Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
Editor's Choice
CARDIOLOGY / CLINICAL RESEARCH
Trehalose-induced alterations in serum expression levels of microRNAs associated with vascular inflammation in patients with coronary artery disease: pilot results from a randomized controlled trial
1
Department of Medical Biotechnology and Nanotechnology, Mashhad University of Medical Sciences, Mashhad, Iran
2
Cardiovascular Inflammation and Redox Biology laboratory, Baker Heart and Diabetes Institute, Melbourne, Victoria, Australia
3
Pharmaceutical Research Center, Pharmaceutical Technology Institute, Mashhad University of Medical Sciences, Mashhad, Iran
4
Immunology of Infectious Diseases Research Center, Research Institute of Basic Medical Sciences, Rafsanjan University of Medical Sciences, Rafsanjan, Iran
5
Department of Internal Medicine, Ali-Ibn Abi-Talib Hospital, School of Medicine, Rafsanjan University of Medical Sciences, Rafsanjan, Iran
6
Department of Traditional Pharmacy, School of Pharmacy, Mashhad University of Medical Sciences, Mashhad, Iran
7
Targeted Drug Delivery Research Center, Pharmaceutical Technology Institute, Mashhad University of Medical Sciences, Mashhad, Iran
8
Medical Toxicology Research Center, Pharmaceutical Technology Institute, Mashhad University of Medical Sciences, Mashhad, Iran
9
Research Department, Royal College of Surgeons in Ireland, Bahrain, Adliya, Bahrain
10
Department of Preventive Cardiology and Lipidology, Medical University of Lodz (MUL), Lodz, Poland
11
Cardiovascular Research Centre, University of Zielona Gora, Poland
12
Biotechnology Research Center, Pharmaceutical Technology Institute, Mashhad University of Medical Sciences, Mashhad, Iran
13
Centre for Research Impact & Outcome, Chitkara College of Pharmacy, Chitkara University, Rajpura, Punjab, India
14
Applied Biomedical Research Center, Basic Sciences Research Institute, Mashhad University of Medical Sciences, Mashhad, Iran
Submission date: 2022-08-08
Acceptance date: 2022-09-28
Online publication date: 2022-09-28
Corresponding author
Amirhossein Sahebkar
Biotechnology Research Center Pharmaceutical Technology Institute School of Medicine School of Pharmacy Mashhad University of Medical Sciences Mashhad, Iran
Biotechnology Research Center Pharmaceutical Technology Institute School of Medicine School of Pharmacy Mashhad University of Medical Sciences Mashhad, Iran
KEYWORDS
TOPICS
ABSTRACT
Introduction:
This study aimed to investigate the trehalose-induced alterations in serum expression levels of miRNAs associated with vascular inflammation in patients with coronary artery disease (CAD) in order to evaluate the effectiveness of intravenous (IV) trehalose administration in reducing arterial wall inflammation.
Material and methods:
This trial enrolled 14 men with a history of myocardial infarction (MI) and systemic inflammation. The patients were randomized in a 2 : 1 ratio to trehalose (15 g/week, IV administration) (N = 10) or placebo (equal volume 0.9% normal saline) (N = 4) for a period of 12 weeks. The relative serum expression levels of miRNA-126, miRNA-24, miRNA-181b, miRNA-10a, and miRNA-92a were assessed.
Results:
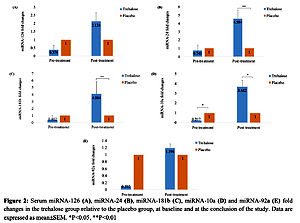
IV trehalose administration significantly increased the serum level of miRNA-24 (2.473 ±0.72; p = 0.037) compared to the baseline but did not alter the other miRNA serum levels. However, at the end of the study, miRNA-24 (4.58 ±0.99; p = 0.002), miRNA-181b (4.08 ±1.75; p = 0.009), and miRNA-10a (3.68 ±0.63; p = 0.013) showed notably higher serum levels in the trehalose relative to the placebo group. Furthermore, the reductions (normalized to baseline) in serum levels of miRNA-126 (p = 0.042) and miRNA-92a (p = 0.001) were smaller in the trehalose versus placebo group, while the serum level of miRNA-24 (p = 0.007) was notably higher than that in the placebo group.
