Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory autoimmune disease characterised by inflammation of the synovial membrane in joints, causing joint destruction and consequently pain and functional disability, diminishing quality of life among patients [1–5]. RA affects approximately 0.5% of the adult population globally [6, 7]. The disease is predominantly treated using disease-modifying anti-rheumatic drugs (DMARDS) and nonsteroidal anti-inflammatory drugs (NSAIDs) [8, 9]. These drugs act by reducing inflammation and delaying bone erosion, and, therefore, they slow down the progression of the disease [10]. Methotrexate (MTX) is the first-line treatment for this disease both as monotherapy and as an adjuvant to biological therapies, due to its high efficacy and rapid action [11, 12]. However, there is considerable interindividual variability in MTX toxicity [13]. Previous studies have found that one-third of patients on MTX treatment do not reach the therapeutic goal due to lack of response to the drug, development of toxicity or loss of efficacy over time [14–17]. Among the most frequent adverse effects are gastrointestinal disorders, liver toxicity, skin reactions, and bone marrow disorders [18, 19]. The factors responsible for this variable toxicity in MTX treatment are not yet fully understood, but recently the genetic polymorphisms linked to the MTX metabolic pathway have become the targets of study for understanding MTX toxicity [20–24].
MTX is a folic acid analogue with anti-inflammatory and anti-proliferative properties [25, 26]. It acts by inhibiting various enzymes in the synthesis pathway of folate, methionine, adenosine, and nucleotides [25], significant among which is methylenetetrahydrofolate reductase (MTHFR). The protein encoded by this gene catalyses the conversion of 5,10-methylenetetrahydrofolate to 5-methyltetrahydrofolate, a co-substrate for homocysteine remethylation to methionine [27]. Two polymorphisms in this enzyme play a part in metabolising MTX. SNP (Single Nucleotide Polymorphism) MTHFR 677C> T (rs1801133) leads to the substitution of alanine for valine at position 233 (Ala233Val) of the protein, reducing the activity of the enzyme. Previous studies have found a link between TT+CT genotypes and MTX liver toxicity, due to increased serum levels of liver enzymes and homocysteine [28, 29]. Another of the polymorphisms studied is MTHFR 1298A> C (rs1801131), which leads to the substitution of glutamine for alanine at codon 429 (Glu429Ala). The G allele has previously been linked to increased risk of adverse effects such as neurotoxicity and liver toxicity [30, 31]. The methylenetetrahydrofolate dehydrogenase 1 (MTHFD1) encodes a protein that possesses three distinct enzymatic activities: 5,10-methylenetetrahydrofolate dehydrogenase, 5,10-methenyltetrahydrofolate cyclohydrolase, and 10-formyltetrahydrofolate synthetase. Each of these activities catalyses one of three sequential reactions in the interconversion of 1-carbon derivatives of tetrahydrofolate, which are substrates for methionine, thymidylate, and de novo purine syntheses. The polymorphism 1958G>A (rs2236225) leads to the substitution of arginine for glycine at gene position 653 (Arg653Gln), reducing the enzymatic activity of the complex [32]. Another enzyme found on the folic acid metabolic pathway is 5-methyltetrahydrofolate-homocysteine methyltransferase (MTR), also known as cobalamin-dependent methionine synthase, which catalyses the final step in methionine biosynthesis [28]. An SNP in MTR gene (2756A>G rs1805087) has been found to reduce enzyme activity [33, 34].
MTX is transported across the cell membrane by a series of transporter proteins, significant amongst which are ABCB1 (P-glycoprotein) and multidrug resistance-associated protein 2 (ABCC2). The membrane-associated proteins encoded by these genes are members of the superfamily of ATP-binding cassette (ABC) transporters. ABC proteins transport various molecules across extra- and intra-cellular membranes [35]. The ABCB1 3435C>T (rs1045642) gene polymorphism has been found to affect response to the drug; specifically, genotype CC could play a part in reducing response to therapy [36]. Similarly, the CT genotype has been linked to increased risk of gastrointestinal toxicity in 336 RA patients of Indian origin [37]. Another of the polymorphisms involved is (rs4148396) in the ABCC2 gene, a transporter protein involved in eliminating metabolites and located on the surface of hepatocytes, the intestine, and the proximal tubules [38]. In a study on 309 Caucasian RA patients on MTX treatment, the CC genotype was found to be associated with increased risk of alopecia [39]. Finally, an SNP in TLR4 (Toll-4 receptor) (896A>G; rs4986790) gene, which codified a protein member of the Toll-like receptor (TLR) family and plays a fundamental role in pathogen recognition and activation of innate immunity, has been linked to the development of mucositis in patients being treated with folic acid and MTX [40].
