Introduction
Since severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was first discovered in Wuhan in December 2019, coronavirus disease-2019 (COVID-19) has spread worldwide. China has made great efforts to control this epidemic. Although more than 12 months has passed since the outbreak of COVID-19, the situation in some countries is not optimistic. A face mask is one of the key tools to protect medical staff in hospitals and clinics and plays an important role in reducing the spread of COVID-19 [1]. Efforts to fight COVID-19 are dual in nature and include supply of vaccines and/or effective drugs to the market to prevent infection. Unrealistic optimism/pessimism may be a key factor in the eradication of COVID-19 [2].
The psychological problems caused by COVID-19 infection cannot be ignored. A study reported that 10.8% of patients experience severe anxiety and depression and need psychological intervention to reduce psychological trauma [3]. Although we have gained some experience in preventing and treating COVID-19, our knowledge and research on COVID-19 are still limited because it is difficult to directly detect airborne viruses [4].
There is a correlation between the incidence of COVID-19 in children and adults that increases with the age of children. However, we currently underestimate the number of pediatric cases [5]. Indeed, elderly patients infected with COVID-19 have relatively severe clinical manifestations due to complications, while young patients with COVID-19 have mild symptoms and the number of critical cases is small. The specific reasons are not clear. COVID-19 may use angiotensin-converting enzyme 2 (ACE2) as a receptor to initiate infection, leading to severe complications from the heart, lungs, and gastrointestinal tract [6].
Yongchuan Hospital Affiliated to Chongqing Medical University is the designated hospital for COVID-19 patients in western Chongqing. As of 14 March 2020, a total of 90 patients had been admitted to the COVID-19 isolation ward. The clinical presentations and symptoms of COVID-19 vary in frequency and severity, and the symptoms differ with the severity of disease. The age-dependent susceptibility to COVID-19 (children are less likely to be infected than adults) results in a reduced number of cases in children. Kuchar et al. [7] reported that SARS-CoV-2 infections in children exhibiting COVID-19 symptoms and requiring hospitalization are significantly lower than in hospitalized adults. Indeed, the immune cross-protection of other coronaviruses may lead to reduced sensitivity [8, 9].
This study retrospectively analyzed the general conditions, clinical symptoms, laboratory test results, imaging findings, and treatment of the 90 patients from different age groups and examined the differences and regularities between young patients and middle-aged and elderly patients. We sought to establish valuable clinical evidence for the prevention and control of COVID-19.
Material and methods
Patient data collection
Yongchuan Hospital is a major tertiary teaching hospital. Yongchuan Hospital has an Infectious Diseases Department, is affiliated with Chongqing Medical University, and is located in Yongchuan district, Chongqing City. Yongchuan Hospital is responsible for treating patients in eight regions and counties from Chongqing City. This study included 90 COVID-19 patients admitted to the hospital during the period from 21 January to 4 March 2020. All of the patients were diagnosed with COVID-19 at the time of admission by RT-PCR. The severity of disease was classified as mild or severe according to the Pneumonitis Diagnosis and Treatment Scheme for Novel Coronavirus Infection (Trial Version 6) [10].
Research methods
We collected data from all patients, including epidemiologic data, clinical manifestations, laboratory test results, imaging findings, and outcome. The patients were evaluated 6 months after hospital discharge. Epidemiologic data were consolidated from case studies and epidemiologic surveys were conducted by the local Centers for Disease Control and Prevention (CDC). The inclusion criteria were as follows: 1. fever, cough, and a history of close contact with other patients. 2. throat or upper respiratory tract secretions shown to be positive for SARS-COV-2 by RT-PCR. 3. influenza A and B, common respiratory virus, SARS-CoV, and MERS-CoV infections were ruled out.
Blood counts and biochemical tests were completed in the Department of Clinical Laboratory of our hospital and low-dose spiral computed tomography (CT) examinations of the chest was performed in the Radiology Department. All data were checked by two executors. In this retrospective study all data were shared with the Chongqing Municipal Health Commission. The patients ranged in age from 3 to 89 years, with an average age of 48 ±18 years. According to the age interquartile range (IQR), the patients were divided into 4 groups: youth (< 39 years), middle-age (39–48 years), middle-elderly (49–60 years), and elderly (> 60 years). The differences in clinical symptoms, laboratory test results, imaging findings, and treatment effects were compared among the four groups.