Conclusions:
Serum levels of miRNAs associated with vascular inflammation were altered following IV trehalose administration. The alterations in serum miRNAs, especially miRNA-126 and miRNA-24, could be considered as helpful biomarkers for the evaluation of trehalose potency in reducing arterial wall inflammation in patients with CAD.
This study aimed to investigate the trehalose-induced alterations in serum expression levels of miRNAs associated with vascular inflammation in patients with coronary artery disease (CAD) in order to evaluate the effectiveness of intravenous (IV) trehalose administration in reducing arterial wall inflammation.
Material and methods:
This trial enrolled 14 men with a history of myocardial infarction (MI) and systemic inflammation. The patients were randomized in a 2 : 1 ratio to trehalose (15 g/week, IV administration) (N = 10) or placebo (equal volume 0.9% normal saline) (N = 4) for a period of 12 weeks. The relative serum expression levels of miRNA-126, miRNA-24, miRNA-181b, miRNA-10a, and miRNA-92a were assessed.
Results:
IV trehalose administration significantly increased the serum level of miRNA-24 (2.473 ±0.72; p = 0.037) compared to the baseline but did not alter the other miRNA serum levels. However, at the end of the study, miRNA-24 (4.58 ±0.99; p = 0.002), miRNA-181b (4.08 ±1.75; p = 0.009), and miRNA-10a (3.68 ±0.63; p = 0.013) showed notably higher serum levels in the trehalose relative to the placebo group. Furthermore, the reductions (normalized to baseline) in serum levels of miRNA-126 (p = 0.042) and miRNA-92a (p = 0.001) were smaller in the trehalose versus placebo group, while the serum level of miRNA-24 (p = 0.007) was notably higher than that in the placebo group.
Conclusions:
Serum levels of miRNAs associated with vascular inflammation were altered following IV trehalose administration. The alterations in serum miRNAs, especially miRNA-126 and miRNA-24, could be considered as helpful biomarkers for the evaluation of trehalose potency in reducing arterial wall inflammation in patients with CAD.
REFERENCES (68)
1.
Mc Namara K, Alzubaidi H, Jackson JK. Cardiovascular disease as a leading cause of death: how are pharmacists getting involved? Integr Pharm Res Pract 2019; 8: 1-11.
3.
Stein EA, Raal FJ. Lipid-lowering drug therapy for CVD prevention: looking into the future. Curr Cardiol Rep 2015; 17: 104.
4.
Libby P. Inflammation in atherosclerosis. Arterioscler Thromb Vasc Biol 2012; 32: 2045-51.
5.
Pothineni NVK, Subramany S, Kuriakose K, et al. Infections, atherosclerosis, and coronary heart disease. Eur Heart J 2017; 38: 3195-201.
6.
Bao MH, Lv QL, Li HG, et al. A novel putative role of TNK1 in atherosclerotic inflammation implicating the Tyk2/STAT1 pathway. Mediators Inflamm 2020: 6268514.
7.
Ballantyne C, Arroll B, Shepherd J. Lipids and CVD management: towards a global consensus. Eur Heart J 2005; 26: 2224-31.
8.
Ridker PM, Rifai N, Pfeffer MA, et al. Inflammation, pravastatin, and the risk of coronary events after myocardial infarction in patients with average cholesterol levels. Circulation 1998; 98: 839-44.
9.
Ridker PM, Rifai N, Clearfield M, et al. Measurement of C-reactive protein for the targeting of statin therapy in the primary prevention of acute coronary events. N Engl J Med 2001; 344: 1959-65.
10.
Cohen B, Singh D. C-reactive protein levels and outcomes after statin therapy. N Engl J Med 2005; 352: 1603-5.
11.
Elbein AD, Pan YT, Pastuszak I, Carroll D. New insights on trehalose: a multifunctional molecule. Glycobiology 2003; 13: 17R-27R.
12.
Jain NK, Roy I. Effect of trehalose on protein structure. Protein Sci 2009; 18: 24-36.
