Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
CLINICAL RESEARCH
Sport-related entheseal adaptation and damage
in elite athletes: a cross-sectional ultrasound study
1
Department of Internal Medicine, Division of Rheumatology, Antalya Education and Research Hospital, Antalya, Turkey
2
Faculty of Medicine, Akdeniz University, Antalya, Turkey
3
Department of Sports Medicine, Izmir City Hospital, İzmir, Turkey
4
Department of Internal Medicine, Division of Rheumatology, Ondokuz Mayıs University, Samsun, Türkiye
5
The Ottawa University, Ottawa, Canada
6
The Ottawa Hospital Research Institute, Ottawa, Canada
Submission date: 2026-01-04
Final revision date: 2026-01-12
Acceptance date: 2026-01-29
Online publication date: 2026-04-09
Corresponding author
Sibel Bakirci
Department of Internal Medicine Division of Rheumatology Antalya Education and Research Hospital Antalya, Turkey
Department of Internal Medicine Division of Rheumatology Antalya Education and Research Hospital Antalya, Turkey
KEYWORDS
TOPICS
ABSTRACT
Introduction:
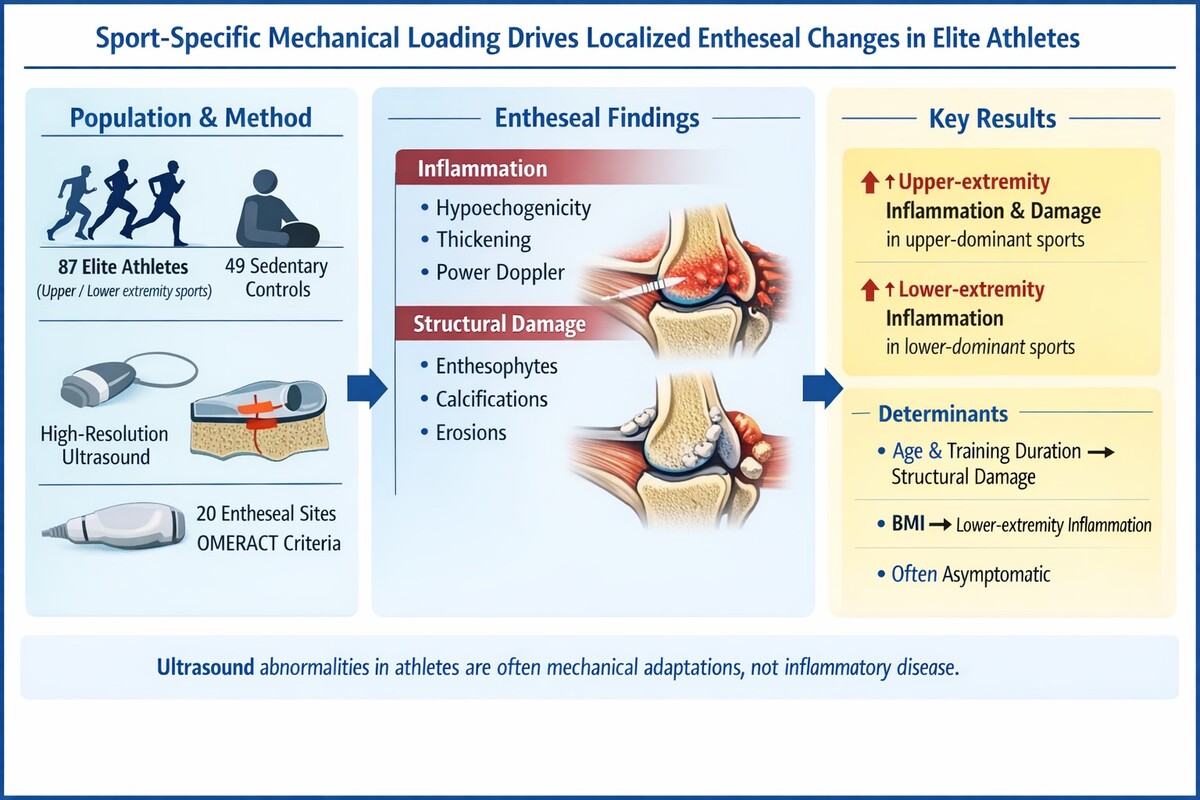
Entheses, the sites where tendons, ligaments, and joint capsules insert into bone, are highly susceptible to mechanical stress. While enthesitis is a cardinal feature of spondyloarthritis (SpA), repetitive mechanical loading in elite athletes can also induce entheseal changes. High-resolution ultrasound (US) allows for detailed assessment of entheseal inflammation and structural damage. However, the specific impact of sport-specific loading patterns on differentiating physiological entheseal adaptation from early pathology in asymptomatic elite athletes remains a critical area for investigation.
