Colorectal cancer (CRC) remains a leading cause of cancer-related death, and many patients still present with metastatic or recurrent disease despite advances in surgery and systemic therapy [1, 2]. Metabolic reprogramming is a hallmark of cancer and an attractive source of biomarkers and therapeutic targets [3]. Arachidonic-acid-derived epoxyeicosatrienoic acids (EETs), generated mainly by CYP2C and CYP2J mono-oxygenases, regulate inflammation, angiogenesis, and cell survival, and have been implicated in several malignancies [4–8]. However, the contribution of CYP2J2 and its EET products to CRC biology is not clearly defined.
Guided by untargeted tissue metabolomics, we investigated arachidonic-acid-related signatures in CRC, evaluated CYP2J2 expression and its relationship with clinicopathologic features and epithelial differentiation markers (CDX2, CK20, CK7), and functionally examined CYP2J2 in CRC cell lines.
Methods
Paired CRC and adjacent mucosa were collected from patients undergoing curative resection. All participants provided written informed consent, and the study was approved by the institutional ethics committee. Treatment-naïve primary CRC patients undergoing curative resection (2020–2024) with paired tumor/adjacent mucosa were included. Patients with a history of prior malignancy/IBD/infection or inadequate tissue were excluded. No patients received neoadjuvant therapy. The study cohort comprised 30 patients aged 40–65, 18 male and 12 female. Tissue extracts were analyzed by LC–MS-based untargeted metabolomics using paired samples from n = 30 colorectal cancer patients. Data were normalized and log₂-transformed before analysis. Differential metabolites were identified using variable-importance-in-projection (VIP) > 1.0 and adjusted p < 0.05 (Benjamini–Hochberg FDR) as significance thresholds. Batch effects were evaluated and corrected using QC-based robust LOESS signal correction (QC-RLSC) prior to statistical analysis. Differential metabolites were identified by multivariable statistics and mapped to KEGG pathways (Supplementary Figure S1); particular attention was given to arachidonic-acid-related mediators, including 5,6-dihydroxyeicosatrienoic acid (5,6-DHET), the soluble epoxide hydrolase product of 5,6-EET.
CYP2J2 mRNA and protein were assessed in CRC and mucosa, and clinicopathologic variables (stage, tumor size, Ki-67 index) were correlated with CYP2J2 expression. Co-expression with CDX2, CK20, and CK7 was analyzed in clinical specimens, and expression of CDX2, CK20, and CK7 was summarized at the cohort level.
HCT116 and HT29 cells were subjected to CYP2J2 gain- and loss-of-function using overexpression plasmids (OE/OX) and shRNA-mediated knockdown (sh/KO). Proliferation was measured by CCK-8 and colony-formation assays, invasion by Matrigel-coated Transwell chambers, apoptosis by TUNEL and flow cytometry, and cell-cycle distribution by propidium-iodide staining. All experiments were performed in triplicate biological replicates. Data are presented as mean ± SD, and statistical comparisons were made using two-tailed Student’s t-tests or one-way ANOVA with Tukey’s post-hoc correction where appropriate. Western blots quantified proliferation-, apoptosis-, and invasion-related proteins.
Results
Metabolomic reprogramming and arachidonic-acid-related changes in CRC
Hierarchical clustering and volcano-plot analysis showed that dozens of metabolites were significantly up- or downregulated in CRC compared with adjacent mucosa (Supplementary Figure S1). Among them, 4-fluorocatechol and phosphoenolpyruvic acid represented markedly downregulated and upregulated species, respectively, and metabolites such as beta-patchoulene and AFN911 had high VIP scores, indicating strong discriminative value. Joint pathway analysis highlighted “TGF-β signaling”, “proteoglycans in cancer”, “glutathione metabolism”, and “central carbon metabolism in cancer”, underscoring broad metabolic rewiring in CRC (Supplementary Figure S2).
