Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
CARDIO-ONCOLOGY / CLINICAL RESEARCH
Does trastuzumab-related cardiotoxicity influence
long-term outcome in patients with HER-2 positive
breast cancer? A prospective study
1
Cardiology Department, Nicolaus Copernicus Memorial Hospital, Lodz, Poland
2
Department of Cardiooncology, Medical University, Lodz
3
Department of Physiology, Development and Neuroscience, University of Cambridge, Cambridge, United Kingdom
4
Department of Chemical Engineering and Biotechnology, University of Cambridge, Cambridge, United Kingdom
5
Department of Hypertension, Medical University of Lodz, Lodz, Poland
Submission date: 2020-04-29
Final revision date: 2020-05-24
Acceptance date: 2020-05-25
Online publication date: 2021-03-16
Publication date: 2026-04-30
Corresponding author
Grzegorz Piotrowski
Department of Cardiology Nicolaus Copernicus Memorial Hospital 62 Pabianicka St. 91-513 Łódź, Poland
Department of Cardiology Nicolaus Copernicus Memorial Hospital 62 Pabianicka St. 91-513 Łódź, Poland
Arch Med Sci 2026;22(2):785-795
KEYWORDS
cardiotoxicitydisease-free survivaloverall survivalbreast cancer adjuvant treatmentnon-metastatic breast cancerbreast cancer prognosis
TOPICS
ABSTRACT
Introduction:
Trastuzumab is a monoclonal antibody directed against the HER-2 receptor that has led, in an adjuvant setting, to higher disease-free (DFS) and overall survival (OS) in HER-2 positive breast cancer (BC) compared with chemotherapy alone. Cardiotoxicity often results in early discontinuation of trastuzumab, which may elevate the risk of cancer recurrence or mortality. Our study aimed to assess how early interruption or early permanent termination of adjuvant trastuzumab treatment influences DFS and OS of patients with HER-2 positive BC.
Material and methods:
This is a prospective observation of 253 women (55 ±10 years of age) with HER-2 positive unilateral, non-metastatic BC treated with trastuzumab in an adjuvant setting. To monitor the safety of the treatment echocardiography was performed at baseline and every 3 months up to 12 months after the end of therapy. If cardiotoxicity developed, trastuzumab was stopped early. Overall survival and DFS were assessed.
Results:
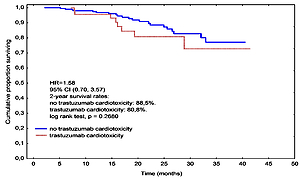
Trastuzumab-associated cardiac complications resulting in treatment discontinuation developed in 52 (20.55%) patients. Median DFS time was 21.1 months in the group with interruption compared with 25.7 months in the group with full trastuzumab treatment, being significantly shorter (HR = 2.32, 95% CI: 1.15–4.71, p = 0.0106). Two year OS in the interruption and no-interruption groups were 80.8% and 88.5%, respectively, which were not statistically significantly different (p = 0.268). In a multivariate regression analysis the cumulative dose of anthracycline (OR = 1.01, 95% CI: 1.00–1.01, p = 0.002) and LVEF at baseline (OR = 0.83, 95% CI: 0.70–0.99, p = 0.0344) were independent predictors of a cardiotoxic effect.
Conclusions:
Trastuzumab-related cardiotoxicity resulting in early treatment discontinuation negatively influences DFS, but does not seem to influence OS.
Trastuzumab is a monoclonal antibody directed against the HER-2 receptor that has led, in an adjuvant setting, to higher disease-free (DFS) and overall survival (OS) in HER-2 positive breast cancer (BC) compared with chemotherapy alone. Cardiotoxicity often results in early discontinuation of trastuzumab, which may elevate the risk of cancer recurrence or mortality. Our study aimed to assess how early interruption or early permanent termination of adjuvant trastuzumab treatment influences DFS and OS of patients with HER-2 positive BC.
Material and methods:
This is a prospective observation of 253 women (55 ±10 years of age) with HER-2 positive unilateral, non-metastatic BC treated with trastuzumab in an adjuvant setting. To monitor the safety of the treatment echocardiography was performed at baseline and every 3 months up to 12 months after the end of therapy. If cardiotoxicity developed, trastuzumab was stopped early. Overall survival and DFS were assessed.
Results:
Trastuzumab-associated cardiac complications resulting in treatment discontinuation developed in 52 (20.55%) patients. Median DFS time was 21.1 months in the group with interruption compared with 25.7 months in the group with full trastuzumab treatment, being significantly shorter (HR = 2.32, 95% CI: 1.15–4.71, p = 0.0106). Two year OS in the interruption and no-interruption groups were 80.8% and 88.5%, respectively, which were not statistically significantly different (p = 0.268). In a multivariate regression analysis the cumulative dose of anthracycline (OR = 1.01, 95% CI: 1.00–1.01, p = 0.002) and LVEF at baseline (OR = 0.83, 95% CI: 0.70–0.99, p = 0.0344) were independent predictors of a cardiotoxic effect.
