Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
CARDIOLOGY / CLINICAL RESEARCH
Interplay between parathyroid hormone concentration and valvular and aortic calcifications
1
Department of Cardiac Surgery and Transplantology, Poznan University of Medical Sciences, Poznan, Poland
2
First Department of Cardiology, Medical University of Warsaw, Poland
3
Department of Radiology, Poznań University of Medical Sciences, Poznan, Poland
4
Thoracic Research Centre, Collegium Medicum Nicolaus Copernicus University, Innovative Medical Forum, Bydgoszcz, Poland
5
First Department of Cardiology, Poznan University of Medical Sciences, Poznan, Poland
6
Institute of Heart Diseases, Wroclaw Medical University, Wroclaw, Poland
7
Department of Biomedical Sciences, and Department of Medicine, Cedars-Sinai Medical Center, Biomedical Imaging Research Institute, Los Angeles, CA, USA
These authors had equal contribution to this work
Submission date: 2024-11-18
Final revision date: 2025-02-04
Acceptance date: 2025-03-28
Online publication date: 2025-06-08
Publication date: 2026-04-30
Corresponding author
Arch Med Sci 2026;22(2):735-742
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Recently, a relationship between hyperparathyroidism and cardiovascular disorders has been highlighted. The current study aimed to identify a potential relationship between parathyroid hormone (PTH) and valvular calcification performance. A secondary aim was to evaluate the potential association between PTH concentration and post-procedural outcomes after transcatheter aortic valve implantation (TAVI).
Material and methods:
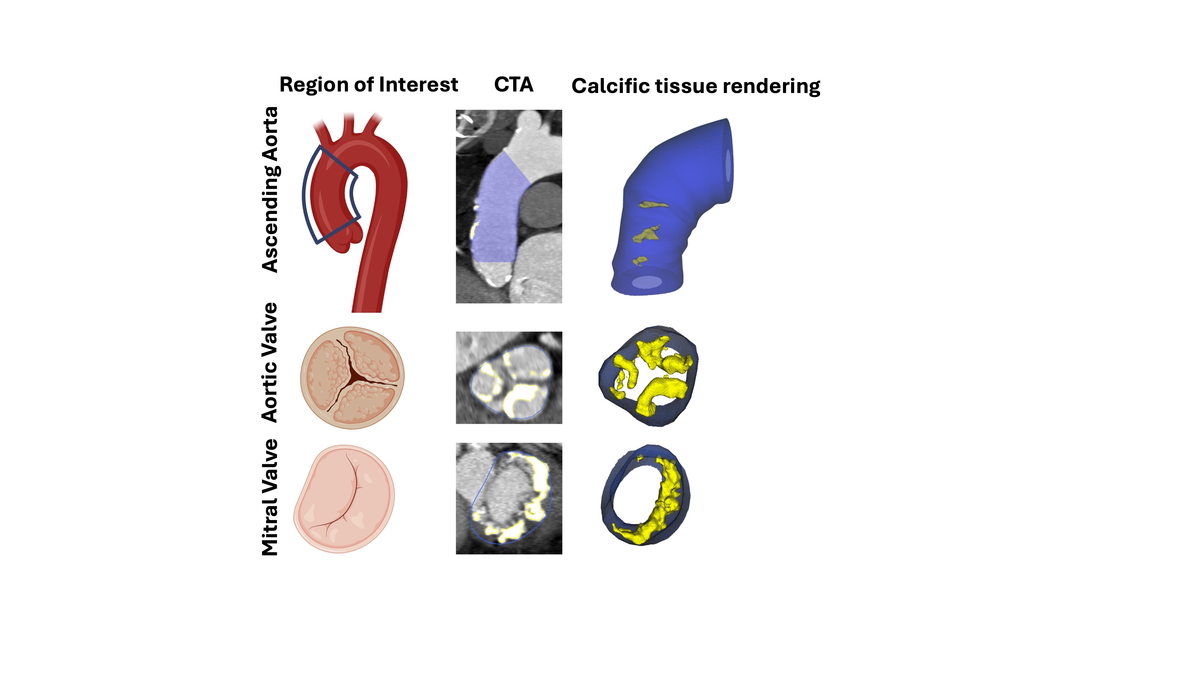
Patients with severe symptomatic aortic stenosis were evaluated for study eligibility. Demographics, clinical data, and blood samples were collected. Pre-procedurally, echocardiography and computed tomography (CT) were performed. Quantitative evaluation of calcific tissue was conducted over the three regions of interest – ascending aorta, aortic and mitral valves – using semiautomated software.
