results
We analysed data from 504 consecutive patients (mean age: 53 ±17 years, with 36.3% males) who were hospitalised due to cardiovascular symptoms post COVID-19 between October 2020 and December 2023 as part of the LATE-COVID study (ClinicalTrials.gov: NCT04799444) [11, 12]. In the studied group symptoms were still present 5 (2–10) months after the acute phase of the infection. Among cardiovascular complications following COVID-19: thromboembolic complications occurred in 1.59% of patients, myocarditis was diagnosed in 0.99% of patients, exacerbation of angina pectoris requiring revascularisation in 6.15%, and without revascularisation in 24.4%, new onset of atrial fibrillation in 1.40%, non-sustained ventricular tachycardia in 2.78%, new onset of SVT in 11.78%, and decrease in ejection fraction (more than 10% reduction from the pre-disease value) in 4.38% of admitted patients. Some patients (10.7%) experienced more than one of the above complications. In general, complications occurred in approximately 41% of all patients. Based on multivariable regression analysis, the following risk factors played the most important role for patients to develop late cardiovascular complications following COVID 19: weight, height, body mass index (BMI), and systolic blood pressure (BP). From echocardiographic parameters, left ventricular end-diastolic volume (LVEDV) and left ventricular end-systolic volume (LVESV) had statistical significance. Among comorbidities, coronary artery disease (CAD), heart failure (HF), asthma/chronic obstructive pulmonary disease (COPD), and adult congenital heart disease (ACHD) predicted post-COVID complications, but only HF, cancer and asthma/COPD remained statistically significant after adjustment for age and sex. From laboratory tests, white blood count (WBC), urea, TSH, and troponin level predicted cardiovascular complications; however, after adjustment for age and gender it did not remain statistically significant (Table I).
Table I
Univariate and multivariate analysis
| Parameter | Univariable regression* | Multivariable regression# | ||
|---|---|---|---|---|
| OR (95% CI) | P-value | OR (95% CI) | P-value | |
| Weight | 1.01 (1.00–1.03) | 0.006 | 1.01 (1.00–1.02) | 0.017 |
| Height | 1.00 (0.99–1.02) | 0.577 | 1.04 (1.01–1.07) | 0.018 |