This was the conceptual framework for our study evaluating the influence of polymorphisms in the genes along the MTX metabolic pathway, including MTHFR 677C>T (rs1801133), 1298A>C (rs1801131), MTHFD1 1958G>A (rs2236225), MTR 2756A>G (rs1805087), ABCB1 3435C>T (rs1045642), ABCC2 C>T (rs4148396), and TLR4 896A>G (rs4986790), on specific types of toxicity of MTX treatment that have not been previously described among Caucasian RA patients.
Material and methods
Study design
An observational, retrospective cohort study conducted in the Virgen de las Nieves University Hospital in Granada (HUVN) with the approval of the reference Ethics Committee in compliance with the Helsinki Declaration. Informed consent was obtained from all patients prior to their inclusion in the study, and their data were treated confidentially.
Target population
The study recruited 200 Caucasian patients, aged over 18 years, and diagnosed with RA as per American College of Rheumatology (ACR) criteria, who were on, or had been on, MTX treatment for at least 1 month. The initial dose was 15 mg/week orally, with escalation from 5 mg/month to 25–30 mg/week or to the maximum tolerable dose. In the case of an insufficient response it was changed to subcutaneous administration [41, 42].
Study location and timeframe
Patients diagnosed with RA and on MTX treatment between February 1990 and June 2018, in HUVN Rheumatology and Pharmacy Departments.
Sociodemographic and clinical variables
The data collected were: sex, age when diagnosed with RA, duration of MTX treatment, smoking status, baseline rheumatoid factor (RF) value, baseline DAS28 [43] value, and adverse reactions relating to blood disorders (anaemia, neutropaenia, macrocytosis, and thrombocytopaenia), smelling (anosmia), digestive system (diarrhoea, nausea and vomiting, mucositis), liver failure, nervous system (dizziness and headaches), psychiatric disorders (anxiety), reproductive system (metrorrhagia), respiratory disorders (pulmonary fibrosis), and skin and subcutaneous tissue disorders (alopecia and acneiform rash).
EULAR response was according to the guidelines given by the European League Against Rheumatism, and was classified as satisfactory (presenting DAS28 < 3.2 and DAS28 improvement > 1.2) or unsatisfactory (presenting DAS28 ≥ 3.2 and DAS28 improvement ≥ 1.2).
Remission and low disease activity were considered when patients achieved DAS28 < 2.4 and DAS28 < 3.6, respectively.
Genetic variables
DNA isolation
Saliva samples were collected in 50 ml BD Falcon™ conical tubes (BD, Plymouth, UK). DNA isolation was performed using the QIAamp DNA Mini Kit (QiagenGmBH, Hilden, Germany) according to the manufacturer’s instructions for DNA purification from blood or saliva and stored at –40°C.
Detection of genetic polymorphisms
The genetic polymorphisms identified as being involved in MTX toxicity – ABCB1 3435C>T (rs1045642), ABCC2 C>T (rs4148396), MTHFR 677C>T (rs1801133), MTHFR 1298A>C (rs1801131), MTR 2756A>G (rs1805087), MTHFD1 1958G>A (rs2236225) and TLR4 896A>G (rs4986790) – were analysed using real-time PCR with TaqMan® probes. The assay ID used for gen ABCB1 (rs1045642) is C_7586657_20, for ABCC2 (rs4148396) is C_11305432_10, for MTHFR (rs1801133) is C_1202883_20, for MTHFR (rs1801131) is C_850486_20, for MTR (rs1805087) is C_12005959_10, for MTHFD1 (rs2236225) is C_1376137_10, and for TLR4 (rs4986790) is C_11722238_20.
Toxicity variables
Toxicity was evaluated using version 4.0 of Common Terminology Criteria for Adverse Events (CTCAE). According to the severity of the adverse reaction, it was graded 0–2 and 3–4. This study considered adverse events of grade 3–4, dichotomised into the presence or absence of type of adverse reaction.
Statistical analysis
Quantitative data were expressed as mean (± standard deviation) for normal-distribution variables or medians and percentiles (25 and 75) for non-normal-distribution variables.
Bivariate analysis was used to evaluate the link between toxicity and polymorphisms using Fisher’s exact test and odds ratio (OR) with the appropriate 95% confidence interval (CI).
Differences in distribution of genotypes for toxicity were analysed using 3 × 2 tables for each genotype. Values of p < 0.05 were considered statistically significant. Strength of association between RA patients and genotypes was estimated using relative risk (RR) with 95% CI.