Results:
The final study group comprised 89 patients (50 females, median (Q1-3) age of 77 (72–82) years. Increased PTH concentration was associated with a higher peak aortic gradient (p = 0.024), but not with mean aortic gradient or mitral annular calcification occurrence. CT analysis revealed an association between increased PTH and mean calcific tissue attenuation in the mitral (p = 0.004) and aortic valves (p < 0.001) and ascending aorta (p < 0.001) but no relationship with calcium volume in the regions of interest. Increased PTH did not differ between patients with and without paravalvular leak or new pacemaker implantation.
Conclusions:
Increased PTH concentration is associated with calcific tissue attenuation but not calcium volume, suggesting that PTH may influence the degree of calcium accumulation in degenerated regions. PTH could potentially serve as a biomarker of calcific loading in valvular heart disease. However, PTH concentration does not appear to be associated with the rate of complications following TAVI.
Recently, a relationship between hyperparathyroidism and cardiovascular disorders has been highlighted. The current study aimed to identify a potential relationship between parathyroid hormone (PTH) and valvular calcification performance. A secondary aim was to evaluate the potential association between PTH concentration and post-procedural outcomes after transcatheter aortic valve implantation (TAVI).
Material and methods:
Patients with severe symptomatic aortic stenosis were evaluated for study eligibility. Demographics, clinical data, and blood samples were collected. Pre-procedurally, echocardiography and computed tomography (CT) were performed. Quantitative evaluation of calcific tissue was conducted over the three regions of interest – ascending aorta, aortic and mitral valves – using semiautomated software.
Results:
The final study group comprised 89 patients (50 females, median (Q1-3) age of 77 (72–82) years. Increased PTH concentration was associated with a higher peak aortic gradient (p = 0.024), but not with mean aortic gradient or mitral annular calcification occurrence. CT analysis revealed an association between increased PTH and mean calcific tissue attenuation in the mitral (p = 0.004) and aortic valves (p < 0.001) and ascending aorta (p < 0.001) but no relationship with calcium volume in the regions of interest. Increased PTH did not differ between patients with and without paravalvular leak or new pacemaker implantation.
Conclusions:
Increased PTH concentration is associated with calcific tissue attenuation but not calcium volume, suggesting that PTH may influence the degree of calcium accumulation in degenerated regions. PTH could potentially serve as a biomarker of calcific loading in valvular heart disease. However, PTH concentration does not appear to be associated with the rate of complications following TAVI.
REFERENCES (38)
1.
Van Hemelrijck M, Taramasso M, Gülmez G, Maisano F, Mestres CA. Mitral annular calcification: challenges and future perspectives. Indian J Thorac Cardiovasc Surg 2020; 36: 397-403.
2.
Trevisan C, Rossi A, Curreri C. Increased parathyroid hormone concentration as a biomarker of atrial fibrillation in severe aortic stenosis: editorial comment. Kardiol Pol 2024; 84: 1055-6.
3.
Li M, Cheng J, Zhao J, et al. Relationship between intact parathyroid hormone and all-cause death, cardiovascular events, and ectopic calcification in patients with diabetic kidney disease: a retrospective study. Diabetes Res Clin Pract 2021; 177: 108926.
4.
Hagström E, Hellman P, Larsson TE, et al. Plasma parathyroid hormone and the risk of cardiovascular mortality in the community. Circulation 2009; 119: 2765-71.
5.
