Inpatient falls remain a major patient safety concern and contribute to fractures, prolonged hospitalization, and mortality [1]. Medication exposure is one of the most common modifiable risk factors for inpatient falls [2]. Opioids and anxiolytics/hypnotics have been consistently linked to falls through impaired cognition and balance [3, 4]. In contrast, the association between gabapentinoid use and fall risk remains relatively inconclusive in inpatient settings despite their frequent prescription for pain management [5–7]. This uncertainty is clinically important in hospitalized adults, who often have multiple comorbidities and concurrent use of other central nervous system (CNS) – acting medications [8].
To address this gap, we conducted a retrospective, single-center propensity score–matched case–control study to evaluate the associations between inpatient use of specific medication classes – including gabapentinoids – and falls. By comparing medication exposure between matched fall and non-fall patients and adjusting for key clinical confounders, we aimed to inform inpatient medication review and fall-prevention strategies.
Methods
Study design and population
We conducted a retrospective, single-center matched case–control study of adults (≥ 20 years) admitted between January 2019 and December 2023. Inpatient falls were identified using the hospital adverse-event reporting system. Controls were patients without inpatient falls during the same period and were propensity score–matched to fall cases (1 : 4) on age, sex, department of admission, and admission Morse Fall Scale (MFS; a validated score reflecting baseline inpatient fall susceptibility) [9, 10]. For controls, the index date was defined as the midpoint of hospitalization. We excluded patients aged < 20 years, those with hospitalization ≤ 2 days, patients who were still hospitalized at the time of data extraction, and patients who were bedridden or unconscious at admission.
Data sources and measures
Data were extracted from the electronic medical records. Variables included age, sex, department of admission, hospital unit, total length of stay (LOS), surgery during hospitalization, admission MFS score, and body mass index (BMI) at admission. Laboratory values obtained on the fall date (cases) or index date (controls) included hemoglobin, serum sodium, serum creatinine, white blood cell count, platelet count, and serum albumin. These measures were summarized as continuous variables where appropriate and as predefined binary indicators (anemia, hyponatremia, elevated serum creatinine, leukocytosis, thrombocytopenia, and hypoalbuminemia). Blood glucose at admission was also collected/measured. Comorbidity burden was assessed using the Charlson Comorbidity Index (CCI) based on the International Classification of Diseases, 10th Revision codes documented within 5 years before admission [11].
Medication exposure
Medication exposure was ascertained using the World Health Organization Anatomical Therapeutic Chemical classification [12]. Medication use was assessed on the day before the fall or on the index date. We evaluated exposure to antiepileptic drugs (N03AB, N03AF, N03AX, N03AG), antipsychotics (N05A), anxiolytics/hypnotics (N03AE, N05B, N05C), gabapentinoids (N02BF), tricyclic antidepressants (N06AA), alpha blockers (G04CA), diuretics (C03), β-blockers (C07), calcium channel blockers (C08), renin–angiotensin system agents (C09), antihistamines (R06A), and opioids (N02A) [12]. Each medication class was treated as a binary exposure (use vs. non-use).
Statistical analysis
Propensity scores were estimated using logistic regression with age, sex, department of admission, and admission MFS score. We performed 1 : 4 nearest-neighbor matching without replacement using a caliper of 0.2 standard deviations of the logit of the propensity score, and assessed post-matching balance using standardized mean differences (SMDs), with SMD < 0.1 indicating negligible imbalance [10, 13]. Propensity scores were used for matching only and were not included in subsequent regression models. We used conditional logistic regression stratified by matched sets to estimate odds ratios (ORs) with 95% confidence intervals (CIs) and p-values. Primary multivariable models were adjusted for BMI at admission, surgery during hospitalization, anemia, hyponatremia, hypoalbuminemia, and thrombocytopenia; total LOS was not included in the primary models to avoid potential overadjustment, but was examined in sensitivity analyses. Additional sensitivity analyses included adjustment for the number of concomitant medications, co-prescribing analyses of gabapentinoids with opioids, and subgroup analyses in patients aged > 75 years. Gabapentinoids, opioids, and anxiolytics/hypnotics were prespecified as primary exposures, while other medication classes were considered exploratory. Missing continuous variables were imputed using mean values, and missing categorical variables were coded as 0. All analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics of the study population
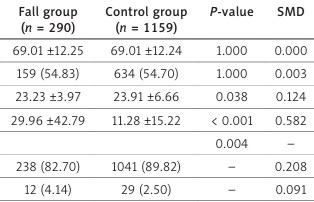
The study included 290 patients in the fall group and 1,159 matched controls (Table I). Mean age was identical in both groups, and the proportion of male patients was comparable. Compared with controls, the fall group had a lower BMI at admission and a longer LOS. Distribution across hospital units differed; fall cases were less likely to be in the general ward and more often in intensive care unit, emergency room, and stroke unit. Surgery during hospitalization was more common in the fall group. Laboratory abnormalities were also more frequent among fall cases, including anemia, hyponatremia, thrombocytopenia, and hypoalbuminemia. Comorbidity burden assessed by the CCI was higher in the fall group.
Table I
Baseline characteristics of the fall group and the matched control group
Association between medication use and inpatient falls
In unadjusted analyses, α blockers, opioids, gabapentinoids, antiepileptic drugs, antipsychotics, and anxiolytics/hypnotics were associated with higher odds of inpatient falls (Table II). In the primary adjusted analysis using conditional logistic regression stratified by matched sets, opioids, gabapentinoids, and anxiolytics/hypnotics remained associated with falls (Table II). Other medication classes showed no significant associations in adjusted models.