13.
Tanaka M, Machida Y, Niu S, et al. Trehalose alleviates polyglutamine-mediated pathology in a mouse model of Huntington disease. Nat Med 2004; 10: 148-54.
14.
Rodríguez-Navarro JA, Rodríguez L, Casarejos MJ, et al. Trehalose ameliorates dopaminergic and tau pathology in parkin deleted/tau overexpressing mice through autophagy activation. Neurobiol Dis 2010; 39: 423-38.
15.
Castillo K, Nassif M, Valenzuela V, et al. Trehalose delays the progression of amyotrophic lateral sclerosis by enhancing autophagy in motoneurons. Autophagy 2013; 9: 1308-20.
16.
Sergin I, Evans TD, Zhang X, et al. Exploiting macrophage autophagy-lysosomal biogenesis as a therapy for atherosclerosis. Nat Commun 2017; 8: 15750.
17.
Forouzanfar F, Guest PC, Jamialahmadi T, Sahebkar A. Hepatoprotective effect of trehalose: insight into its mechanisms of action. Adv Exp Med Biol 2021; 1328: 489-500.
18.
Khalifeh M, Barreto G, Sahebkar A. Therapeutic potential of trehalose in neurodegenerative diseases: the knowns and unknowns. Neural Regen Res 2021; 16: 2026-7.
19.
Khalifeh M, Barreto GE, Sahebkar A. Trehalose as a promising therapeutic candidate for the treatment of Parkinson’s disease. Br J Pharmacol 2019; 176: 1173-89.
20.
Khalifeh M, Read MI, Barreto GE, Sahebkar A. Trehalose against Alzheimer’s disease: insights into a potential therapy. BioEssays 2020; 42: 1900195.
21.
Sahebkar A, Hatamipour M, Tabatabaei SA. Trehalose administration attenuates atherosclerosis in rabbits fed a high-fat diet. J Cell Biochem 2019; 120: 9455-9.
22.
Yaribeygi H, Yaribeygi A, Sathyapalan T, Sahebkar A. Molecular mechanisms of trehalose in modulating glucose homeostasis in diabetes. Diabetes Metab Syndr Clin Res Rev 2019; 13: 2214-8.
23.
Minutoli L, Altavilla D, Bitto A, et al. Trehalose: a biophysics approach to modulate the inflammatory response during endotoxic shock. Eur J Pharmacol 2008; 589: 272-80.
24.
Zhang Y, Higgins CB, Mayer AL, et al. TFEB-dependent induction of thermogenesis by the hepatocyte SLC2A inhibitor trehalose. Autophagy 2018; 14: 1959-75.
25.
Wang Q, Ren J. mTOR-Independent autophagy inducer trehalose rescues against insulin resistance-induced myocardial contractile anomalies: role of p38 MAPK and Foxo1. Pharmacol Res 2016; 111: 357-73.
26.
Evans TD, Jeong SJ, Zhang X, Sergin I, Razani B. TFEB and trehalose drive the macrophage autophagy-lysosome system to protect against atherosclerosis. Autophagy 2018; 14: 724-6.
27.
Hosseinpour-Moghaddam K, Caraglia M, Sahebkar A. Autophagy induction by trehalose: molecular mechanisms and therapeutic impacts. J Cell Physiol 2018; 233: 6524-43.
28.
Germolec DR, Shipkowski KA, Frawley RP, Evans E. Markers of inflammation. Methods Mol Biol 2018; 1803: 57-79.
29.
Wang J, Chen J, Sen S. MicroRNA as biomarkers and diagnostics. J Cell Physiol 2016; 231: 25-30.
30.
Garo LP, Murugaiyan G. Contribution of MicroRNAs to autoimmune diseases. Cell Mol Life Sci 2016; 73: 2041-51.
31.
O’connell RM, Rao DS, Chaudhuri AA, Baltimore D. Physiological and pathological roles for microRNAs in the immune system. Nat Rev Immunol 2010; 10: 111-22.
32.