Malluche HH, Blomquist G, Monier-Faugere MC, Cantor TL, Davenport DL. High parathyroid hormone level and osteoporosis predict progression of coronary artery calcification in patients on dialysis. J Am Soc Nephrol 2015; 26: 2534-44.
6.
Kobayashi T, Kitahara H, Kato K, Saito Y, Kobayashi Y. Impact of parathyroid hormone level on intracoronary calcification and short- and long-term outcomes in dialysis patients undergoing percutaneous coronary intervention. Circ J 2023; 87: 247-55.
7.
Wu GY, Xu BD, Wu T, et al. Correlation between serum parathyroid hormone levels and coronary artery calcification in patients without renal failure. Biomed Rep 2016; 5: 601-6.
8.
Osto E, Fallo F, Pelizzo MR, et al. Coronary microvascular dysfunction induced by primary hyperparathyroidism is restored after parathyroidectomy. Circulation 2012; 126: 1031-9.
9.
Nägele MP, Barthelmes J, Kreysing L, et al. Endocrine hormone imbalance in heart failure with reduced ejection fraction: a cross-sectional study. Health Sci Rep 2022; 5: e880.
10.
Scicchitano P, Iacoviello M, Passantino A, et al. Plasma levels of intact parathyroid hormone and congestion burden in heart failure: clinical correlations and prognostic role. J Cardiovasc Dev Dis 2022; 9: 334.
11.
Dahlen B, Müller F, Tröbs SO, et al. Sex-specific relationship between parathyroid hormone and platelet indices in phenotypes of heart failure-results from the MyoVasc Study. Front Cardiovasc Med 2021; 8: 682521.
12.
Olasińska-Wiśniewska A, Urbanowicz T, Kübler P, et al. Increased parathyroid hormone concentration as a biomarker of atrial fibrillation in severe aortic stenosis. Kardiol Pol 2024; 82: 1076-82.
13.
Vahanian A, Beyersdorf F, Praz F, et al.; ESC/EACTS Scientific Document Group. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 2022; 43: 561-632.
14.
Salahuddin A, Aronow WS, Spevack DM. At what flow rate does the aortic valve gradient become severely elevated? Implications for guideline recommendations on aortic valve area cutoffs. Arch Med Sci 2021; 20: 713-8.
15.
Grodecki K, Olasińska-Wiśniewska A, Cyran A, et al. Quantification of aortic valve fibrotic and calcific tissue from CTA: prospective comparison with histology. Radiology 2024; 312: e240229.
16.
Grodecki K, Tamarappoo BK, Huczek Z, et al. Non-calcific aortic tissue quantified from computed tomography angiography improves diagnosis and prognostication of patients referred for transcatheter aortic valve implantation. Eur Heart J Cardiovasc Imaging 2021; 22: 626-35.
17.
Mueller KAL, Langnau C, Harm T, et al. Macrophage migration inhibitory factor promotes thromboinflammation and predicts fast progression of aortic stenosis. Arter Thromb Vasc Biol 2024; 44: 2118-35.
18.
Olasińska-Wiśniewska A, Urbanowicz T, Grodecki K, et al. Monocyte-to-lymphocyte ratio correlates with parathyroid hormone concentration in patients with severe symptomatic aortic stenosis. Adv Med Sci 2023; 68: 396-401.
19.
Butcher JT, Nerem RM. Valvular endothelial cells and the mechanoregulation of valvular pathology. Philos Trans R Soc Lond B Biol Sci 2007; 362: 1445-57.
20.
Richards JM, Kunitake JAMR, Hunt HB, et al. Crystallinity of hydroxyapatite drives myofibroblastic activation and calcification in aortic valves. Acta Biomater 2018; 71: 24-36.
21.
Müller AM, Cronen C, Kupferwasser LI, Oelert H, Müller KM, Kirkpatrick CJ. Expression of endothelial cell adhesion molecules on heart valves: up-regulation in degeneration as well as acute endocarditis. J Pathol 2000; 191: 54-60.
22.