Table II
Association between medication use and inpatient falls
| Medication | Unadjusted odsds ratio (95% confidence interval) | P-value | Adjusted odds ratio (95% confidence interval)* | P-value |
|---|---|---|---|---|
| Diuretics | 1.372 (0.998–1.887) | 0.052 | 1.088 (0.767–1.545) | 0.635 |
| B-blockers | 1.453 (0.984–2.144) | 0.060 | 1.245 (0.811–1.910) | 0.316 |
| Calcium channel blockers | 1.234 (0.905–1.681) | 0.183 | 1.294 (0.924–1.814) | 0.134 |
| Renin-angiotensin system blockers | 1.043 (0.770–1.412) | 0.785 | 0.932 (0.669–1.298) | 0.676 |
| A-blockers | 1.479 (1.028–2.128) | 0.035 | 1.225 (0.822–1.825) | 0.318 |
| Antihistamines | 1.248 (0.898–1.733) | 0.187 | 1.335 (0.927–1.921) | 0.118 |
| Opioids | 2.038 (1.569–2.648) | < 0.001 | 1.666 (1.251–2.219) | 0.001 |
| Gabapentinoids | 1.892 (1.311–2.729) | < 0.001 | 1.884 (1.255–2.826) | 0.002 |
| Antiepileptic drugs | 2.038 (1.216–3.416) | 0.007 | 1.927 (0.991–3.505) | 0.094 |
| Antipsychotics | 1.802 (1.209–2.686) | 0.004 | 1.406 (0.908–2.176) | 0.128 |
| Anxiolytics/hypnotics | 1.823 (1.325–2.508) | < 0.001 | 1.947 (1.367–2.772) | < 0.001 |
| Tricyclic antidepressants | 1.553 (0.809–2.982) | 0.186 | 2.131 (0.937–4.580) | 0.109 |
Sensitivity analyses
Results were consistent in a sensitivity analysis additionally adjusting for the total length of stay (Supplementary Table SI). Additional adjustment for the number of concomitant medications did not materially change the associations for opioids, gabapentinoids, or anxiolytics/hypnotics. Concurrent gabapentinoid and opioid use was associated with higher odds of falls (adjusted OR = 2.524; 95% CI: 1.376–4.629; p = 0.003), whereas the gabapentinoid-by-opioid interaction term was not significant (p = 0.91). Subgroup analyses by age yielded directionally similar results (Supplementary Table SII).
Discussion
In this propensity score–matched case–control study of hospitalized adults, use of opioids, gabapentinoids, and anxiolytics/hypnotics was associated with falls after adjustment. The associations for opioids and anxiolytics/hypnotics are consistent with prior evidence linking these agents to falls through sedation and balance disturbance [4, 14]. Beyond reproducing these established risks, a key finding was that gabapentinoid exposure was also associated with inpatient falls in this study, adding evidence to an association that has remained with heterogeneous findings across prior studies [5, 6]. Collectively, our results suggest that gabapentinoids should be considered among medications that may increase fall risk in hospitalized adults. Potential mechanisms for a gabapentinoid–fall association likely reflect known CNS adverse effects, including dizziness, somnolence, and impaired postural control [5–7, 15]. These effects may be more consequential in older inpatients with reduced physiologic reserve and baseline gait impairment. Because gabapentinoids are predominantly renally excreted, impaired renal function may also potentiate dose-related adverse effects [15]. Prior studies have also raised concerns that co-prescribing gabapentinoids with opioids or other CNS depressants may increase clinically meaningful adverse outcomes [3, 7]. In our sensitivity analyses, concurrent gabapentinoid and opioid exposure was associated with higher odds of falls than neither exposure, supporting increased vigilance when these agents are co-prescribed; however, the interaction test was not significant, and synergistic effects should not be assumed.
Methodologically, matching on age, sex, department of admission, and admission MFS score improved comparability and partially addressed baseline fall susceptibility. Conditional logistic regression accounted for the matched design and adjusted for clinically relevant covariates, and sensitivity analyses incorporating a proxy for polypharmacy yielded broadly consistent findings for the main CNS-acting/active medications. Nevertheless, because gabapentinoids are often co-prescribed with other CNS-active medications, residual confounding related to CNS medication burden may persist. Accordingly, our findings should be interpreted as associations observed in routine inpatient prescribing patterns rather than definitive evidence of fully independent drug effects. These results may inform inpatient medication review by highlighting gabapentinoids, opioids, and anxiolytics/hypnotics as targets for risk-mitigating efforts, with an emphasis on individualized risk assessment.
Several limitations warrant consideration. The retrospective observational design limits causal inference. Propensity score matching was limited to age, sex, department of admission, and admission MFS score, focusing on baseline fall susceptibility rather than overall illness severity. Because medication exposure was defined at a single index time point and time-stamped administration data were unavailable, time-at-risk was not fully aligned and time-dependent analyses were not feasible, raising the possibility of time-related bias. In addition, gabapentinoids were analyzed as a single class for clinical interpretability; thus, gabapentin- vs. pregabalin-specific effects and dose-, duration-, or renal function–adjusted dose–response relationships could not be assessed. Although admission MFS captures components related to acute fall susceptibility (including mental status and prior falling), residual confounding due to unmeasured factors (e.g., frailty, delirium, and illness severity) may remain. LOS was handled in sensitivity analyses to address potential overadjustment and reverse causation; however, residual confounding cannot be fully excluded. Finally, this single-center study had limited power for subgroup analyses.
Our findings suggest an association between inpatient falls and CNS-acting/active medications. In conclusion, gabapentinoids should be considered along with other fall-risk–increasing drugs (e.g., opioids and anxiolytics/hypnotics) during an inpatient medication review. Prospective studies are needed to clarify the role of gabapentinoids, particularly with time-varying exposure, dose and renal function considerations.