| Body mass index | 1.06 (1.02–1.09) | 0.001 | 1.04 (1.00–1.07) | 0,049 |
| Systolic blood pressure | 1.02 (1.01–1.03) | 0.003 | 1.01 (1.00–1.02) | 0.021 |
| Diastolic blood pressure | 1.00 (0.98–1.01) | 0.836 | 1.00 (0.98–1.02) | 0.781 |
| Heart rate (HR) | 1.00 (0.98–1.01) | 0.644 | 1.00 (0.99–1.01) | 0.909 |
| Smoking | 1.26 (1.02–1.56) | 0.029 | 1.13 (0.90–1.41) | 0.279 |
| Symptoms | ||||
| Dyspnoea | 1.30 (0.86–1.96) | 0.214 | 1.12 (0.73–1.72) | 0.616 |
| Physical activity intolerance | 1.68 (1.17–2.41) | 0.005 | 1.35 (0.92–1.98) | 0.123 |
| Asthma cardiale | 2.34 (1.63–3.41) | <0.001 | 2.32 (1.57–3.42) | <0.001 |
| Blood pressure disturbances | 1.45 (0.93–2.28) | 0.099 | 1.35 (0.85–2.14) | 0.203 |
| Palpitation | 1.10 (0.73–1.59) | 0.632 | 1.10 (0.74–1.61) | 0.644 |
| Weight loss | 1.55 (0.73–3.29) | 0.251 | 1.30 (0.60–2.83) | 0.503 |
| Brain fog | 0.64 (0.24–1.72) | 0.376 | 0.51 (0.18–1.41) | 0.195 |
| General malaise | 1.30 (0.74–2.59) | 0.356 | 1.31 (0.73–2.35) | 0.360 |
| Headache | 0.90 (0.38–2.18) | 0.816 | 1.03 (0.42–2.49) | 0.952 |
| Ascites | 1.67 (0.95–2.92) | 0.075 | 1.32 (0.74–2.36) | 0.343 |
| Comorbidities | ||||
| Coronary artery disease | 2.06 (1.22–3.50) | 0.007 | 1.32 (0.76–2.32) | 0.326 |
| Stroke | 1.33 (0.61–2.89) | 0.471 | 1.08 (0.48–2.40) | 0.857 |
| Heart failure | 2.20 (1.35–3.59) | 0.002 | 1.63 (0.97–2.73) | 0.064 |
| Hypertension | 1.42 (0.99–2.02) | 0.056 | 0.92 (0.62–1.38) | 0.691 |
| Cancer | 0.67 (0.37–1.24) | 0.202 | 0.42 (0.22–0.80) | 0.008 |
| Pulmonary diseases | 1.81 (1.05–3.12) | 0.034 | 1.66 (0.95–2.93) | 0.077 |
| Diabetes mellitus | 1.60 (0.94–2.73) | 0.083 | 1.14 (0.64–2.01) | 0.657 |
| Atrial fibrillation | 1.46 (0.88–2.41) | 0.142 | 0.89 (0.51–1.53) | 0.663 |
| Arrhythmias | 1.04 (0.67–1.62) | 0.847 | 1.05 (0.66–1.65) | 0.846 |
| ACHD | 0.43 (0.22–0.82) | 0.011 | 0.57 (0.28–1.17) | 0.125 |
| Left bundle branch block | 0.71 (0.21–2.38) | 0.577 | 0.81 (0.22–2.87) | 0.741 |
| Thyroid disease | 1.00 (0.63–1.57) | 0.995 | 0.90 (0.55–1.46) | 0.671 |
| 24 hour ECG monitoring | ||||
| Minimal heart rate | 1.01 (0.99–1.03) | 0.412 | 1.00 (0.98–1.02) | 0.683 |
| Maximal heart rate | 1.00 (0.99–1.01) | 0.449 | 1.00 (0.99–1.01) | 0.853 |
| Mean heart rate | 1.00 (0.98–1.02) | 0.926 | 1.01 (0.99–1.03) | 0.543 |