Observation items
Clinical symptoms, such as fever, non-productive and productive cough, fatigue, shortness of breath, myalgias, and diarrhea, and the laboratory test results, including routine blood tests, C-reaction protein (CRP) level, interleukin-6 (IL-6) level, blood coagulation profile, and lymphocyte subset counts, were collected. Pneumonia was diagnosed based on a low-dose 256 detector row CT of the chest and examined by two radiologists. The clinical discharge criteria for patients were based on the COVID-19 diagnosis and treatment protocol (trial version 6), as follows: 1) body temperature returned to normal for at least 3 days. 2) clinical symptoms of the respiratory tract were significantly improved. 3) re-examination of pulmonary imaging showed significant improvement of acute exudative lesions. 4) two consecutive negative nucleic acid tests of respiratory tract specimens (at least 1 day between sampling intervals) [10].
Statistical analysis
SPSS 26.0 statistical software was used for statistical analysis of the results, and a p < 0.05 was considered statistically significant. Measurement data were expressed as the mean ± standard deviation (SD). The Student-Newman-Keuls (SNK) method was used for intergroup comparisons following the one-way ANOVA analysis. Enumeration data were analyzed using the χ2 test.
Results
General information
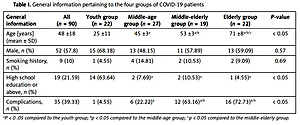
There were 22 (24.4%), 27 (30.0%), 19 (21.2%), and 22 patients (24.4%) in the youth, middle-age, middle-elderly, and elderly groups, respectively. The youth group accounted for 63.64% (14/22) of the patients with a high school education or above, which was significantly different from the other 3 groups (p < 0.05). The incidence of complications in the youth group (4.55%) was significantly lower than in the other 3 groups. There was no significant difference in the distribution of gender or smoking history in the 4 groups (p > 0.05, Table I).
Table I
General information pertaining to the four groups of COVID-19 patients
| General information | All (n = 90) | Youth group (n = 22) | Middle-age group (n = 27) | Middle-elderly group (n = 19) | Elderly group (n = 22) | P-value |
|---|---|---|---|---|---|---|
| Age [years] (mean ± SD) | 48 ±18 | 25 ±11 | 45 ±3a | 53 ±3a/b | 71 ±8a/b/c | < 0.05 |
| Male, n (%) | 52 (57.8) | 15 (68.18) | 13 (48.15) | 11 (57.89) | 13 (59.09) | 0.57 |
| Smoking history, n (%) | 9 (10) | 1 (4.55) | 4 (14.81) | 2 (10.53) | 2 (9.09) | 0.69 |
| High school education or above, n (%) | 19 (21.59) | 14 (63.64) | 2 (7.69)a | 2 (10.53)a | 1 (4.55)a | < 0.05 |
| Complications, n (%) | 35 (39.33) | 1 (4.55) | 6 (22.22)a | 12 (63.16)a/b | 16 (72.73)a/b | < 0.05 |
Clinical symptoms
The main clinical manifestations of COVID-19 patients included fever, non-productive and productive cough, fatigue, shortness of breath, myalgias, diarrhea, headaches, pharyngeal neuralgia, chills, and anorexia, as well as other types of discomfort in a small number of patients. The proportion of main clinical symptoms did not differ significantly among the four age groups (p > 0.05, Table II).
Table II
Main clinical symptoms of COVID-19 patients in the four groups
Laboratory findings
On admission, 90 patients underwent hematologic, biochemical, and routine serology testing. There was no significant difference in the white blood cell count among the four groups (p > 0.05). Procalcitonin (PCT), an indicator of inflammation, did not differ significantly among the four groups (p > 0.05). Compared with the other three groups, the lymphocyte and CD4 + T counts of the youth group were significantly higher (p < 0.05), while the CRP and IL-6 levels were significantly lower (p < 0.05, Table III).
