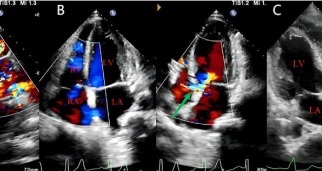
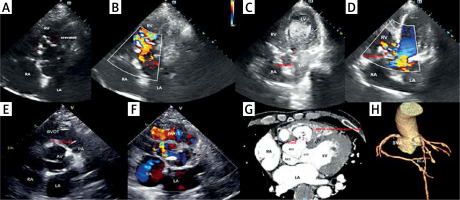
A 55-year-old man, with prior history of syphilis, was admitted to the hospital due to chest tightness with shortness of breath. Cardiac auscultation revealed a coarse continuous murmur of grade III or higher in the aortic valve auscultation area. Laboratory tests showed elevated total antibody to syphilis, and negative serotonin for syphilis. Electrocardiogram demonstrated complete right bundle branch conduction block and secondary ST segment alterations. Echocardiography (Figures 1 A, B) displayed four perforations of the right sinus of Valsalva (SoV) aneurysm (maximum diameter 4–5 mm) that had ruptured into the right ventricle (RV). There was a sac-like structure that had undergone calcification between the right aortic sinus and the RV with a large shunt flow from the right coronary sinus to the aneurysm and to the RV. Furthermore, it also showed calcification of the aortic valve with mild-to-moderate regurgitation and ascending aorta wall thickening, and 54% ejection fraction. The above finding was further confirmed by cardiac computed tomography angiography (CTA) (Figures 1 G–H). Due to the poor physical condition of the patient and the high risk of surgery, the family requested catheterization for the intervention. Subsequently, the patient underwent percutaneous closure of the SoV aneurysm rupture, which showed three parallel ruptures (maximum diameter 6.0 mm), no significant ventricular septal defect (VSD) shunt, and severe pulmonary hypertension (109/40/69 mm Hg) which was measured by the catheter without significant stenosis of the RV outflow tract. The position of the occluder was confirmed by SonoVue contrast-enhanced ultrasound with favorable results, only with a trivial residual shunt (Figures 1 C–F). Then, the pulmonary artery pressure that was reviewed again with the catheter was 95/43/62 mm Hg, which decreased significantly, and the symptoms of shortness of breath improved. At the time of discharge, there were no shortness of breath features and the patient showed improvement in motor symptoms with the medications for heart failure and pulmonary artery pressure.
Figure 1
Echocardiographic images and cardiac computed tomography angiography of the patient with the right sinus of Valsalva aneurysm. A, B – Echocardiographic images before transcatheter closure. C – SonoVue contrast-enhanced ultrasound during transcatheter closure. D – Echocardiography image during transcatheter closure. E, F – Echocardiography image after transcatheter closure. G, H – cardiac computed tomography angiography of the patient
LA – left atrium, LV – left ventricle, RA – right atrium, RV – right ventricle, RVOT – right ventricular outflow tract, SVA – sinus of Valsalva aneurysm, LCC – left coronary cusp, RCC – right coronary cusp, NCC – noncoronary cusp. Red star is the occluder.
Six months after discharge, the patient did not have chest tightness or shortness of breath. However, the patient was hospitalized in the gastroenterology department for recurrent melena and hematochezia, which was considered to be caused by vascular malformation in the small intestine leading to vascular rupture.