| Supraventricular extrasystoles | 1.00 (1.00–1.00) | 0.398 | 1.00 (1.00–1.00) | 0.389 |
| Ventricular extrasystoles | 1.00 (1.00–1.00) | 0.050 | 1.00 (1.00–1.00) | 0.112 |
| Echocardiography | ||||
| Left atrial volume index | 1.01 (1.00–1.02) | 0.151 | 1.00 (0.09–1.01) | 0.749 |
| Right atrial area | 1.03 (1.00–1.07) | 0.079 | 1.01 (0.97–1.05) | 0.577 |
| LVEDV | 1.00 (1.00–1.01) | 0.145 | 1.01 (1.00–1.01) | 0.012 |
| LVESV | 1.01 (1.00–1.01) | 0.083 | 1.01 (1.00–1.02) | 0.024 |
| Ejection fraction | 0.98 (0.95–1.00) | 0.032 | 0.98 (0.95–1.00) | 0.057 |
| Tricuspid annular motion | 1.02 (0.98–1.07) | 0.369 | 1.01 (0.96–1.06) | 0.668 |
| S’ | 1.06 (0.99–1.13) | 0.079 | 1.04 (0.98–1.11) | 0.198 |
| Laboratory tests | ||||
| White blood count | 1.01 (0.95–1.08) | 0.657 | 1.03 (0.96–1.09) | 0.432 |
| Red blood count | 0.68 (0.49–0.96) | 0.027 | 0.91 (0.67–1.24) | 0.554 |
| Haemoglobin | 0.90 (0.80–1.00) | 0.053 | 0.98 (0.87–1.11) | 0.756 |
| Platelets | 1.00 (1.00–1.00) | 0.644 | 1.00 (1.00–1.01) | 0.103 |
| Total cholesterol | 1.00 (1.00–1.00) | 0.141 | 1.00 (1.00–1.00) | 0.297 |
| Low density lipoproteins | 1.00 (1.00–1.01) | 0.217 | 1.00 (0.99–1.01) | 0.603 |
| High density lipoproteins | 1.00 (0.99–1.00) | 0.349 | 0.99 (0.98–1.00) | 0.249 |
| Triglicerides | 1.00 (1.00–1.00) | 0.867 | 1.00 (1.00–1.00) | 0.845 |
| Creatinine | 1.10 (0.87–1.41) | 0.425 | 1.06 (0.83–1.35) | 0.641 |
| Glomerular filtration rate | 0.99 (0.98–1.00) | 0.064 | 1.01 (1.00–1.02) | 0.112 |
| Urea | 1.02 (1.00–1.03) | 0.020 | 1.00 (1.00–1.02) | 0.660 |
| Alanine aminotransferase | 1.00 (1.00–1.01) | 0.802 | 1.00 (1.00–1.01) | 0.486 |
| Aspartate aminotransferase | 1.01 (1.00–1.02) | 0.130 | 1.01 (1.00–1.02) | 0.046 |
| C-reactive protein | 1.02 (0.75–1.37) | 0.913 | 1.00 (0.72–1.38) | 0.993 |
| D-dimer | 1.00 (1.00–1.00) | 0.646 | 1.00 (1.00–1.00) | 0.368 |
| NTproBNP | 1.00 (1.00–1.00) | 0.312 | 1.00 (1.00–1.00) | 0.958 |
| Troponin | 1.02 (1.00–1.04) | 0.030 | 1.01 (1.00–1.03) | 0.081 |
| TSH | 0.84 (0.72–0.99) | 0.041 | 0.90 (0.76–1.06) | 0.189 |
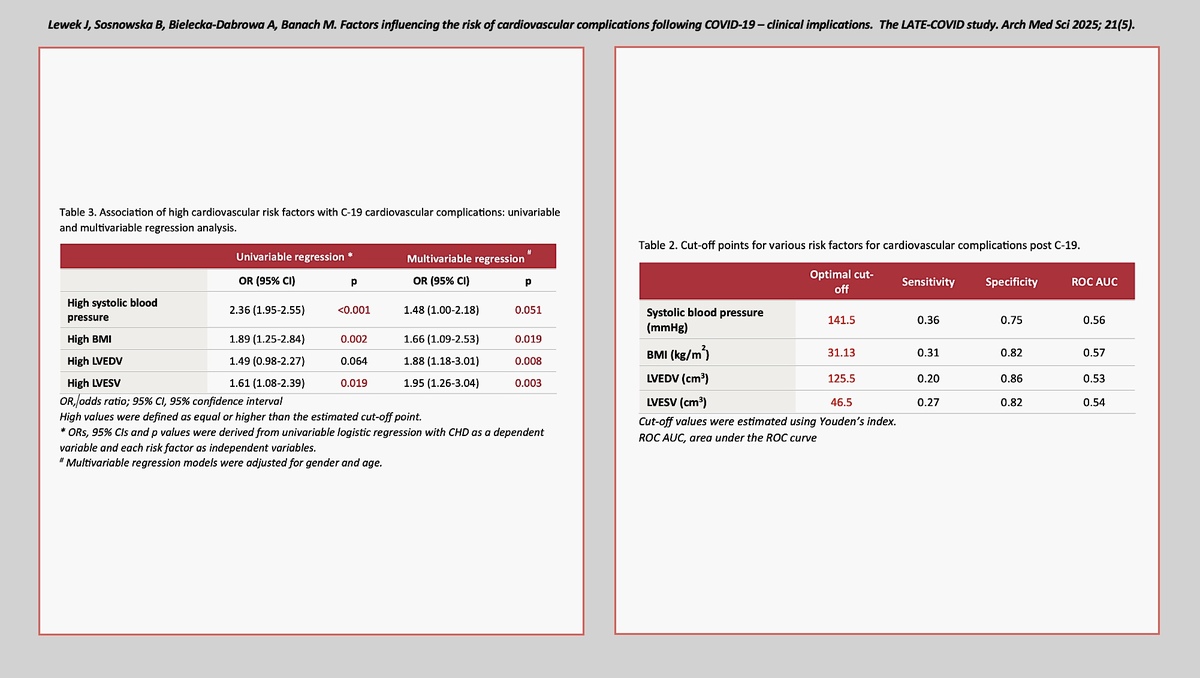
Among continuous factors influencing the development of long COVID cardiovascular complications, we calculated optimal cut-off values for systolic blood pressure, BMI, LVEDV, and LVESV (Table II).
