Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
LIPID DISORDERS / CLINICAL RESEARCH
Postmarketing nutrivigilance safety profile: a line
of dietary food supplements containing red yeast rice
for dyslipidemia
1
Polish Mother’s Memorial Hospital Research Institute (PMMHRI), Lodz, Poland
2
Department of Hypertension, Medical University of Lodz (MUL), Lodz, Poland
3
Cardiovascular Research Centre, University of Zielona Gora, Zielona Gora, Poland
4
First Department of Internal Medicine, Diabetes Center, Division of Endocrinology
and Metabolism, Medical School, Aristotle University of Thessaloniki,
AHEPA Hospital, Thessaloniki, Greece
5
Institute of Cardiology and Regenerative Medicine, Faculty of Medicine, University
of Latvia, Riga, Latvia
6
Pauls Stradins Clinical University Hospital, Riga, Latvia
7
Department of Health Promotion Sciences Maternal and Infantile Care, Internal
Medicine and Medical Specialties (PROMISE), University of Palermo, Palermo, Italy
8
2nd Department of Cardiology of the East Slovak Institute of Cardiovascular Disease
and Faculty of Medicine PJ Safarik University, Kosice, Slovak Republic
9
School of Pharmacy and Biomolecular Sciences, Liverpool John Moores University,
Liverpool, UK
10
Liverpool Centre for Cardiovascular Science, Liverpool, UK
11
Department of Internal Medicine, University Hospital Center Zagreb, Zagreb, Croatia
12
IRCCS Azienda Ospedaliero-Universitaria Di Bologna, Bologna, Italy
13
Alma Mater Studiorum University of Bologna, Bologna, Italy
Submission date: 2021-02-26
Acceptance date: 2021-02-26
Online publication date: 2021-03-04
Publication date: 2021-07-16
Corresponding author
Arch Med Sci 2021;17(4):856-863
KEYWORDS
TOPICS
ABSTRACT
Introduction:
In the absence of a European standardized postmarketing food supplement surveillance system (nutrivigilance), some member states and companies have developed their own approaches to monitoring potential adverse reactions to secure a high level of product safety. This paper describes the use of a nutrivigilance system in monitoring the incidence of spontaneously reported suspected adverse reactions associated with food supplements containing red yeast rice (RYR).
Material and methods:
We report the data from a widely used product marketed under the trademark Armolipid/Armolipid Plus. Postmarketing information was collected in a voluntary nutrivigilance system established by the manufacturing company (Meda Pharma SpA, a Viatris Company, Monza, Italy). From 1st October 2004 to 31st December 2019, this system captured cases of suspected adverse reactions spontaneously reported by consumers, healthcare professionals, health authorities, regardless of causality.
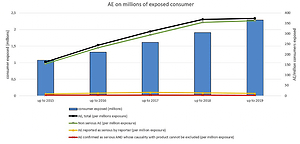
Results:
The total number of case reports received mentioning the RYR food supplement product line was 542, in which 855 adverse events (AEs) were reported. The total reporting rate of AEs was estimated to be 0.037% of 2,287,449 exposed consumers. Of the 542 cases, 21 (0.0009% of exposed consumers) included suspected serious adverse events (SAEs). After careful investigation, 6 cases (0.0003% of consumers exposed) and 6 AEs were assessed by the manufacturer as serious and potentially related to exposure to the above-mentioned RYR-based nutraceutical.
Conclusions:
This nutrivigilance-derived data analysis clearly demonstrates a low prevalence of suspected adverse events associated with the red yeast rice product line. Consumer safety of food supplements could be generally improved by raising awareness of the importance of following the indications and warnings detailed in a food supplement’s labeling.
In the absence of a European standardized postmarketing food supplement surveillance system (nutrivigilance), some member states and companies have developed their own approaches to monitoring potential adverse reactions to secure a high level of product safety. This paper describes the use of a nutrivigilance system in monitoring the incidence of spontaneously reported suspected adverse reactions associated with food supplements containing red yeast rice (RYR).
Material and methods:
We report the data from a widely used product marketed under the trademark Armolipid/Armolipid Plus. Postmarketing information was collected in a voluntary nutrivigilance system established by the manufacturing company (Meda Pharma SpA, a Viatris Company, Monza, Italy). From 1st October 2004 to 31st December 2019, this system captured cases of suspected adverse reactions spontaneously reported by consumers, healthcare professionals, health authorities, regardless of causality.
Results:
The total number of case reports received mentioning the RYR food supplement product line was 542, in which 855 adverse events (AEs) were reported. The total reporting rate of AEs was estimated to be 0.037% of 2,287,449 exposed consumers. Of the 542 cases, 21 (0.0009% of exposed consumers) included suspected serious adverse events (SAEs). After careful investigation, 6 cases (0.0003% of consumers exposed) and 6 AEs were assessed by the manufacturer as serious and potentially related to exposure to the above-mentioned RYR-based nutraceutical.
Conclusions:
This nutrivigilance-derived data analysis clearly demonstrates a low prevalence of suspected adverse events associated with the red yeast rice product line. Consumer safety of food supplements could be generally improved by raising awareness of the importance of following the indications and warnings detailed in a food supplement’s labeling.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.