Descriptive analysis was performed using R 3.0.1 [44].
The Hardy-Weinberg equilibrium and pairwise haplotype frequencies were estimated using the free, open-source whole genome association analysis toolset PLINK [45].
Results
Patient characteristics
The baseline characteristics of the 200 RA patients are shown in Table I. Median age was 45 years (35, 53), 161 were female (161/200; 80.5%), and 143 were non-smokers (143/200; 71.5%). The median of the duration on MTX treatment was 46 months (17, 88). As regards the baseline RF value, it was positive for 68.3% (63/199) of patients, and for RA activity analysed by DAS28 value the mean obtained was 5.42 ±1.15. The grade 3–4 toxicity profile value was: 6.0% (12/200) anaemia, 7.5% (15/200) neutropaenia, 2.5% (5/200) macrocytosis, 1.5% (3/200) thrombocytopenia, 1% (2/200) anosmia, 16.0% (32/200) diarrhoea, 23.0% (46/200) nausea and vomiting, 11.0% (22/200) mucositis, 46.4% (90/200) liver failure, 9.0% (18/194) dizziness, 10.0% (20/200) headache, 0.5% (1/200) anxiety, 1.5% (3/200) metrorrhagia, 3.5% (7/200) pulmonary fibrosis, 12.5% (25/200) alopecia, and 4.5% (9/200) acneiform rash (Table I).
Table I
Clinical-pathological characteristics of the 200 RA patients treated with methotrexate
Influence of clinical-pathological characteristics on toxicity
Nausea and vomiting showed a statistically significant association with treatment duration (p = 0.0423; OR = 2.13; 95% CI: 1.03–4.54) (Supplementary Table SI). No association was observed between the other toxicities and patient clinical-pathological characteristics (Supplementary Tables SII–SXVI).
Genotyping distribution
The distribution of all the polymorphisms studied match what is expected as per the Hardy-Weinberg equilibrium model (Supplementary Table SXVII). The linkage disequilibrium D´ and r2 values are shown in Supplementary Table SXVIII.
Influence of polymorphisms on toxicity
Haematological toxicity
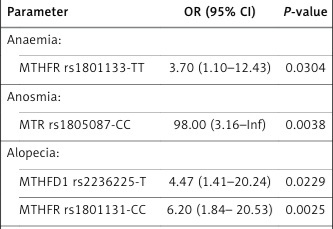
Bivariate analysis showed that patients with CC genotype for ABCC2 rs4148396 polymorphism were at greater risk of developing macrocytosis (p = 0.0022) (Supplementary Table SXIX). The multivariate logistic regression model revealed the influence of this polymorphism on macrocytosis (Table II). In the bivariate analysis, MTHFR rs1801133 was associated with anaemia. In particular, patients with the TT genotype were at greater risk of anaemia (p = 0.0328; OR = 3.70; 95% CI: 13–12.09) (Supplementary Table SXX). This association was also clear in the multivariate logistic regression analysis (Table II). Other haematological toxicities (neutropaenia and thrombocytopaenia) showed no significant association with the polymorphisms analysed (Supplementary Tables SXXI, SXXII).
Table II
The influence of gene polymorphisms on toxicity
Nervous system toxicity
Bivariate analysis showed that the MTHFR rs1801133 and ABCC2 rs4148396 gene polymorphisms were associated with a higher risk of dizziness and headaches, respectively. Specifically, patients with MTHFR rs1801133-T allele were at greater risk of experiencing dizziness (p = 0.0355; OR = 8.15; 95% CI: 1.06–62.75) (Supplementary Table SXXIII). Similarly, patients with ABCC2 rs4148396-TT genotype were at greater risk of experiencing headaches (p = 0.0496; OR = 2.67; 95%CI: 0.01–7.01) (Supplementary Table SXXIV). Multivariate logistic regression analysis was conducted to evaluate the impact of these polymorphisms on nervous system toxicity, and both were found to be statistically significant (Table II).