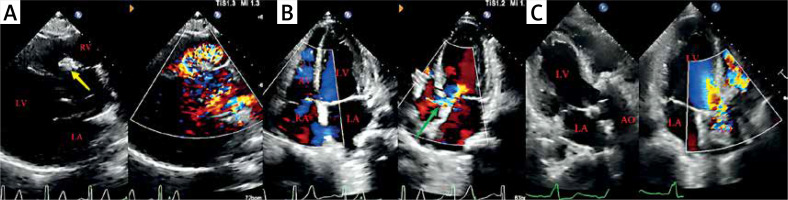
On follow-up after half a year, he was readmitted for shortness of breath and chest tightness. At the apex and the left sternal border a pathological holosystolic murmur was heard. Re-examination of the laboratory results showed elevated total antibodies to syphilis and positive serotonin with a titer of 1 : 1. Echocardiography (Figures 2 A, B) showed whole heart enlargement and right ventricular wall thickening. The right SoV aneurysm expansion area was 30 × 24 mm with partial calcification of the aneurysm wall, on which hyperechogenicity of the occluder can be seen and protrusion into the right ventricular outflow tract, and a shunt signal of about 3.2 mm in width can be seen on the left side of the blocker, with a peak flow velocity of 3.88 m/s, and a peak pressure difference of 60 mm Hg. The perimembranous ventricular septum appears to be continuously interrupted with a width of about 4 mm, with a velocity of 4.97 m/s, and a peak pressure difference of 99 mm Hg. Aside from this, there was pulmonary artery dilatation, moderate-to-severe aortic regurgitation, and normal left ventricular systolic function. Surgical treatment should be provided to prevent the development of potentially life-threatening complications. Intraoperatively, a 2.5 × 2.5 cm septal defect was seen immediately below the right coronary annulus. Subsequently, aortic valve replacement, occluder removal, repair of ruptured aortic sinus aneurysm, repair of ventricular septal defect, aortic root angioplasty, and implantation of a temporary pacemaker were performed. Postoperative echocardiography (Figure 2 C) showed successful repair of the VSD and right coronary sinus aneurysm rupture, significant reduction of aortic regurgitation, and normalization of pulmonary artery pressure. Pathology of the aortic valve suggested fibrous tissue hyperplasia with collagenization and calcification, focal mucinous degeneration, and a small amount of lymphocytic infiltration. Post-operatively, the patient was medicated with furosemide, sildenafil, macitentan tablets, and warfarin, recovering well, and was finally discharged 16 days after surgery. In addition, patients are released from the hospital to receive penicillin treatment in dermatology outpatient clinics.
Figure 2
Echocardiographic images of the patient before and after the second surgery. A, B – Echocardiographic images before the second surgery; the yellow arrow is the occluder; the green arrow is the ventricular septal defect. C – Echocardiographic images after surgical operation
LA – left atrium, LV – left ventricle, RA – right atrium, RV – right ventricle, AO – ascending aorta.
Aneurysm of the aortic sinus, also referred to as the sinus of Valsalva aneurysm (SVA), is a rare cardiac abnormality that is often combined with VSD and aortic regurgitation. It can be caused by congenital factors or acquired diseases secondary to syphilis, Behçet’s disease, infective endocarditis, aortic sclerosis, rheumatic heart disease, trauma, etc [1]. In comparison, congenital aneurysms are more common, accounting for 0.5% to 3.0% of congenital heart disease. Secondary SVAs are relatively uncommon, and those associated with syphilis infection are even rarer [2]. In this case, the patient also had a combined ventricular septal defect, but unfortunately, it was missed during the first hospitalization. We speculated that the following factors might be responsible for this missed diagnosis: (a) the SVA may partially or even completely obscure the VSD, in turn affecting our judgment; (b) the continuous Doppler systolic shunt signals of the VSD may be masked by the biphasic continuous shunt signals of the right SVA (RSVA), resulting in the missed diagnosis.
Syphilitic aortitis has been reported in 70–80% of untreated syphilis patients, and 10% can cause significant cardiovascular complications [3], mainly including aortic sinus aneurysm, aortic valve insufficiency, and coronary artery stenosis [4]. The pathology of syphilitic aortic aneurysm is characterized by the absence of elastic fibers and muscle tissue, the enrichment of plasma cells and lymphocytes, and the proliferation of fibrous tissue in the arterial wall. The lesions are mainly located in the thoracic aorta and rarely in the abdominal aorta [5]. Patients with syphilitic aortic aneurysms can be asymptomatic for many years with no obvious clinical manifestations, but the mortality rate is high. In severe cases, rupture of the aneurysm can lead to cardiac compression and sudden death, which are the main causes of late death, and aggressive surgery is a more effective treatment. Both direct inflammation of the valve and dilatation of the ascending aorta can cause aortic regurgitation (AR), and severe AR can lead to heart failure and thus require valve replacement [6]. Aortic sinus aneurysm rupture or AR is one of the leading causes of death from cardiovascular syphilis; consequently, patients with syphilis should be alert to cardiovascular illness.