Table II
Cut-off points for various risk factors for cardiovascular complications post C-19
| Parameter | Optimal cut-off | Sensitivity | Specificity | ROC AUC |
|---|---|---|---|---|
| Systolic blood pressure [mm Hg] | 141.5 | 0.36 | 0.75 | 0.56 |
| BMI [kg/m2] | 31.13 | 0.31 | 0.82 | 0.57 |
| LVEDV [cm3] | 125.5 | 0.20 | 0.86 | 0.53 |
| LVESV [cm3] | 46.5 | 0.27 | 0.82 | 0.54 |
Hypertension, defined by an systolic blood pressure (SBP) of 141.5 mm Hg, showed a sensitivity of 0.36 and a specificity of 0.75, with a ROC AUC of 0.56. Although the sensitivity is relatively low, the specificity indicates a moderate ability to correctly identify individuals without adverse outcomes. This suggests that while not all hypertensive patients will experience severe COVID-19 outcomes, those who do not have hypertension are less likely to face such complications. The area under the receiver operating characteristic curve (ROC AUC) area under the curve (AUC) value, while not high, still indicates some discriminatory power of SBP in predicting adverse outcomes.
Similarly, obesity, defined by a BMI of 31.13 kg/m², had a sensitivity of 0.31 and a specificity of 0.82, with a ROC AUC of 0.57. The higher specificity compared to sensitivity suggests that BMI is a better predictor for excluding individuals who will not experience severe outcomes rather than identifying those who will. The ROC AUC value indicates a modest ability to discriminate between those who will and will not experience adverse outcomes.
The LVEDV and LVESV were also evaluated as potential predictors of cardiovascular complications in long COVID. The optimal cut-off for LVEDV was 125.5 cm³, with a sensitivity of 0.20 and a specificity of 0.86, and a ROC AUC of 0.53. For LVESV, the cut-off was 46.5 cm³, with a sensitivity of 0.27 and a specificity of 0.82, and a ROC AUC of 0.54. These values suggest that while these measures have high specificity, their low sensitivity limits their utility in predicting adverse outcomes. However, their high specificity indicates that individuals with values below these cut-offs are less likely to develop cardiovascular complications.
The association of high cardiovascular risk factors with COVID-19 cardiovascular complications through univariate and multivariate regression analyses are presented in Table III. Univariable regression analysis showed that high systolic blood pressure (OR = 2.36, 95% CI: 1.95–2.55, p < 0.001), high BMI (OR = 1.89, 95% CI: 1.25–2.84, p = 0.002), high LVEDV (OR = 1.49, 95% CI: 0.98–2.27, p = 0.064), and high LVESV (OR = 1.61, 95% CI: 1.08–2.39, p = 0.019) were significantly associated with increased odds of cardiovascular complications. In multivariable regression models adjusted for gender and age, high systolic blood pressure (OR = 1.48, 95% CI: 1.00–2.18, p = 0.051), high BMI (OR = 1.66, 95% CI: 1.09–2.53, p = 0.019), high LVEDV (OR = 1.88, 95% CI: 1.18–3.01, p = 0.008), and high LVESV (OR = 1.95, 95% CI: 1.26–3.04, p = 0.003) remained significant predictors.
Table III
Association of high cardiovascular risk factors with C-19 cardiovascular complications: univariate and multivariate regression analysis
| Parameter | Univariate regression* | Multivariate regression# | ||
|---|---|---|---|---|
| OR (95% CI) | P-value | OR (95% CI) | P-value | |
| High systolic blood pressure | 2.36 (1.95–2.55) | < 0.001 | 1.48 (1.00–2.18) | 0.051 |
| High BMI | 1.89 (1.25–2.84) | 0.002 | 1.66 (1.09–2.53) | 0.019 |
| High LVEDV | 1.49 (0.98–2.27) | 0.064 | 1.88 (1.18–3.01) | 0.008 |
| High LVESV | 1.61 (1.08–2.39) | 0.019 | 1.95 (1.26–3.04) | 0.003 |
Discussion
In our study, we observed that symptoms persisted for a duration of 5 months (ranging from 2 to 10 months) following the acute phase of COVID-19 infection. This finding aligns with the broader spectrum of post-COVID-19 symptom persistence reported in the literature.