Table III
Laboratory findings for SARS-CoV-2-infected patients
| Laboratory test | All (n = 90) | Youth group (n = 22) | Middle-age group (n = 27) | Middle-elderly group (n = 19) | Elderly group (n = 22) |
|---|---|---|---|---|---|
| WBC [× 109/l] | 5.44 ±2.25 | 5.96 ±2.46 | 5.84 ±2.63 | 5.02 ±2.06 | 4.78 ±1.42 |
| PCT [ng/ml] | 0.09 ±0.14 | 0.11 ±0.23 | 0.10 ±0.16 | 0.09 ±0.06 | 0.08 ±0.05 |
| L [× 109/l] | 1.22 ±0.58 | 1.63 ±0.71 | 1.10 ±0.47a | 1.13 ±0.52a | 1.04 ±0.45a |
| Youth group (n = 22) | p = 0.005 | p = 0.026 | p = 0.003 | ||
| CRP [mmol/l] | 18.12 ±24.12 | 7.20 ±9.15 | 16.70 ±26.57 | 28.62 ±34.20a | 21.75 ±16.26 |
| Youth group (n = 22) | p = 0.026 | ||||
| IL-6 [pg/ml] | 13.51 ±17.72 | 6.35 ±7.58 | 10.32 ±15.86a | 14.51 ±15.10 | 23.73 ±24.28a/b |
| Elderly group (n = 22) | p = 0.005 | p = 0.040 | |||
| CD4+ T lymphocyte count [per µl] | 494.43 ±279.30 | 631.41 ±317.62 | 457.22 ±241.66 | 514.42 ±311.30 | 385.86 ±200.49a |
| Youth group (n = 22) | p = 0.020 |
CT examination
All patients had chest CT scans on admission, and the severity of acute lung exudative lesions was determined using the pneumonia severity index (PSI) [11]. Ninety percent (81/90) of the chest CT scans showed ground-glass opacities, patchy lesions, consolidation, cords, and network shadows. The number of patients without lesions in each group did not differ significantly among the four groups (p > 0.05). The PSI at the time of hospital admission and discharge in the middle-aged, middle-elderly, and elderly groups was significantly higher than in the youth group (p < 0.05, Table IV).
Table IV
CT results of 90 SARS-CoV-2-infected patients
| CT examination | All (n = 90) | Youth group (n = 22) | Middle-age group (n = 27) | Middle-elderly group (n = 19) | Elderly group (n = 22) | P-value |
|---|---|---|---|---|---|---|
| No. of lesions, n (%) | 9 (10.0) | 6 (27.27) | 0 (0) | 2 (10.53) | 1 (4.55) | 0.38 |
| PSI on admission | 32.44 ±21.74 | 8.13 ±4.73 | 24.12 ±17.59a | 36.25 ±15.47a | 52.50 ±21.15a/b | < 0.05 |
| PSI at discharge | 21.71 ±18.52 | 9.64 ±12.53 | 21.92 ±17.65a | 26.05 ±17.68a | 29.20 ±20.26a | 0.00 |
Treatment
Among the 90 patients, 77 (85.6%) were mild, 13 (14.4%) were severe, and the number of mild cases in the youth group accounted for 100%. All patients received antiviral treatment. For patients with bacterial pneumonia, we used piperacillin-tazobactam treatment through an intravenous drip (4.5 g every 8 h). For patients with gradual deterioration of the oxygenation index, rapid progression based on imaging findings, and excessive activation of the inflammatory response, methylprednisolone was administered at a dose of 1–2 mg/kg/day for 3–5 days. The antibiotic and hormone treatment plans were adjusted according to the patient’s condition. Among the 90 patients, 32 (35.6%) received antibiotic therapy, 25 (27.8%) received hormone (methylprednisolone) therapy, 13 (14.4%) received gamma globulin therapy, and 8 (8.9%) received ventilator-assisted ventilation (Table V). No patients in the youth group were mechanically ventilated, which was statistically different from the middle-aged and elderly groups (p < 0.05). There was no difference in the use of antibiotics, hormones, or gamma globulin between groups (p > 0.05).