Other toxicity subtypes
Bivariate analysis showed that patients with MTHFR rs1801131-C allele were at greater risk of developing mucositis and acneiform rash (p = 0.02765; OR = 3.02; 95% CI: 1.20–7.57 and p = 0.0307; OR = 5.74; 95% CI: 1.16–28.27, respectively) (Supplementary Tables SXXV, SXXVI). The multivariate logistic regression model showed that MTHFR rs1801131 was the only independent factor associated with developing mucositis and acneiform rash (Table II). In the bivariate analysis, patients with MTR rs1805087-CC genotype were at greater risk of developing anosmia (p = 0.0298; RR = 98.00; 95% CI: 4.41–2180.05) (Supplementary Table SXXVII), which was confirmed in the multivariate analysis (Table II). In the bivariate analysis, patients with MTHFD1 rs2236225-T allele and MTHFR rs1801131-CC genotype were at greater risk of developing alopecia (p = 0.0248; OR = 3.83; 95% CI: 1.10–13.30 and p = 0.0072; OR = 5.11; 95% CI: 1.37–17.70, respectively) (Supplementary Table SXXVIII), which was statistically significant in the multivariate analysis (Table II). Liver failure was associated with MTHFD1 rs2236225 gene polymorphism. Patients with MTHFD1 rs2236225-TT genotype showed higher toxicity due to liver failure (p = 0.0332; OR = 2.34; 95% CI: 1.12–4.86) (Supplementary Table SXXIX). The multivariate logistic regression analysis showed that the MTHFD1 rs2236225 gene polymorphism was the only independent factor associated with liver failure (Table II).
For the remaining polymorphisms, no significant associations were found between the genotype and appearance of toxicity (Supplementary Tables SXXX–SXXXIV).
Discussion
Over 2 million patients experience adverse reactions to medication annually, with around 197,000 deaths as a result [46]. In the case of RA, MTX is the first-line treatment, but 30% of RA patients on MTX therapy cease treatment due to side effects such as liver failure or gastrointestinal disorders. Furthermore, there is considerable variability between patients as regards developing toxicity, which limits treatment [47–50]. Among the causes leading to this variability are polymorphisms in the genes in the MTX metabolic pathway [51–59].
This study included 200 RA patients in a single institution, who had been on MTX treatment for at least one month, with a view to investigating the influence of polymorphisms in ABCB1, ABCC2, MTHFR, MTR, MTHFD1, and TLR4 genes on the risk of developing drug-related toxicity. Among these patients, MTHFR rs1801133-C allele and TT genotype, MTHFR rs1801131-C allele and CC genotype, MTR rs1805087-CC genotype, MTHFD1 rs2236225-TT genotype and T allele, and ABCC2 rs4148396-TT genotype are all predictors of at least one grade 3-4 adverse reaction developing (Table II).
The influence of polymorphisms in MTHFR (rs1801131 and rs1801133) on MTX toxicity has been extensively researched [31, 60, 61]. In our study, the polymorphism rs1801133 has been associated with the development of anaemia in patients with the TT genotype (p = 0.0304; OR = 3.70; 95% CI: 1.10–12.43) and of experiencing dizziness in patients with the C allele (p = 0.0438; OR = 8.15; 95% CI: 1.61–148.68). No previous studies have assessed the effect of this polymorphism on both toxicity subtypes in RA patients on MTX therapy. In line with our findings, a previous study in India on 236 RA patients treated with MTX found a link between the T allele for MTHFR rs1801133 and the development of general toxicity (RR = 2.01; 95% CI: 1.09–3.70), primarily with increased levels of liver enzymes (RR = 2.38; 95% CI: 1.06–5.34) [28]. Similarly, a study on Caucasian patients with osteosarcoma (63 patients) and acute lymphoblastic leukaemia (52 patients) on MTX therapy showed a link between the T allele and liver toxicity (p = 0.04; OR = 1.8; 95% CI: 1.0–3.2) [61]. In relation to MTHFR rs1801131, our study revealed a significant link between the C allele and the risk of developing acneiform rash (p = 0.0322; OR = 5.74; 95% CI: 1.34–39.21) and mucositis (p = 0.0188; OR = 3.02; 95% CI: 1.22–7.91). Patients carrying the CC genotype also showed greater risk of developing alopecia (p = 0.0072; OR = 5.11; 95% CI: 1.37–17.70). The link between this polymorphism and the development of dermatological toxicity has not been previously studied. However, in line with our findings, a study on 1004 Asian patients with lung cancer receiving platinum-based chemotherapy found a link between patients with the CT genotype and the development of gastrointestinal toxicity (p = 0.004, OR = 0.40; 95% CI: 0.22–0.75) [20].
In relation to MTHFD1 rs2236225 gene polymorphism, we found an association between the MTHFD1 rs2236225-T allele and the risk of alopecia (p = 0.0248; OR = 3.83; 95% CI: 1.10–13.30) as well as between the MTHFD1 rs2236225-TT genotype and risk of liver failure (p = 0.0229; OR = 2.34; 95% CI: 1.14–4.96). Liver failure is the most frequently occurring adverse reaction and therefore the one that causes the most concern for MTX treatment, and alopecia is another frequent side effect, but there are no previous studies that assess the effect of MTHFD1 rs2236225 polymorphism on liver toxicity or alopecia in RA patients. However, its effect in 41 patient from Slovenia with pleural mesothelioma has been assessed regarding response to pemetrexed treatment, which was found to be lower in patients with the T allele (p = 0.0005; OR = 0.12; 95% CI: 0.03–0.54) [62].