Material and methods:
This cross-sectional study evaluated 87 elite athletes from various sports (athletics, basketball, cycling, golf, handball, climbing, ar-chery) and 49 sedentary controls. Elite status was defined as training > 10 h/week for over 5 years. All participants underwent US evaluation of 20 bilateral entheseal sites. US findings were scored semi-quantitatively (0–3) for inflammation (hypoechogenicity, thickening, power Doppler [PD] signals) and damage (calcifications, erosions, enthesophytes) according to OMERACT definitions. Athletes were grouped by dominant extremity use (upper, lower, or both). Statistical comparisons and linear regression analyses were performed to identify associations between entheseal scores and demographic/training variables.
Results:
Elite athletes exhibited significantly higher entheseal inflammation and damage scores compared to sedentary controls (p < 0.05). Athletes practicing upper extremity-dominant sports (e.g., archery, climbing) showed significantly higher upper extremity inflam-mation (median [IQR], 1 [0–3] vs. 0 [0–1.5]; p = 0.010) and damage scores (median [IQR], 0 [0–3] vs. 0 [0–0]; p < 0.001) than con-trols. Similarly, lower extremity-dominant athletes (e.g., athletics, cycling) had significantly higher lower extremity inflammation scores (median [IQR], 2 [1–4] vs. 0 [0–1]; p < 0.001). Linear regression revealed that upper extremity inflammation was inde-pendently associated with age and weekly training duration, while lower extremity inflammation was solely associated with body mass index (BMI). Notably, a high prevalence of US abnormalities was observed despite many athletes being asymptomatic.
Conclusions:
Sport-specific mechanical loading is strongly associated with localized entheseal changes detectable by US in elite athletes. These findings underscore the importance of contextualizing US results within an athlete’s specific sport and loading history to distinguish between subclinical entheseal adaptation and true pathology.
Entheses, the sites where tendons, ligaments, and joint capsules insert into bone, are highly susceptible to mechanical stress. While enthesitis is a cardinal feature of spondyloarthritis (SpA), repetitive mechanical loading in elite athletes can also induce entheseal changes. High-resolution ultrasound (US) allows for detailed assessment of entheseal inflammation and structural damage. However, the specific impact of sport-specific loading patterns on differentiating physiological entheseal adaptation from early pathology in asymptomatic elite athletes remains a critical area for investigation.
Material and methods:
This cross-sectional study evaluated 87 elite athletes from various sports (athletics, basketball, cycling, golf, handball, climbing, ar-chery) and 49 sedentary controls. Elite status was defined as training > 10 h/week for over 5 years. All participants underwent US evaluation of 20 bilateral entheseal sites. US findings were scored semi-quantitatively (0–3) for inflammation (hypoechogenicity, thickening, power Doppler [PD] signals) and damage (calcifications, erosions, enthesophytes) according to OMERACT definitions. Athletes were grouped by dominant extremity use (upper, lower, or both). Statistical comparisons and linear regression analyses were performed to identify associations between entheseal scores and demographic/training variables.
Results:
Elite athletes exhibited significantly higher entheseal inflammation and damage scores compared to sedentary controls (p < 0.05). Athletes practicing upper extremity-dominant sports (e.g., archery, climbing) showed significantly higher upper extremity inflam-mation (median [IQR], 1 [0–3] vs. 0 [0–1.5]; p = 0.010) and damage scores (median [IQR], 0 [0–3] vs. 0 [0–0]; p < 0.001) than con-trols. Similarly, lower extremity-dominant athletes (e.g., athletics, cycling) had significantly higher lower extremity inflammation scores (median [IQR], 2 [1–4] vs. 0 [0–1]; p < 0.001). Linear regression revealed that upper extremity inflammation was inde-pendently associated with age and weekly training duration, while lower extremity inflammation was solely associated with body mass index (BMI). Notably, a high prevalence of US abnormalities was observed despite many athletes being asymptomatic.
