Delirium, an acute brain dysfunction, is one of the most prevalent complications occurring in intensive care unit (ICU) patients and those who undergo cardiac surgery. It is related to worse clinical outcomes including a longer ICU stay, longer duration of mechanical ventilation, increased risk of cognitive impairment and higher mortality rates [1–5]. Depression and cognitive impairment following critical illness are associated with delirium, which could contribute to several long-term negative consequences including reduced quality of life, increased healthcare costs and institutionalization [6–8]. A chronic brain syndrome is common in about 40% of delirium patients [9], while full resolution of symptoms of delirium was observed in only 4% of patients at discharge and reduction in functions remained even 6 months after hospital discharge [10, 11].
Although the exact pathophysiological mechanisms of delirium remain unclear, disruption of neurotransmission, inflammation or acute stress responses are considered as potential mechanisms involved in the development of delirium [12]. Early diagnosis and accurate treatment of each underlying cause prior to the intervention is recommended to lessen the occurrence of postoperative delirium [13]. It is suggested that inflammation might play a role in the development of delirium as previous research indicated that higher serum levels of C-reactive protein (CRP) are related to fewer delirium-free days [14]. In addition, cytokines such as tumor necrosis factor-α and interleukins 1 and 2 could interfere with neurotransmitter function. Thus, increase in cytokines might be associated with the development of delirium [14]. Acute inflammation is common in several conditions including sepsis, trauma and acute respiratory distress syndrome (ARDS), which might increase delirium incidence in patients in ICU [15–17]. It is proposed that neuroinflammation, oxidative damage and apoptosis followed by systemic inflammation, characterized by elevated CRP, could contribute to cerebral hypoperfusion and delirium [18, 19].
Delirium occurs in about 80% of ICU patients and contributes to hypoperfusion in frontal, temporal and subcortical brain regions, which all might lead to the progression of neuropsychological deficits [15, 16, 20]. Nevertheless, pharmacological agents capable of preventing or treating delirium during critical illness are lacking [18].
It has been recently suggested that statins might be safe and useful drugs to prevent and treat delirium in ICU patients due to their potential anti-inflammatory (peripheral and central) effects [18]. Statins are usually prescribed to reduce low-density lipoprotein cholesterol (LDL-C) levels and subsequently cardiovascular morbidity and mortality [21]. However, it is revealed that statins have lipid-independent pleiotropic effects [22–27] and attenuate neuro-inflammation, which might have beneficial effects on central neural system injury resulting in the improvement of delirium and its associated long-term cognitive injury [18]. In a previous trial, it was shown that simvastatin administration significantly reduced CRP in healthy volunteers and in critically ill patients with acute lung injury [28]. In a previous prospective cohort, a significant correlation was found between statin therapy and lower daily risk of delirium in critically ill patients [19]. Similarly, statin therapy leads to reduced cardiac surgery-induced delirium [17].
Due to the strong impact of delirium on clinical outcomes of critical ill patients, finding an efficient treatment without undesirable side effects is necessary. However, the effect of statins as safe drugs for the attenuation of delirium in ICU patients has been poorly investigated, with no report on the efficacy of atorvastatin. Therefore, the aim of the current study was to assess the efficacy of atorvastatin administration on delirium status of patients in the ICU.
Methods
This was a double-blind randomized controlled trial designed to determine the effect of atorvastatin on delirium status in patients referred to the general intensive care unit (GICU) of the Shahid Sadoughi teaching Hospital, Yazd, Iran, from June to December of 2015.
Study subjects
In this study, GICU patients were assessed for delirium status and if they had at least 2 days delirium, they were randomly assigned to two groups to receive either atorvastatin 40 mg/day (intervention group) or placebo (control group) without any statin therapy. The study researchers, nurses and the other hospital staff were blinded regarding the assigned intervention. The inclusion criteria were patients with age ≥ 18 years who were admitted to the GICU and had delirium for at least 2 days with a Glasgow Coma Scale (GCS) score between 13 and 15. Exclusion criteria were suffering from any known psychological disorder, hepatic or renal dysfunctions, abnormal levels of hepatic enzymes and treatment with macrolide drugs. Patients who had a history of hypersensitivity to atorvastatin were also excluded.
Outcome measures
A well-trained nurse assessed the levels of sedation twice a day at 10:00 a.m. and 18:00 p.m. using the Richmond Agitation-Sedation Scale (RASS) [29]. “Delirium free” status was defined as a day without delirium [30]. For ICU patients, normal cognitive status (i.e., awakeness and no delirium) was defined if in a day there was no delirium or the patient was not in a coma (any cause including sedation) [30].