Table V
Treatment scheme for a patient infected with COVID-19
| Treatment | All (n = 90) | Youth group (n = 22) | Middle-age group (n = 27) | Middle-elderly group (n = 19) | Elderly group (n = 22) | P-value |
|---|---|---|---|---|---|---|
| Antibiotics, n (%) | 32 (35.56) | 6 (27.27) | 8 (29.63) | 8 (42.11) | 10 (45.45) | 0.50 |
| Methylprednisolone therapy, n (%) | 25 (27.78) | 3 (13.64) | 6 (22.22) | 6 (31.58) | 10 (45.45) | 0.10 |
| Gamma globulin, n (%) | 13 (14.44) | 1 (4.55) | 5 (18.52) | 3 (15.79) | 4 (18.18) | 0.50 |
| Ventilator-assisted ventilation, n (%) | 8 (8.89) | 0 | 3 (11.11)a | 0 | 5 (22.73)a | 0.02 |
| Mild cases, n (%) | 77 (85.56) | 22 (100) | 23 (85.19)a | 17 (89.47)a | 15 (68.18)a | 0.02 |
Discussion
Adolescents and young adults are more often involved in social activities, communication and learning, and finding stable jobs, which increases susceptibility to the worldwide spread of COVID-19. Thus, public health officials should pay more attention to young patients infected with COVID-19. In the course of treatment, we found that the clinical manifestations of patients differed as a function of age. Young patients have mild symptoms and elderly patients have severe clinical manifestations, which are consistent with reports in the literature. Liao et al. [12] also found that the majority of young patients with COVID-19 infections were asymptomatic, rarely had critical conditions, and most had better prognosis and outcomes after treatment. The specific reasons for this were not further analyzed by the authors, and our study suggests that one of the possible reasons is the existence of complications of underlying diseases in middle-aged and elderly patients [13, 14]. To date, the specific causes of mild clinical manifestations in young patients have not been reported. For other respiratory viral infections, including SARS, age-dependent differences in severity have been observed. For COVID-19, there are strong indications that the reported cases have age-dependent severity and mortality rates, which may extend to the possibility of clinically reportable symptoms after infection [15–17].
In this study, the ages of the 90 patients were stratified by interquartile spacing, and there was no significant difference in the number of cases among the four groups with respect to age and gender. The clinical characteristics of patients infected with SARS-CoV-2 were different among the four age groups. We observed that the main clinical symptoms of the four groups were fever, non-productive and productive cough, fatigue, and shortness of breath. In fact, there was no significant difference in the number of patients with the main clinical symptoms between the youth group and the other three groups. However, we observed significant differences in the PSI at the time of hospital admission and discharge between the youth and other three groups, which was consistent with previous reports [18–20].
Nine retrospective studies (1332 cases) were included. The results demonstrated that the combined proportions of findings were as follows: ground-glass opacities (GGOs), 0.79 (95% CI: 0.68–0.89); consolidation, 0.34 (95% CI: 0.23–0.47), combined GGOs and consolidation, 0.46 (95% CI: 0.37–56); air bronchogram sign, 0.41 (95% CI: 0.26–0.55); crazy paving pattern, 0.32 (95% CI: 0.17–0.47); interlobular septal thickening, 0.55 (95% CI: 0.42–0.67); reticulation, 0.30 (95% CI: 0.12–0.48); bronchial wall thickening, 0.24 (95% CI: 0.11–0.40); vascular enlargement, 0.74 (95% CI: 0.64–0.86); subpleural linear opacities, 0.28 (95% CI: 0.12–0.48); intrathoracic lymph node enlargement, 0.03 (95% CI: 0.00–0.07); and pleural effusions, 0.03 (95% CI: 0.02–0.06).
The distribution was characterized as peripheral, bilateral, and > 2 lobes involved. Therefore, based on the above-mentioned chest CT features of COVID-19, chest CT should be considered a promising method for identifying COVID-19 [21].