Similarly, the complications of COVID-19 observed in our study are similar to those previously described. In 2022, the American College of Cardiology published its inaugural expert consensus on the cardiovascular consequences of COVID-19, addressing both short-term and long-term effects [13]. Numerous papers address the issue of cardiovascular complications. However, most of them describe the acute phase of COVID-19. Data regarding persistence of cardiovascular symptoms are limited.
Cardiological complications of a past SARS-CoV-2 infection include the following: thromboembolic complications, myocardial infarction, cardiomyopathy, arrhythmias, and HF [14, 15]. The prevalence of those diseases varies depending on the population studied [14, 16–18]. The cardiovascular complications observed in our study following COVID-19 infection align with the range of symptoms reported in existing literature, although some variations are notable [15, 19, 20]. Our findings indicate that thromboembolic complications occurred in 1.59% of patients, myocarditis in 0.99%, and new onset of atrial fibrillation in 1.40%. These rates are generally lower compared to the broader ranges reported in the literature, where myocarditis is noted in 4–60% of cases and atrial fibrillation in 18% [21]. Notably, our study found a higher incidence of supraventricular tachycardia (11.78%) compared to the reported 4–6% for ventricular tachycardia/fibrillation [21, 22]. Additionally, exacerbation of angina pectoris requiring revascularisation was observed in 6.15% of patients, and without revascularisation in 24.4%, highlighting a significant burden of ischaemic complications. The overall complication rate of approximately 41% in our cohort underscores the substantial cardiovascular impact of COVID-19, consistent with the high prevalence of myocardial oedema and abnormalities on cardiac magnetic resonance reported in up to 78% of cases. These findings emphasise the need for vigilant cardiovascular monitoring and management in patients recovering from COVID-19.
In our study, we identified weight, height, BMI, BP, LVEDV, LVESV, and comorbidities such as CAD, HF, asthma/COPD, and ACHD as predictors of cardiovascular complications. The role of comorbidities was emphasised in previous publications. Patients with pre-existing cardiovascular disease are at higher risk of developing further cardiovascular complications [23]. In other studies key predictors of cardiovascular complications post COVID-19 are older age (particularly over 60 years), male sex, body mass index, smoking, medical history, and medication [23]. A systematic review by Tsampasian et al. identified pre-existing conditions such as coronary syndromes and heart failure as well as diabetes and obesity as significant risk factors for cardiovascular complications with conflicting results regarding other comorbidities [24].
Our paper is unique because it includes patients who were admitted to the cardiology department due to cardiovascular symptoms following COVID-19. However, not all those symptoms remained clinically significant. From the population studied we extracted a population of patients with current complications after SARS-CoV-2 infection. However, there are also some limitations that should be mentioned. Given the cross-sectional nature of this study, we are unable to draw definitive conclusions regarding the causality of the observed associations. Nonetheless, our findings may assist in the preliminary evaluation and prediction of risk factors for cardiovascular diseases in post-COVID-19 convalescent patients who were hospitalised due to their symptoms, potentially indicating the emergence of a new cardiovascular condition [25, 26]. Other limitations are the absence of a control group comprising individuals without SARS-CoV-2 infection and the lack of follow-up period to fully comprehend the progression of newly diagnosed cardiovascular diseases.
In conclusion, the incidence of delayed cardiovascular complications in long COVID is notably high. We identified the following risk factors/conditions that independently affected cardiovascular complications: BMI, SBP, cardiac asthma, LVEDV, LVESV, HF, cancer, and asthma/COPD. These findings highlight the necessity of monitoring these risk factors in post-COVID-19 patients to mitigate the risk of developing cardiovascular complications.