Conclusions:
Sport-specific mechanical loading is strongly associated with localized entheseal changes detectable by US in elite athletes. These findings underscore the importance of contextualizing US results within an athlete’s specific sport and loading history to distinguish between subclinical entheseal adaptation and true pathology.
REFERENCES (18)
1.
Benjamin M, McGonagle D. The enthesis organ concept and its relevance to the spondyloarthropathies. Arthritis Rheum 2009; 61: 142-7.
2.
Bakirci S, Solmaz D, Stephenson W, Eder L, Roth J, Aydin SZ. Entheseal changes in response to age, body mass index, and physical activity: an ultrasound study in healthy people. J Rheumatol 2020; 47: 968-72.
3.
Kehl AS, Koreth J, Manadan AM, et al. Enthesitis: pathogenesis, diagnosis, and treatment. Am J Med 2016; 129: 1014-21.
4.
Sabido-Sauri R. Enthesopathies – mechanical, inflammatory or both? Best Pract Res Clin Rheumatol 2024; 38: 101908.
5.
Simon D, Lories RJ, D’Agostino MA, et al. Biomechanical stress in the context of competitive sports training triggers enthesitis. Arthritis Res Ther 2021; 23: 172.
6.
De Lorenzis E, D’Agostino MA, Lories RJ, et al. Concepts of entheseal pain. Arthritis Rheumatol 2023; 75: 493-8.
7.
Di Matteo A, D’Agostino MA, Lories RJ, et al. Reliability of a consensus-based ultrasound definition and scoring for enthesitis in spondy-loarthritis and psoriatic arthritis: an OMERACT US initiative. Ann Rheum Dis 2024; 83 (Suppl 1): 114.
8.
Song Y, D’Agostino MA, Lories RJ, et al. A narrative review of the design of ultrasound indices for enthesitis in spondyloarthritis. J Clin Med 2022; 11: 1084.
9.
Di Matteo A, D’Agostino MA, Lories RJ, et al. DEUS Enthesitis Index (DEI): a new tool integrating. RMD Open 2025; 11: e005496.
10.
Bruyn GA, D’Agostino MA, Lories RJ, et al. OMERACT definitions for ultrasonographic pathologies of the entheses. J Rheumatol 2019; 46: 1388-94.
11.
Dean E, D’Agostino MA, Lories RJ, et al. The GRAPPA Sonographic Enthesitis Workshop. J Rheumatol 2019; 46: 1395-400.
12.
Mascarenhas S, Couette N. A systematic review of the inclusion of non-inflammatory ultrasonographic enthesopathy findings in enthesitis scoring indices. Diagnostics 2021; 11: 669.
13.
Yadav A, Mehra N, Pal S, et al. Evaluation of enthesitis in patients with juvenile idiopathic arthritis by power color and spectral Doppler ul-trasonography. Eur J Rheumatol 2020; 7: 173-8.
14.
Lanfranchi MA, D’Agostino MA, Lories RJ, et al. Are ultrasound findings similar in patients with axial spondyloarthritis and athletes? A comparative study. J Rheumatol 2017; 44: 609-15.
15.
Perrotta FM, D’Agostino MA, Lories RJ, et al. Ultrasonographic evaluation of entheseal fibrocartilage in patients with psoriatic arthritis, ath-letes and healthy controls: a comparison study. Diagnostics 2023; 13: 1446.
16.
Benítez-Martínez JC, D’Agostino MA, Lories RJ, et al. Lower limb dominance, morphology, and sonographic abnormalities of the patellar tendon in elite basketball players: a cross-sectional study. J Athlet Training 2019; 54: 1280-6.
17.
Ali Ismail AM, Abd El-Azeim AS, El-Sayed Felaya EE. Integrated neuromuscular inhibition technique versus spray and stretch technique in neck pain patients with upper trapezius trigger points. J Manual Manip Ther 2024; 32: 141-9.
18.
Yasin FB, Abdulla FA, Madhanagopal J, Shallan A, Sheeha BB, Ismail AMA. New flexi force sensor integrated seat pan device with real-time biofeedback for instant poor posture alert among office workers: a pilot study. Irish J Med Sci 2025; 194: 2225-32.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.