According to RASS [31], scores 0–4 were indicative of alert patients or patients who agitated prior to stimulation. Scores –1 to –3 indicated unalert patients (not spontaneously alert) based on the duration of eye contact, when patients were called by name to look at the rater. Physical stimulation (i.e. shoulder shake and/or sternal rub) was used for patients who did not respond to verbal stimulation and according to their response received a score of –4 or –5. For calm patients who were not alert prior to verbal and physical stimulation, scores –1 to –5 were considered even if they became agitated on stimulation [31].
The Acute Physiology and Chronic Health Evaluation score (APACHE II) was used considering 12 routine physiological measurements, age and previous health status to provide a general measure of severity of disease with a range of 0 to 71. Increasing scores indicated the subsequent risk of hospital death [32, 33].
Sociodemographic factors and clinical status of patients including age, sex, hypercholesterolemia, ischemic heart disease, diabetes, and admission after aortic aneurysm surgery and ventilated days were also collected.
All experimental procedures were approved by the Ethics Committee of the Shahid Sadoughi University of Medical Sciences, Yazd, Iran. The participant’s autonomy and anonymity as well as confidentiality were considered as ethical issues. A family member of the patient was asked to fill in a written consent form before participation of the patient in the study. The whole protocol was registered in the Iranian Registry of Clinical Trials under the accession code of IRCT201606131836N9.
Statistical analysis
All data were analyzed using SPSS, version 16, at a significance level of < 0.05. The Kolmogorov-Smirnov test was applied to assess normality of the data. Independent and pair t-test were used to analyze differences between and within groups, respectively. The χ2 test was used to analyze differences between groups in categorical variables. Wilcoxon and Mann-Whitney tests were used to analyze non-parametric factors between and within groups, respectively. Logistic regression analysis was used to evaluate the correlation between atorvastatin therapy and delirium status.
Results
General characteristics
A total of 98 patients were enrolled in this study, among whom 2 subjects died and 6 patients were discharged 1 day after entering the study and finally data were analyzed for 90 patients. Forty patients (20 women) and 50 patients (35 women) were included in the intervention and control groups, respectively. As shown in Table I, there were no significant differences between the mean age, sex, the prevalence of diabetes mellitus, surgery and consumption of corticosteroids and nonsteroidal anti-inflammatory drugs (NSAIDs) between the two groups. However, the prevalence of ischemic heart disease and cerebrovascular disease and hypercholesterolemia was significantly higher among case patients than controls (Table I).
Table I
Clinical characteristics of study population, stratified by atorvastatin use
| Variables | Atorvastatin use (40 mg/day) | P-value | |
|---|---|---|---|
| No (50) | Yes (40) | ||
| Age | 65 ±23.87 | 51 ±19.33 | 0.14* |
| Female (%) | 35 (70%) | 20 (50%) | 0.65** |
| IHD or CVD | 10 (20%) | 20 (50%) | < 0.004* |
| Diabetes mellitus | 10 (20%) | 4 (10%) | 0.15* |
| Hypercholesterolemia | 0 (0%) | 8 (20%) | < 0.001* |
| Surgery | 20 (40%) | 12 (30%) | 0.50* |
| NSAIDs | 35 (60%) | 24 (80%) | 0.60* |
Effect of atorvastatin administration on delirium status and clinical outcomes
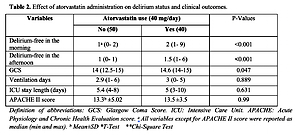
Median delirium-free status in both morning and afternoon was significantly higher in patients receiving atorvastatin compared with the control group (p < 0.001) (Table II). In comparison with the patients without atorvastatin consumption, GCS was significantly higher in atorvastatin-receiving participants (p = 0.047) (Table II). No significant difference was observed between groups regarding ventilation duration, length of stay and APACHE II score.
Table II
Effect of atorvastatin administration on delirium status and clinical outcomes
| Variables | Atorvastatin use (40 mg/day) | P-values | |
|---|---|---|---|
| No (50) | Yes (40) | ||
| Delirium-free in the morning | 1 (0–2)a | 2 (1–9) | < 0.001 |
| Delirium-free in the afternoon | 1 (0–1) | 1.5 (1–6) | > 0.001 |
| GCS | 14 (12.5–15) | 14.6 (14–15) | 0.047 |
| Ventilation days | 2.9 (1–6) | 3 (0–5) | 0.889 |
| ICU stay length [days] | 5.4 (4–8) | 5 (3–10) | 0.631 |
| APACHE II score | 13.3 ±5.02b | 13.5 ±3.5 | 0.99 |
As shown in Table III atorvastatin consumption significantly reduced RASS in morning and afternoon compared with baseline (p < 0.05). Further analysis using linear regression indicated that there were positive correlations between atorvastatin administration and delirium-free state in the morning (β = 2.67, 95% CI: 1.4–5.0; p = 0.029) and afternoon (β = 3.00, 95% CI: 1.5–6.1; p = 0.012).
