Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
UROLOGY / CLINICAL RESEARCH
Combining the PI-RADS v2.1 score with PSAD improves the diagnostic accuracy of transition zone prostate cancer
1
Department of Radiology, The Second Affiliated Hospital, Xi’ an Jiaotong University, China
2
Department of Plastic and Reconstructive Surgery, Xijing Hospital, Fourth Military Medical University, China
Submission date: 2025-05-20
Final revision date: 2025-08-06
Acceptance date: 2025-10-17
Online publication date: 2026-04-03
Publication date: 2026-04-30
Corresponding author
Jinman Zhong
Department of Radiology The Second Affiliated Hospital Xi’ an Jiaotong University, China
Department of Radiology The Second Affiliated Hospital Xi’ an Jiaotong University, China
Arch Med Sci 2026;22(2):884-891
KEYWORDS
prostate cancertransition zonebenign prostatic hyperplasiaprostate-specific antigen densityProstate Imaging Reporting and Data System
TOPICS
ABSTRACT
Introduction:
Transition zone (TZ) prostate cancer poses diagnostic challenges due to overlapping imaging features with benign prostatic hyperplasia (BPH). This study aimed to investigate whether Prostate Imaging Reporting and Data System (PI-RADS) v2.1 combined with prostate-specific antigen density (PSAD) can enhance diagnostic accuracy in distinguishing TZ cancer from BPH.
Material and methods:
The retrospective study included 377 patients divided into two groups: the TZ cancer group (n = 139) and the BPH group (n = 238). Two radiologists independently reviewed prostate MR images of each patient and assigned a PI-RADS score for the TZ lesion. Clinical characteristics were compared between the two groups using the c2 test, t-test, or Mann-Whitney U-test. The diagnostic performance of PI-RADS scores, PSAD, and the combined parameters was determined by ROC curve analysis.
Results:
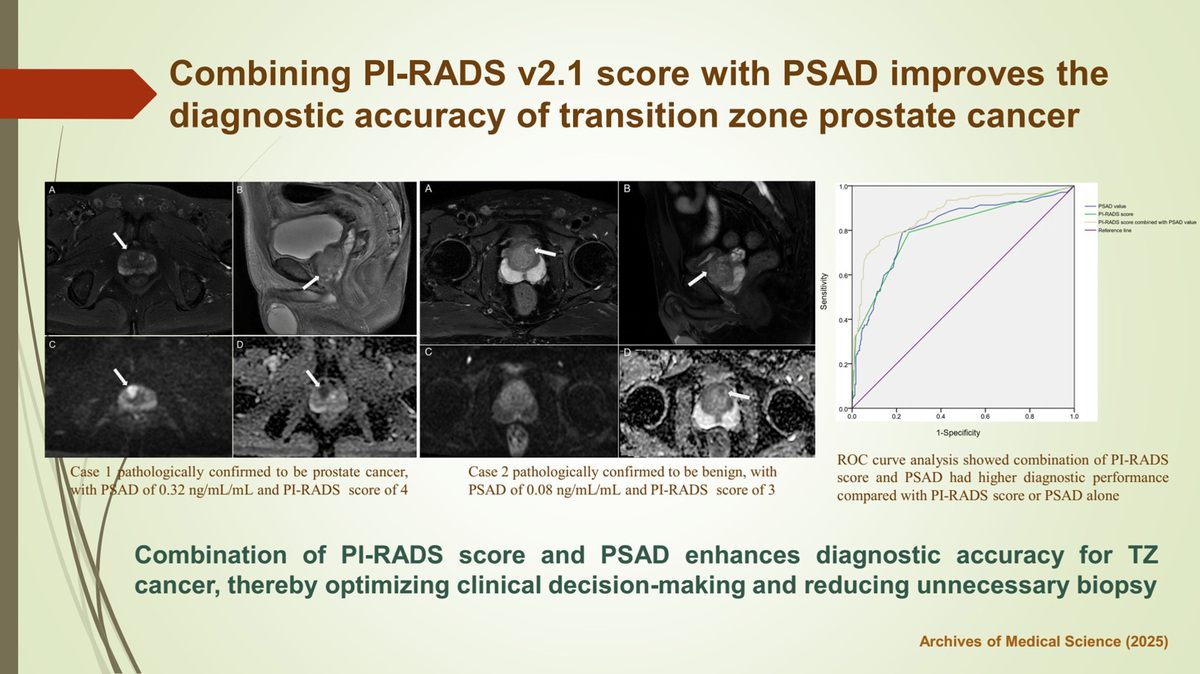
Statistically significant differences were found in PSA, prostate volume, PSAD, and PI-RADS scores between the two groups (all p < 0.0001). No difference in age was observed between the two groups (p = 0.602). The area under the curve (AUC) for PI-RADS v2.1 alone was 0.802 (95% CI: 0.759–0.841) with a sensitivity of 79.14% and specificity of 74.37% at a cutoff of ³ 4. The AUC for PSAD alone was 0.808 (95% CI: 0.765–0.847) with a sensitivity of 79.14% and specificity of 77.31% at a cutoff of 0.22 ng/ml/ml. Combining PI-RADS and PSAD yielded an AUC of 0.873 (95% CI: 0.835–0.905), with a sensitivity of 79.26% and specificity of 86.55%.
Conclusions:
The combination of PI-RADS v2.1 and PSAD enhances the diagnostic accuracy for TZ cancer, thereby reducing unnecessary invasive procedures.
Transition zone (TZ) prostate cancer poses diagnostic challenges due to overlapping imaging features with benign prostatic hyperplasia (BPH). This study aimed to investigate whether Prostate Imaging Reporting and Data System (PI-RADS) v2.1 combined with prostate-specific antigen density (PSAD) can enhance diagnostic accuracy in distinguishing TZ cancer from BPH.
Material and methods:
The retrospective study included 377 patients divided into two groups: the TZ cancer group (n = 139) and the BPH group (n = 238). Two radiologists independently reviewed prostate MR images of each patient and assigned a PI-RADS score for the TZ lesion. Clinical characteristics were compared between the two groups using the c2 test, t-test, or Mann-Whitney U-test. The diagnostic performance of PI-RADS scores, PSAD, and the combined parameters was determined by ROC curve analysis.
Results:
Statistically significant differences were found in PSA, prostate volume, PSAD, and PI-RADS scores between the two groups (all p < 0.0001). No difference in age was observed between the two groups (p = 0.602). The area under the curve (AUC) for PI-RADS v2.1 alone was 0.802 (95% CI: 0.759–0.841) with a sensitivity of 79.14% and specificity of 74.37% at a cutoff of ³ 4. The AUC for PSAD alone was 0.808 (95% CI: 0.765–0.847) with a sensitivity of 79.14% and specificity of 77.31% at a cutoff of 0.22 ng/ml/ml. Combining PI-RADS and PSAD yielded an AUC of 0.873 (95% CI: 0.835–0.905), with a sensitivity of 79.26% and specificity of 86.55%.
Conclusions:
The combination of PI-RADS v2.1 and PSAD enhances the diagnostic accuracy for TZ cancer, thereby reducing unnecessary invasive procedures.
REFERENCES (22)
1.
Bergengren O, Pekala KR, Matsoukas K, et al. 2022 update on prostate cancer epidemiology and risk factors-a systematic review]. Eur Urol 2023; 84: 191-206.
2.
Caner E, Serkan A, Neslihan K, et al. A new ASAP scoring system and risk table to predict second prostate biopsy outcomes. Arch Med Sci 2024; 20: 1894-901.
3.
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021; 71: 209-49.
4.
Ali A, Du Feu A, Oliveira P, et al. Prostate zones and cancer: lost in transition? Nat Rev Urol 2022; 19: 101-15.
5.
Elbuluk O, Muradyan N, Shih J, et al. Differentiating transition zone cancers from benign prostatic hyperplasia by quantitative multiparametric magnetic resonance imaging. J Comput Assist Tomogr 2016; 40: 218-24.