The laboratory test findings showed significant changes in lymphocyte counts and CRP and IL-6 levels in the youth group, indicating that young patients were unlikely to develop severe disease [22, 23]. This finding was attributed to underlying health issues in the elderly population, such as more co-morbidities, immunosenescence, decreased immune function, poor nutritional state, and increased frailty. Specifically, diabetes, heart disease, and other chronic illnesses can lead to more severe symptoms and complications in patients infected with COVID-19. Additionally, as people age, the immune system gradually loses its resiliency; thus patients are more susceptible to infections of any kind, especially a novel pathogen like COVID-19, while young people with robust immune function are less susceptible to SARS-CoV-2 [24].
Diabetes patients with COVID-19 have the worst prognosis because diabetes is characterized by low-grade chronic inflammation caused by excessive visceral adipose tissue. Chronic inflammation combined with hyperglycemia can lead to abnormal and ineffective immune responses. In addition, diabetes often co-exists with hypertension, and treatment with ACE inhibitors or angiotensin receptor blockers (ARB) may have harmful effects on patients with diabetes and COVID-19 [25].
During aging, the immune system changes in two main ways: there is a gradual decline in immune function (immunosenescence), and another classic immune system change during aging is a chronic increase in systemic inflammation (inflammaging), which is caused by an overactive but ineffective alert system [26]. A large amount of the most recent data on the pathologic and molecular changes of COVID-19 patients shows that immunosenescence and inflammaging are the main driving forces for the high mortality rate in elderly patients [27]. However, a comparative study of patients with moderate and severe disease found that gender and age are not risk factors for severe illness, but rather a combination of underlying factors, such as respiratory rate, lymphocyte count, blood glucose, albumin, urea, CRP, and IL-6 levels, and imaging findings [28].
We observed that the number of severe cases in the younger group was very low. Indeed, none of the youth in the current study had respiratory failure or required a ventilator to assist with respiratory support. The clinical characteristics of young patients are mostly mild with the following two important characteristics: the youth group included highly educated people with fixed professional and social identities, and the group used news media, the internet, new media platforms, and other media to enable them to recognize and understand COVID-19 earlier than other age groups. For those patients who have permanent work or a designated place of study, the range of activities may have been restricted, thus making it difficult to maintain close contact with COVID-19 patients. Second, the CD4+ T lymphocyte counts of the youth group were significantly higher than the other three groups at the time of hospital admission, indicating that this group has good cellular immune function.
It has been reported that the antibody levels in patients with mild clinical manifestations undergo rapid regression [29]. Fighting against SARS-CoV-2 does not depend on how many antibodies there are or how long the antibodies exist in the human body, but on the ability to mobilize the immune system quickly and effectively.
The lymphocyte counts in COVID-19 patients gradually decrease with progression of the disease, but the mechanism underlying the massive decrease in lymphocyte count in patients with severe disease is unclear [30]. As the disease progresses, the absolute number of CD8 + T and NK cells is significantly reduced, but the cell sub-populations are highly activated. After the patient’s condition improves, the number of CD8 + T and NK cells and the immune status recover to a certain extent [31]. Helper T lymphocytes are responsible for coordinating the entire immune response process, including assisting B cells to produce antibodies and assisting NK cells to perform immune functions [32, 33]. Therefore, in young people, SARS-CoV-2 infection seems to be effectively controlled by good cellular immune function, thus manifesting as asymptomatic or mildly symptomatic infections. However, in order to further confirm these findings, it is necessary for future research to compare the severity of illness of COVID-19 patients with low education in the same age group.
In conclusion, among young patients, a higher level of education, higher awareness, and a high degree of acceptance of COVID-19 epidemic prevention and control may reduce the chance of exposure to SARS-CoV-2. Moreover, the youth group has good immune function, and can effectively control the virus. Thus, young patients infected with SARS-CoV-2 showed mild clinical manifestations. Therefore, we should not only strengthen the publicity and education about COVID-19, but also strengthen our physical fitness and design and develop a vaccine that can fully stimulate the immune system to control SARS-CoV-2 infection.