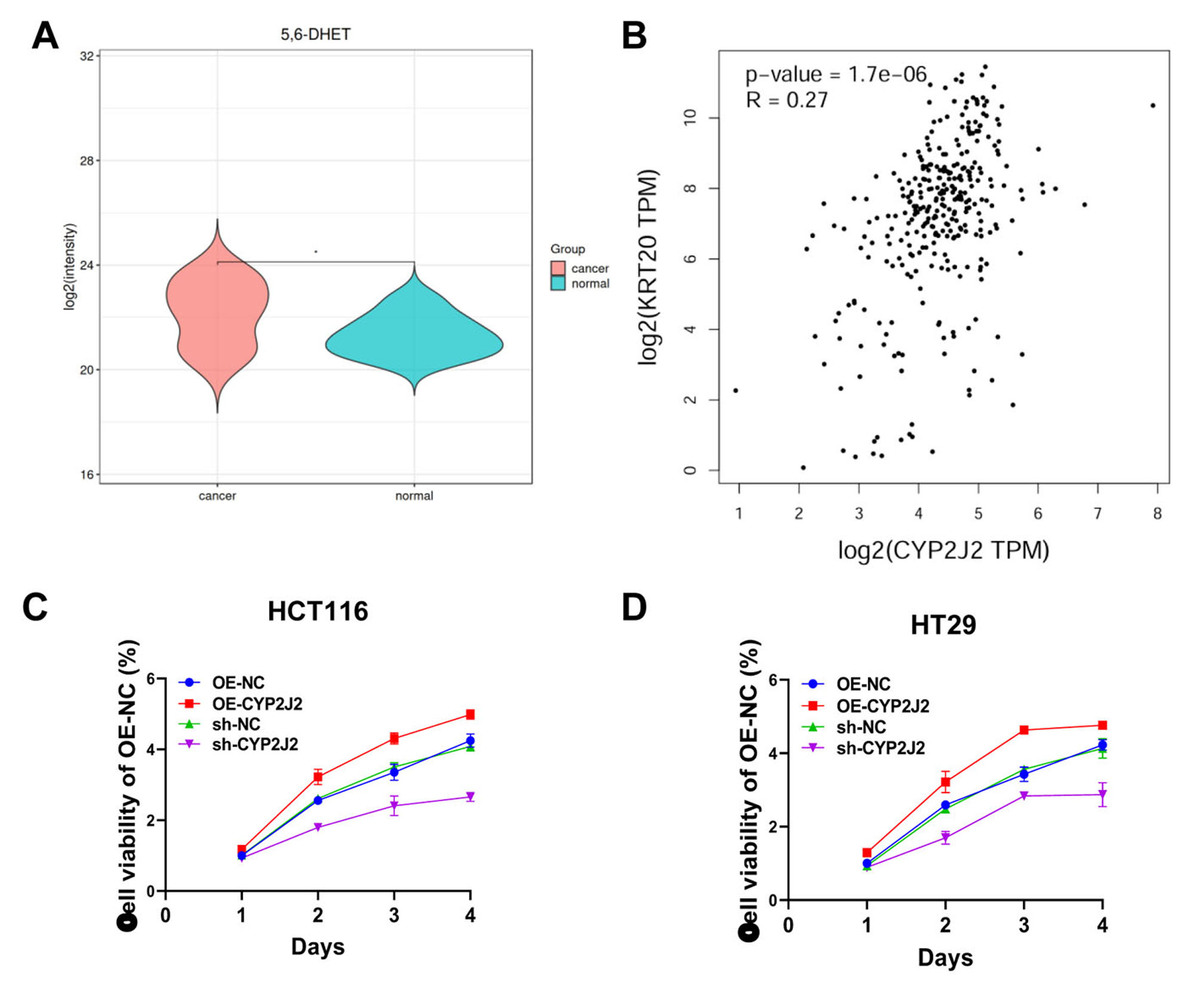
Arachidonic-acid-related metabolites were prominently altered. In particular, 5,6-DHET was significantly enriched in CRC tissue compared with normal mucosa, suggesting enhanced CYP-epoxygenase activity and EET turnover in the tumor microenvironment, consistent with possible involvement of the EET–soluble epoxide hydrolase axis in CRC pathogenesis (Figure 1 A).
Figure 1
A – Violin plot showing the distribution of 5,6-dihydroxyeicosatrienoic acid (5,6-DHET) in colorectal cancer and normal tissues. Color: Red represents colorectal cancer tissues, blue represents normal tissues. B – Scatter plot showing the correlation between CYP2J2 and KRT20 (CK20) expression. Axes indicate log2(CYP2J2 TPM) and log2(KRT20 TPM). n = 269. p-value: Significance of the correlation between the two gene expression levels. C, D – Cell proliferation curves are presented as mean ±} SD (n = 3 biological replicates). Statistical significance between groups across time was assessed by two-way ANOVA (group × time) with Sidak’s multiple-comparisons test. Control groups: OE-NC (overexpression negative control), sh-NC (knockdown negative control). Experimental groups: OE-CYP2J2 (overexpression of CYP2J2), sh-CYP2J2 (knockdown of CYP2J2)
CYP2J2 expression, epithelial markers, and clinicopathologic correlations
CYP2J2 expression was significantly higher in CRC tissue than in matched mucosa. Survival analysis showed no significant difference between CYP2J2-high and CYP2J2-low groups (log-rank p = 0.27), indicating that CYP2J2 alone is not an independent prognostic factor in this cohort. However, CYP2J2 levels increased with advancing clinical stage (p < 0.01) and correlated strongly with tumor size (R² = 0.85, p < 0.001) and Ki-67 index (R² = 0.88, p < 0.001), linking CYP2J2 to tumor burden and proliferative activity rather than long-term outcome (Supplementary Figure S3).