6.
Turkbey B, Rosenkrantz AB, Haider MA, et al. Prostate imaging reporting and data system version 2.1: 2019 update of prostate imaging reporting and data system version 2. Eur Urol 2019; 76: 340-51.
7.
Chatterjee A, Thomas S, Oto A. Prostate MR: pitfalls and benign lesions. Abdom Radiol 2020; 45: 2154-64.
8.
Chatterjee A, Gallan AJ, He D, et al. Revisiting quantitative multi-parametric MRI of benign prostatic hyperplasia and its differentiation from transition zone cancer. Abdom Radiol 2019; 44: 2233-43.
9.
Chesnais AL, Niaf E, Bratan F, et al. Differentiation of transitional zone prostate cancer from benign hyperplasia nodules: evaluation of discriminant criteria at multiparametric MRI. Clin Radiol 2013; 68: e323-30.
10.
Hoeks CM, Hambrock T, Yakar D, et al. Transition zone prostate cancer: detection and localization with 3-T multiparametric MR imaging. Radiology 2013; 266: 207-17.
11.
Pellegrino F, Stabile A, Sorce G, et al. Added value of prostate-specific antigen density in selecting prostate biopsy candidates among men with elevated prostate-specific antigen and PI-RADS >/=3 lesions on multiparametric magnetic resonance imaging of the prostate: a systematic assessment by PI-RADS Score. Eur Urol Focus 2024; 10: 634-40.
12.
Arafa MA, Farhat KH, Rabah DM, et al. Prostate-specific antigen density as a proxy for predicting prostate cancer severity: is there any difference between systematic and targeted biopsy? Saudi J Med Med Sci 2023; 11: 299-304.
13.
Lin S, Jiang W, Ding J, et al. Risk factor analysis and optimal cutoff value selection of PSAD for diagnosing clinically significant prostate cancer in patients with negative mpMRI: results from a high-volume center in Southeast China. World J Surg Oncol 2024; 22: 140.
14.
Yusim I, Krenawi M, Mazor E, et al. The use of prostate specific antigen density to predict clinically significant prostate cancer. Sci Rep 2020; 10: 20015.
15.
Schoots IG, Padhani AR. Risk-adapted biopsy decision based on prostate magnetic resonance imaging and prostate-specific antigen density for enhanced biopsy avoidance in first prostate cancer diagnostic evaluation. BJU Int 2021; 127: 175-8.
16.
Sakai I, Harada K, Kurahashi T, et al. Analysis of differences in clinicopathological features between prostate cancers located in the transition and peripheral zones. Int J Urol 2006; 13: 368-72.
17.
Bo M, Ventura M, Marinello R, et al. Relationship between prostatic specific antigen (PSA) and volume of the prostate in the benign prostatic hyperplasia in the elderly. Crit Rev Oncol Hematol 2003; 47: 207-11.
18.
Park DH, Yu JH. Prostate-specific antigen density as the best predictor of low- to intermediate-risk prostate cancer: a cohort study. Transl Cancer Res 2023; 12: 502-14.
19.
Wen J, Liu W, Shen X, et al. PI-RADS v2.1 and PSAD for the prediction of clinically significant prostate cancer among patients with PSA levels of 4-10 ng/ml. Sci Rep 2024; 14: 6570.
20.
Liu X, Xiong Q, Zeng W, et al. Comparison of the utility of PI-RADS 2.1, ADC values, and combined use of both, for the diagnosis of transition zone prostate cancers. J Comput Assist Tomogr 2024; 48: 206-11.
21.
Wei C, Pan P, Chen T, et al. A nomogram based on PI-RADS v2.1 and clinical indicators for predicting clinically significant prostate cancer in the transition zone. Transl Androl Urol 2021; 10: 2435-46.
22.
Cornford P, van den Bergh R, Briers E, et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on prostate cancer. Part II-2020 update: treatment of relapsing and metastatic prostate cancer. Eur Urol 2021; 79: 263-82.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.