Respiratory infections induced by postoperative bed rest, such as pneumonia and empyema, could lead to mortality of lung cancer patients [1], and the mortality rate of pneumonia is high regardless of the fact that the postoperative mortality of lung resection is reduced [2].
Risk factors such as age, smoking history, weak respiratory functions, invasive operations, intraoperative complications, or pathologic stages are known to be more likely to lead to postoperative respiratory infections [1, 3–5], or even prolonged postoperative hospital stay [6].
Moxa, a traditional Chinese herb which is minced before being burned in moxibustion, has been found to affect neuroendocrine immune functions and induce the production of heat shock proteins to activate the process of pain reduction and self-healing [7, 8]. Moreover, several reports have documented that moxibustion could influence gene expression, thus being beneficial to autoimmune and inflammatory diseases such as ulcerative colitis [9, 10], Crohn’s disease [11], arthritis [12] and intestinal mucositis [13]. Also, a previous study on recurrent respiratory tract infection in children suffering from cerebral palsy also indicated that moxibustion could exhibit a long-term clinical effect [14]. Moreover, moxibustion at the acupoint of ST36 (Zusanli) was proved to activate the protective responses against HSV-1 infection in male BALB/c mice by activating production of cytokines including interleukin and TNF-α [15].
Therefore, the aim of this study is to further investigate the potential role of moxibustion treatment in the management of respiratory infection in bed-ridden patients who are suffering from osteoporotic fracture of the spine.
Methods
The human study was approved by the institutional ethics committee and all experiments were performed in strict accordance with the 2013 version of the Declaration of Helsinki. Written informed consent was obtained from all participants or their first-degree relatives before the initiation of the human study. We recruited a total of 96 senile patients with pulmonary infection who were bed-ridden due to osteoporotic fracture of the spine in this study. The patients were randomly assigned to a MOX (–) group and a MOX (+) group in which the patients received moxibustion treatment at the points of BL13 (Feishu), RN8 (Shenque) and RN4 (Guanyuan). For the MOX treatment performed once each day for 12 weeks, 1.8 g of moxa cones (Hanyi, Nanyang, Henan, CN) were placed burning on a 3.3 cm × 0.7 cm herbal cake (Hanyi, Nanyang, Henan, CN).
The animal study was approved by the institutional animal ethics committee and all experiments were performed in strict accordance with the 8th edition of the Guide for the Care and Use of Laboratory Animals by the National Research Council (US) Committee. 32 specific-pathogen-free (SPF) male C57BL/6 mice 6–8 weeks of age were randomly divided into 4 groups: a SHAM group a PRIMED group, a MOX 15’ group and a MOX 30’ group. The mice were established as acute infected animals by i.p. injection of 10 LD50 Staphylococcus aureus after 2 days of i.p. injection of 6% starch broth (Sigma-Aldrich, St. Louis, MI, US). At 48 h after the moxibustion treatment, peripheral blood was collected from all mice, followed by euthanasia by i.p. injection of 100 mg/kg sodium pentobarbital. The peritoneal fluids were immediately collected for subsequent bacterial colony formation analysis to evaluate the bacterial clearance, i.e., the number of Staphylococcus aureus colonies as colony-forming units (CFU).
Peripheral blood samples collected from patients and animal models were prepared to study the levels of C-reactive protein (CRP), myeloperoxidase (MPO), interleukin (IL) 18 (IL-18), IL-1β, IL-10, IL-6 and tumor necrosis factor-α (TNF-α) using the appropriate assay kits following the instructions provided by the manufacturer.
A p-value no more than 0.05 was deemed as the level of statistical significance. All statistical analyses were carried out with GraphPad Prism software (GraphPad, Santa Barbara, CA, US).
Results
Human study
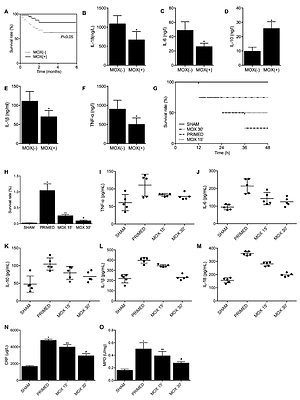
As indicated in Table I, demographic and baseline data of all bed-ridden senile patients were collected and compared in the table, and no significant differences were found in respect to the parameters. However, the survival curve up to 6 months showed that patients who received moxibustion treatment had a higher survival rate compared with patients in the MOX (–) group (Figure 1 A). Moreover, the MOX (+) group had lower levels of TNF-α (Figure 1 B), IL-6 (Figure 1 C), IL-1β (Figure 1 E) and IL-18 (Figure 1 F) compared with the MOX (–) group, and the level of IL-10 (Figure 1 D) was higher in the MOX (+) group, indicating the potential association between MOX treatment and reduced inflammation responses in the patients.