Fathullahzadeh S, Mirzaei H, Honardoost MA, Sahebkar A, Salehi M. Circulating microRNA-192 as a diagnostic biomarker in human chronic lymphocytic leukemia. Cancer Gene Ther 2016; 23: 327-32.
33.
Gorabi AM, Ghanbari M, Sathyapalan T, Jamialahmadi T, Sahebkar A. Implications of microRNAs in the pathogenesis of atherosclerosis and prospects for therapy. Curr Drug Targets 2021; 22: 1738-49.
34.
Gorabi AM, Kiaie N, Sathyapalan T, Al-Rasadi K, Jamialahmadi T, Sahebkar A. The role of microRNAs in regulating cytokines and growth factors in coronary artery disease: the ins and outs. J Immunol Res 2020; 2020: 5193036.
35.
Mahmoudi A, Butler AE, Jamialahmadi T, Sahebkar A. The role of exosomal miRNA in nonalcoholic fatty liver disease. J Cell Physiol 2022; 237: 2078-94.
36.
Mirzaei HR, Sahebkar A, Mohammadi M, et al. Circulating micrornas in hepatocellular carcinoma: potential diagnostic and prognostic biomarkers. Curr Pharm Des 2016; 22: 5257-69.
37.
de Oliveira ARCP, Castanhole-Nunes MMU, Biselli-Chicote PM, et al. Differential expression of angiogenesis-related miRNAs and VEGFA in cirrhosis and hepatocellular carcinoma. Arch Med Sci 2020; 16: 1150-7.
38.
Li H, Liu D, Liu L, Huang S, Ma A, Zhang X. The role of HOTAIR/miR-152-3p/LIN28B in regulating the progression of endometrial squamous carcinoma. Arch Med Sci 2021; 17: 434-48.
39.
Li W, Wang S, Xu J, et al. Inferring latent microRNA-disease associations on a gene-mediated tripartite heterogeneous multiplexing metwork. IEEE/ACM Trans Comput Biol Bioinform 2022; 19: 3190-201. doi: 10.1109/TCBB.2022.3143770.
40.
Churov A, Summerhill V, Grechko A, Orekhova V, Orekhov A. MicroRNAs as potential biomarkers in atherosclerosis. Int J Mol Sci 2019; 20: 5547.
41.
Widmer RJ, Chung WY, Herrmann J, Jordan KL, Lerman LO, Lerman A. The association between circulating microRNA levels and coronary endothelial function. PLoS One 2014; 9: e109650.
42.
Parahuleva MS, Lipps C, Parviz B, et al. MicroRNA expression profile of human advanced coronary atherosclerotic plaques. Sci Rep 2018; 8: 1-9.
43.
Fang Y, Davies PF. Site-specific microRNA-92a regulation of Krüppel-like factors 4 and 2 in atherosusceptible endothelium. Arterioscler Thromb Vasc Biol 2012; 32: 979-87.
44.
Loyer X, Potteaux S, Vion AC, et al. Inhibition of microRNA-92a prevents endothelial dysfunction and atherosclerosis in mice. Circ Res 2014; 114: 434-43.
45.
Harris TA, Yamakuchi M, Ferlito M, Mendell JT, Lowenstein CJ. MicroRNA-126 regulates endothelial expression of vascular cell adhesion molecule 1. Proc Natl Acad Sci 2008; 105: 1516-21.
46.
Feinberg MW, Moore KJ. MicroRNA regulation of atherosclerosis. Circ Res 2016; 118: 703-20.
47.
Sun X, Icli B, Wara AK, et al. MicroRNA-181b regulates NF-B–mediated vascular inflammation. J Clin Investig 2012; 122: 1973-90.
48.
Sun X, Sit A, Feinberg MW. Role of miR-181 family in regulating vascular inflammation and immunity. Trends Cardiovasc Med 2014; 24: 105-12.
49.
Fang Y, Shi C, Manduchi E, Civelek M, Davies PF. MicroRNA-10a regulation of proinflammatory phenotype in athero-susceptible endothelium in vivo and in vitro. Proc Natl Acad Sci 2010; 107: 13450-5.
50.