Goettsch C, Iwata H, Aikawa E. Parathyroid hormone: critical bridge between bone metabolism and cardiovascular disease. Arterioscler Thromb Vasc Biol 2014; 34: 1333-5.
23.
Fujii H. Association between parathyroid hormone and cardiovascular disease. Ther Apher Dial 2018; 22: 236-41.
24.
Neves KR, Graciolli FG, dos Reis LM, et al. Vascular calcification: contribution of parathyroid hormone in renal failure. Kidney Int 2007; 71: 1262-70.
25.
Nusca A, Viscusi MM, Circhetta S, et al. Impact of burden and distribution of aortic valve calcification on the hemodynamic performance and procedural outcomes of a self-expanding, intra-annular transcatheter aortic valve system. Int J Cardiovasc Imaging 2024; 40: 2545-58.
26.
Yao JQ, Deng ZJ, Fang MX, et al. Expression of serum inflammatory cytokines and oxidative stress markers and their correlation with coronary artery calcium score in patients with coronary heart disease. Arch Med Sci 2020; 19: 1709-13.
27.
Myśliwiec M, Bandura M, Wołoszyn-Durkiewicz A, et al. 2024 Polish recommendations for the management of familial hypercholesterolemia in children and adolescents. Arch Med Sci 2024; 20: 1741-53.
28.
Banach M, Surma S, Bielecka-Dąbrowa A, et al. Rosuvastatin-based combination treatment with acetylsalicylic acid or ezetimibe in the management of patients at high and very high cardiovascular risk. Expert Opinion Paper of the Polish Lipid Association 2025. Arch Med Sci 2025; 21: 1-15.
29.
Marquis-Gravel G, Redfors B, Leon MB, Généreux P. Medical treatment of aortic stenosis. Circulation 2016; 134: 1766-84.
30.
Afshar M, Yazdan-Ashoori S, Engert JC, Thanassoulis G. Drugs for prevention and treatment of aortic stenosis: how close are we? Can J Cardiol 2021; 37: 1016-26.
31.
Cowell SJ, Newby DE, Prescott RJ, et al.; Scottish Aortic Stenosis and Lipid Lowering Trial, Impact on Regression (SALTIRE) Investigators. A randomized trial of intensive lipid-lowering therapy in calcific aortic stenosis. N Engl J Med 2005; 352: 2389-97.
32.
Chan KL, Teo K, Dumesnil JG, Ni A, Tam J; ASTRONOMER Investigators. Effect of Lipid lowering with rosuvastatin on progression of aortic stenosis: results of the aortic stenosis progression observation: measuring effects of rosuvastatin (ASTRONOMER) trial. Circulation 2010; 121: 306-14.
33.
Sosnowska B, Stepinska J, Mitkowski P, et al. Recommendations of the Experts of the Polish Cardiac Society (PCS) and the Polish Lipid Association (PoLA) on the diagnosis and management of elevated lipoprotein(a) levels. Arch Med Sci 2024; 20: 8-27.
34.
Santangelo G, Faggiano A, Bernardi N, Carugo S, Giammanco A, Faggiano P. Lipoprotein(a) and aortic valve stenosis: a casual or causal association? Nutr Metab Cardiovasc Dis 2022; 32: 309-17.
35.
Thanassoulis G, Campbell CY, Owens DS, et al.; CHARGE Extracoronary Calcium Working Group. Genetic associations with valvular calcification and aortic stenosis. N Engl J Med 2013; 368: 503-12.
36.
Capoulade R, Chan KL, Yeang C, et al. Oxidized phospholipids, lipoprotein(a), and progression of calcific aortic valve stenosis. J Am Coll Cardiol 2015; 66: 1236-46.
37.
Sakai M, Tokunaga S, Kawai M, et al. Evocalcet prevents ectopic calcification and parathyroid hyperplasia in rats with secondary hyperparathyroidism. PLoS One 2020; 15: e0232428.
38.
Solomon A, Birkenfeld S. Rapid progression of aortic stenosis after initiation of teriparatide treatment: a case report. Cardiovasc Endocrinol Metab 2020; 10: 56-8.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.