Table III
Effect of atorvastatin administration on Richmond Agitation-Sedation Scale (RASS) and C-reactive-protein, before and after intervention
| Variables | Groups | Before (mean ± SD) | After (mean ± SD) | P-value |
|---|---|---|---|---|
| RASS, morning | Atorvastatin group | 1.6 ±0.84 | 0.35 ±0.37 | 0.001* |
| No atorvastatin group | 1.2 ±0.66 | 0.9 ±0.32 | 0.33* | |
| P-value | 0.13* | 0.03* | ||
| RASS, afternoon | Atorvastatin group | 1.3 ±0.48 | 0.33 ±0.25 | 0.03* |
| No atorvastatin group | 1.4 ±0.57 | 0.82 ±0.36 | 0.24* | |
| P-value | 0.68* | 0.04* |
Discussion
The main finding of the current study is that delirium-free status in both morning and afternoon were significantly higher in patients receiving atorvastatin (40 mg/day) compared with the control group. To the authors’ knowledge, this is the first study investigating the effect of atorvastatin on delirium status in ICU patients. Delirium is a severe dysfunction of the brain, which is associated with attention and cognition problems [34]. Disruption of clinical care including medication administration, nutrition, wound care, personal hygiene, discharge planning and family stress are the other negative health consequences of delirium in hospitalized patients [35]. Therefore, our results indicate that atorvastatin might have a beneficial effect in reducing brain dysfunction in ICU patients.
In line with our study, findings of two prospective observational cohorts showed that statin therapy was associated with less delirium in ICU patients [30, 36]. On the other hand, an observational cohort revealed no significant reduction of delirium in patients who were given statins prior to coronary revascularization [37]. Also, the results of a previous randomized controlled trial investigating the effect of rosuvastatin (40 mg loading dose and then 20 mg daily until the earliest of 3 days after discharge from intensive care, study day 28, or death) on delirium in patients with sepsis-associated acute respiratory distress syndrome showed that rosuvastatin had no effect on delirium status of the patients [38]. These inconsistent findings might be explained by differences in the hydrophilic and lipophilic properties of statins. Atorvastatin, used in this study, and simvastatin are lipophilic drugs that can enter into cells, be distributed in different tissues and pass the blood-brain barrier [38, 39] but hydrophilic statins such as pravastatin and rosuvastatin are mainly liver-specific [39].
The results of a recent systematic review investigating the pharmacological prevention and treatment of delirium in ICU patients showed that in only two included studies regarding statin therapy did these drugs cause a significant postoperative reduction in delirium rates in patients ≥ 60 years [40]. However, a recent systematic review and meta-analysis which reviewed the results of 6 studies with high heterogeneity (two studies in ICU patients and four studies in cardiac surgery patients) indicated that statins had no effects on delirium status in critically ill and cardiac surgery patients. It was suggested that more studies are required to clarify the relationship between statin therapy, particularly with respect to the type of statin and dosage, and delirium status, its mechanisms and outcomes in ICU patients [41].
In addition to their anti-inflammatory effects, statins may improve delirium through effects on N-methyl-d-aspartate (NMDA)-mediated glutamate excitotoxicity [42, 43] or endothelial function [44, 45], which might have beneficial effects on neuronal function. In addition, it has been reported that in response to the statin therapy, cerebral blood flow was increased in the ischemic penumbra, and the behavioral deficits were improved in the brain injury [46, 47].
Despite the novelty and the interventional design, some limitations should be acknowledged. First, we conducted this study in the general ICU, which included a diverse range of patients; hence, the generalization of the results to other hospitalized patients may not be applicable. Second, patients in the general ICU were administered a wide range of drugs, which might result in potential bias in the findings. However, using randomization could help to reduce these concerns. Third, we only assessed the effects of a single statin (atorvastatin) at a fixed dose and it is not clear whether other statins could have different effects and whether the observed effects of atorvastatin are dose-dependent. Given the findings of this study, additional trials exploring the efficacy of atorvastatin in reducing delirium status in critically ill patients seem to be helpful. Fourth, this study had a small size and was conducted as a single-center trial, thus making the generalizability of the findings difficult. Finally, the results need to be confirmed in larger and multi-center trials employing more specific indices of delirium evaluation such as the CAM-ICU (Cognitive Assessment Method for ICU) or ICDSC (Intensive Care Delirium Screening Checklist).
In conclusion, the findings of the present study showed that atorvastatin administration at a dose of 40 mg/day reduced delirium at both morning and afternoon time points in general ICU patients. Further investigations are required to confirm our findings and to compare the effects of different statins on delirium as well as the underlying mechanisms in ICU patients.