Echigo R, Shimohata N, Karatsu K, et al. Trehalose treatment suppresses inflammation, oxidative stress, and vasospasm induced by experimental subarachnoid hemorrhage. J Transl Med 2012; 10: 80.
51.
Sahebkar A, Hatamipour M, Tabatabaei SA. Trehalose administration attenuates atherosclerosis in rabbits fed a high-fat diet. J Cell Biochem 2019; 120: 9455-9459.
52.
Kaplon RE, Hill SD, Bispham NZ, et al. Oral trehalose supplementation improves resistance artery endothelial function in healthy middle-aged and older adults. Aging 2016; 8: 1167.
53.
Zhou Q, Gallagher R, Ufret-Vincenty R, Li X, Olson EN, Wang S. Regulation of angiogenesis and choroidal neovascularization by members of microRNA-23 27 24 clusters. Proc Natl Acad Sci 2011; 108: 8287-92.
54.
Fiedler J, Jazbutyte V, Kirchmaier BC, et al. MicroRNA-24 regulates vascularity after myocardial infarction. Circulation 2011; 124: 720-30.
55.
Wang J, Huang W, Xu R, et al. Micro RNA-24 regulates cardiac fibrosis after myocardial infarction. J Cell Mol Med 2012; 16: 2150-60.
56.
Rathcke CN, Vestergaard H. YKL-40-an emerging biomarker in cardiovascular disease and diabetes. Cardiovasc Diabetol 2009; 8: 61.
57.
Deng X, Liu Y, Luo M, et al. Circulating miRNA-24 and its target YKL-40 as potential biomarkers in patients with coronary heart disease and type 2 diabetes mellitus. Oncotarget 2017; 8: 63038-46.
58.
Fichtlscherer S, De Rosa S, Fox H, et al. Circulating microRNAs in patients with coronary artery disease. Circ Res 2010; 107: 677-84.
59.
Wang W, Li Z, Zheng Y, Yan M, Cui Y, Jiang J. Circulating microRNA-92a level predicts acute coronary syndrome in diabetic patients with coronary heart disease. Lipids Health Dis 2019; 18: 22.
60.
Liu Y, Li Q, Hosen MR, et al. Atherosclerotic conditions promote the packaging of functional microRNA-92a-3p into endothelial microvesicles. Circ Res 2019; 124: 575-87.
61.
Liu G, Li Y, Gao XG. microRNA-181a is upregulated in human atherosclerosis plaques and involves in the oxidative stress-induced endothelial cell dysfunction through direct targeting Bcl-2. Eur Rev Med Pharmacol Sci 2016; 20: 3092-100.
62.
Weber M, Baker MB, Patel RS, Quyyumi AA, Bao G, Searles CD. MicroRNA expression profile in CAD patients and the impact of ACEI/ARB. Cardiol Res Pract 2011; 2011: 532915.
63.
Kuo JT, Tsai HE, Lin CT, et al. Low levels of microRNA-10a in cardiovascular endothelium and blood serum are related to human atherosclerotic disease. Cardiol Res Pract 2021; 2021: 1452917.
64.
Fras Z, Tršan J, Banach M. On the present and future role of Lp-PLA2 in atherosclerosis-related cardiovascular risk prediction and management. Arch Med Sci 2020; 17: 954-64.
65.
Pirro M, Simental-Mendía LE, Bianconi V, Watts GF, Banach M, Sahebkar A. Effect of statin therapy on arterial wall inflammation based on 18F-FDG PET/CT: a systematic review and meta-analysis of interventional studies. J Clin Med 2019; 8: 118.
66.
Khalifeh M, Penson PE, Banach M, Sahebkar A. Statins as anti-pyroptotic agents. Arch Med Sci 2021; 17: 1414-7.
67.
Banach M, Burchardt P, Chlebus K, et al. PoLA/CFPiP/PCS/PSLD/PSD/PSH guidelines on diagnosis and therapy of lipid disorders in Poland 2021. Arch Med Sci 2021; 17: 1447-547.
68.
Jamialahmadi T, Emami F, Bagheri RK, et al. The effect of trehalose administration on vascular inflammation in patients with coronary artery disease. Biomed Pharmacother 2022; 147: 112632.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.