Co-expression analyses demonstrated that CYP2J2 is integrated into the intestinal epithelial differentiation program. CDX2 and CK20 were positively expressed in all CRC samples, whereas CK7 was consistently negative, confirming typical colorectal immunophenotypes. Quantitatively, CYP2J2 and CDX2 showed only a weak correlation (R = 0.19, p = 0.0009), whereas CYP2J2 and CK20 exhibited a more pronounced positive correlation (R = 0.27, p = 1.7 × 10–6) (Figure 1 B and Supplementary Figure S3). Thus, CYP2J2 is preferentially co-expressed with CK20-defined intestinal epithelial cells while retaining relative independence from CDX2, supporting its role as an epithelial, CRC-linked metabolic enzyme.
CYP2J2 promotes proliferation, invasion, and cell-cycle progression while inhibiting apoptosis
In both HCT116 and HT29 cells, CYP2J2 overexpression significantly increased proliferation in CCK-8 and colony-formation assays, whereas shRNA-mediated knockdown reduced proliferative capacity, indicating a positive relationship between CYP2J2 levels and cell growth. Transwell assays showed that CYP2J2 overexpression enhanced, and CYP2J2 knockdown diminished, invasive ability, indicating a key role for CYP2J2 in promoting colorectal cancer cell invasion (Supplementary Figure S4).
TUNEL and flow-cytometric analyses revealed that CYP2J2 knockdown markedly increased apoptosis, while CYP2J2 overexpression reduced apoptotic fractions, suggesting that CYP2J2 expression inhibits apoptosis and favors cell survival (Supplementary Figure S4). Cell-cycle profiling demonstrated that CYP2J2 overexpression decreased the G0/G1 fraction and increased the S and G2/M fractions; conversely, CYP2J2 knockdown caused G0/G1 accumulation and reduced cycling fractions (Supplementary Figure S5). Western-blot analysis further showed that proliferation-related proteins were upregulated and apoptosis-associated proteins were downregulated in CYP2J2-overexpressing cells, with the opposite pattern after CYP2J2 knockdown (Supplementary Figures S6 and S7). Together, these data indicate that CYP2J2 plays a crucial role in regulating the cell cycle and apoptosis in CRC cells.
Discussion
This study integrates metabolomics, clinical tissue analyses, and functional studies to suggest that CYP2J2 may serve as a metabolic node potentially linking reprogramming with malignant behavior in CRC. Untargeted metabolomics revealed a CRC-specific signature characterized by dysregulated central-carbon, glutathione, TGF-β-related, and cancer-associated proteoglycan pathways and by accumulation of arachidonic-acid-derived 5,6-DHET. Within this context, CYP2J2 was overexpressed, strongly correlated with tumor size and Ki-67 index, and preferentially co-expressed with the intestinal epithelial marker CK20, while CDX2 showed only weak co-variation and CK7 remained negative. These findings situate CYP2J2 within a typical CRC differentiation background but highlight its specific association with proliferative burden.
Functionally, CYP2J2 enhanced proliferation, invasion, and cell-cycle progression and suppressed apoptosis in CRC cell lines, consistent with a model in which CYP2J2-derived EETs foster EGFR–MAPK–PI3K signaling and metastatic potential, although direct evidence was not obtained in this study. Collectively, our results support CYP2J2 as a biomarker reflecting proliferative aggressiveness rather than survival per se, and as a promising therapeutic target. Isoform-selective inhibition of CYP2J2 or modulation of the EET–soluble epoxide hydrolase axis, potentially combined with standard chemo- or radiotherapy, warrants further evaluation in preclinical CRC models, while carefully considering the physiological roles of CYP2J2 in normal tissues. Limitations of this study include the relatively small metabolomics cohort, the lack of in vivo validation, and the absence of direct measurements of EETs/EET-pathway activity, which are inferred here primarily from increased 5,6-DHET levels.