Table I
Basic characteristics of the recruited bed-ridden senile patients
Figure 1
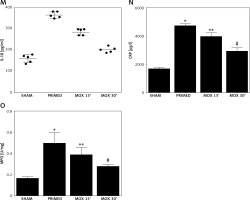
A – The survival rate of the MOX (+) patient group was higher than that of the MOX (+) patient group. B – The level of TNF-α was lower in the MOX (+) patient group than that in the MOX (–) patient group. C – The production of IL-6 was suppressed in the MOX (+) patient group compared with that in the MOX (–) patient group. D – Compared with the MOX (–) patient group, the IL-10 level was higher in the MOX (+) patient group. E – Compared with the MOX (–) patient group, the IL-1β level in the MOX (+) patient group was significantly lower. F – Compared with the MOX (–) patient group, the IL-18 level in the MOX (+) patient group was evidently reduced. G – Survival curves of difference groups of mice. H – The bacteria left in different groups of mice. I – ELISA analysis of TNF-α production in different groups of mice. J – ELISA analysis of IL-6 production in different groups of mice. K – ELISA analysis of IL-10 level in different groups of mice. L – ELISA analysis of IL-1β level in different groups of mice. M – ELISA analysis of IL-18 level in different groups of mice. N – ELISA analysis of CPR production in different groups of mice. O – ELISA analysis of MPO production in different groups of mice


Animal study
The survival analysis showed the lowest survival rate in the PRIMED group, while both moxibustion treatments for 15 min or 30 min elevated the suppressed survival rate (Figure 1 G). Meanwhile, moxibustion was found to substantially reduce the high bacteria count in the infected mice (Figure 1 H). Moreover, the production of TNF-α (Figure 1 I), IL-6 (Figure 1 J), IL-10 (Figure 1 K), IL-1β (Figure 1 L) and IL-18 (Figure 1 M) in the PRIMED group was evidently higher compared with that of the SHAM group, whereas treatment with moxibustion restored the expression of these factors in a time-dependent manner. Also, ELISA assay of the serum level of CPR (Figure 1 N) and MPO (Figure 1 O) also presented evidently promoted CRP and MPO production in the PRIMED group, while moxibustion significantly reduced the CRP and MPO production in the MOX 15’ group and MOX 30’ group. Altogether, the above observations validated the time-dependent inhibitory effect of moxibustion treatment upon inflammatory responses in the bacteria-infected mice.
Discussion
The over-activation of inflammasome NOD-like receptor protein 3 (NLRP3) induces the maturation and release of proinflammatory cytokines IL-1β and IL-18 [16]. Since IL-1β participates in the process of inflammatory injuries and damaging the colonic barrier, the modulation of NLRP3 could inhibit inflammatory responses and reconstruct colonic mucosal immune homeostasis [17]. Meanwhile, NLRP3 is also found to be regulated by the signaling pathway of nuclear factor κB (NF-κB), which participates in the inflammatory responses of inflammatory bowel disease (IBD) [18]. The high concentration of extracellular adenosine triphosphate (ATP) produced at inflammatory sites could activate the P2X7 receptor (P2X7R) [19], and P2X7R and ATP could collaboratively activate the pannexin-1 channel to promote transcription of NLRP3 mRNA [20]. According to Tourkochristou et al., upon the activation of NLRP3, apoptosis-associated speck-like protein (ASC) could trigger activation of caspase-1, which helps to produce mature IL-1β and IL-18 and induce chronic intestinal inflammation [21–23]. Also, it was found that the continuous interaction between the harmful gut bacteria and the pathogen-associated molecular patterns will accelerate activation of the NLRP3 inflammasome, which results in severer inflammatory reactions by the subsequent upregulation of the downstream pro-inflammatory cytokines such as TNF-α, IL-6, IL-1β and IL-18, thereby further aggravating the inflammatory reaction [24, 25]. Accordingly, compared with the MOX (–) patient group, we found that moxibustion in the MOX (+) patient group not only suppressed the expression of NLRP3, but also inhibited expression of pro-inflammatory factors such as TNF-α, IL-1β, IL-6 and IL-18 while increasing the level of IL-10. Similar results were obtained from the animal models primed with bacteria which subsequently received moxibustion treatment.
In this study, we not only studied the effect of moxibustion against lung infection, but also observed whether the moxibustion duration potentially influences the effect of moxibustion. Therefore, we treated the bacteria-infected mouse models for 15 min and 30 min, and we found that the 30-min treatment with moxibustion exerted a more beneficial effect against bacterial infection compared with the 15-min treatment with moxibustion, while both the 15-min moxibustion treatment and 30-min moxibustion treatment exhibited a significant beneficial effect compared with the untreated mice.
In conclusion, by observing the lung-infected patients and mouse models, we found that the MOX treatment could significantly alleviate the inflammatory responses, thus leading to a better survival rate of patients bed-ridden due to osteoporotic fracture of the spine.