As regards ABCC2 rs4148396 polymorphism, our study showed a link between the TT genotype and greater risk of experiencing headaches (p = 0.0466; OR = 2.67; 95% CI: 0.98–6.94). The effect of this polymorphism on central nervous system toxicity has not been studied previously in RA patients, although its association with toxicity has been evaluated in a study on 154 Caucasian patients with colorectal cancer receiving chemotherapy (5-fluorouracil/leucovorin/oxaliplatin), which revealed a correlation between the T allele of ABCC2 rs4148396 and neurotoxicity (p = 0.0048; OR = 4.69; 95% CI: 1.60–13.74) [63].
Another of the MTX metabolic pathway polymorphisms analysed is MTR rs1805087. We found a link between the CC genotype of this polymorphism and anosmia (p = 0.0038; OR = 98.00; 95% CI: 3.16–infinite); however, the sample size was only 2 patients. There are no previous studies that evaluate the role of this polymorphism in developing anosmia, but its role as a biomarker for gastrointestinal disorders and liver toxicity has been evaluated in 405 European patients, and although no significant differences were found in this respect, they were found in the time taken for disease to become inactive (p = 0.042; OR = 1.64; 95% CI: 1.02–2.63) [64].
Lastly, other polymorphisms analysed for which we found no significant differences are ABCB1 rs1045642 and TLR4 rs4986790. In line with our findings, various studies have assessed the role of the ABCB1 rs1045642 polymorphism in patients receiving platinum-based chemotherapy. A study on 120 Caucasian patients with colorectal cancer on oxaliplatin therapy evaluated the effect of ABCB1 rs1045642 gene polymorphism on neurotoxicity, finding no significant differences (p = 0.951; OR = 0.97; 95% CI: 0.39–2.41) [65]. Another study on 141 patients with non-small-cell lung cancer receiving carboplatin/cisplatin treatment studied the effect of this polymorphism on different toxicity sub-types (asthenia, gastrointestinal toxicity, liver toxicity, infection, nephrotoxicity, and neurotoxicity) and found no significant association [66]. For TLR4 rs4986790 gene polymorphism, a study on 1481 RA patients found no significant association between this polymorphism and toxicity relating to cardiovascular risk, which is in line with our findings [67].
The patients included in our study were recruited from the same hospital, which ensures sample homogeneity as regards treatment and period of administration, and toxicity data. Its main limitation relates to sample size, due to the small number of patients showing a link between some of the polymorphisms described and toxicity.
This notwithstanding, and given that there are currently no tests available for predicting MTX toxicity, it may be concluded that pharmacogenetic studies of the polymorphisms involved in MTX metabolic pathways may be clinically useful for rheumatologists as an aid for making decisions regarding therapy, such as dose adjustment or concomitant administration of folic or folinic acid, or an alternative treatment. In this way, therapeutic algorithms for RA that may integrate clinical, genetic, and other biomarker data are critically important to individualise and optimise therapy with antirheumatic agents. In this regard, the next crucial step is the clinical verification of these promising biomarkers using studies that assess multiple SNPs, longer-term follow-up, and larger sample sizes. Therefore, further research is needed to clarify the true role of these polymorphisms in the toxicity to MTX therapy in AR patients.
Moreover, along with the growing pharmacogenetic knowledge in rheumatology, there is increasing difficulty to explain to physicians the use, advantages, and disadvantages of pharmacogenetics and how to interpret and apply genetic test result to guide drug therapy decisions. Thus, effective health education for physicians is required for a successful choice and/or adjustment of antirheumatic agents and for an understandable information towards the patients.
In conclusion, these results suggest that RA patients treated with MTX, who have MTHFR rs1801133-TT genotype, are at higher risk of developing anaemia. Patients with MTHFR rs1801133-C allele are at greater risk of experiencing dizziness, and patients with MTHFR rs1801131-C allele or CC genotype are at greater risk of developing mucositis, acneiform rash, and alopecia. Our results also revealed that patients with MTHFD1 rs2236225-T allele or TT genotype are at greater risk of liver failure and alopecia. Similarly, the TT genotype of ABCC2 rs4148396 gene polymorphism conferred higher risk of suffering headaches in our patients.