Conclusions:
Trastuzumab-related cardiotoxicity resulting in early treatment discontinuation negatively influences DFS, but does not seem to influence OS.
REFERENCES (44)
1.
Slamon DJ, Godolphin W, Jones LA, et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 1989; 244: 707-12.
2.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987; 235: 177-82.
3.
Slamon D, Eiermann W, Robert N, et al. On behalf of the Breast Cancer International Research Group. Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med 2011; 365: 1273-83.
4.
Moja L, Tagliabue L, Balduzzi S, et al. Trastuzumab containing regimens for early breast cancer. Cochrane Database Syst Rev 2012; 4: CD006243.
5.
de Azambuja E, Procter MJ, van Veldhuisen DJ, et al. Trastuzumab-associated cardiac events at 8 years of median follow-up in the Herceptin Adjuvant trial (BIG1-01). J Clin Oncol 2014; 32: 2159-65.
6.
Perez EA, Suman VJ, Davidson NE, et al. Cardiac safety analysis of doxorubicin and cyclophosphamide followed by paclitaxel with or without trastuzumab in the North Central Cancer Treatment Group N9831 adjuvant breast cancer trial. J Clin Oncol 2008; 26: 1231-8.
7.
Romond EH, Jeong JH, Rastogi P, et al. Seven-year follow-up assessment of cardiac function in NSABP B-31, a randomized trial comparing doxorubicin and cyclophosphamide followed by paclitaxel (ACP) with ACP plus trastuzumab as adjuvant therapy for patients with node-positive, human epidermal growth factor receptor 2-positive breast cancer. J Clin Oncol 2012; 30: 3792-9.
8.
Tan-Chiu E, Yothers G, Romond E, et al. Assessment of cardiac dysfunction in a randomized trial comparing doxorubicin and cyclophosphamide followed by paclitaxel, with or without trastuzumab as adjuvant therapy in node-positive, human epidermal growth factor receptor 2-overexpressingbreast cancer: NSABP B-31. J Clin Oncol 2005; 23: 7811-9.
9.
Piccart-Gebhart MJ, Procter M, Leyland-Jones B, et al. Trastuzumab after adjuvant chemotherapy in HER2-positivebreast cancer. N Engl J Med 2005; 353: 1659-72.
10.
Tolaney SM, Barry WT, Dang CT, et al. Adjuvant paclitaxel and trastuzumab for node-negative, HER2-positivebreast cancer. N Engl J Med 2015; 372: 134-41.
11.
Jones SE, Collea R, Paul D, et al. Adjuvant docetaxel and cyclophosphamide plus trastuzumab inpatients with HER2-amplified early stage breast cancer: a single group, open-label, phase 2 study. Lancet Oncol 2013; 14: 1121-8.
12.
Telli ML, Hunt SA, Carlson RW, Guardino AE. Trastuzumab-related cardiotoxicity: calling into question the concept of reversibility. J Clin Oncol 2007; 25: 3525-33.
13.
Bowles EJ, Wellman R, Feigelson HS, et al.; Pharmacovigilance Study Team. Risk of heart failure in breast cancer patients after anthracycline and trastuzumab treatment: a retrospective cohort study. J Natl Cancer Inst 2012; 104: 1293-305.
14.
Banke A, Fosbøl EL, Ewertz M, et al. Long-term risk of heart failure in breast cancer patients after adjuvant chemotherapy with or without trastuzumab. JACC Heart Fail 2019; 7: 217-24.
15.
Barron CC, Tyagi NK, Alhussein MM, et al. Adjuvant trastuzumab therapy: can we balance efficacy and safety? Oncologist 2019; 24: 1405-9.
16.
Mackey JR, Clemons M, Cote MA, et al. Cardiac management adjuvant trastuzumab therapy: recommendation of the Canadian Working Group. Current Oncol 2008; 15: 24-35.
17.
Jones AL, Barlow M, Barrett-Lee PJ, et al. Management of cardiac health in trastuzumab treated patients with breast cancer: updated United Kingdom National Cancer Research Institute recommendations for monitoring. Br J Cancer 2009; 100: 684-92.
18.
Swedberg K, Cleland J, Dargie H, et al. Guidelines for the diagnosis and treatment of chronic heart failure: full text (update 2005). The task force for the diagnosis and treatment of CHF of the European Society of Cardiology. Eur Heart J 2005; 26: 1115-40.
19.
Douglas PS, DeCara JM, Devereux RB, et al. Echocardiographic imaging in clinical trials: American Society of Echocardiography Standards for echocardiography core laboratories: endorsed by the American College of Cardiology Foundation. J Am Soc Echocardiogr 2009; 22: 755-65.
20.
Guarneri V, Lenihan DJ, Valero V, et al. Long-term cardiac tolerability of trastuzumab in metastatic breast cancer: the M.D. Anderson Cancer Center experience. J Clin Oncol 2006; 24: 4107-15.
21.
Piotrowski G, Gawor R, Bourge RC, et al. Heart remodelling induced by adjuvant trastuzumab-containing chemotherapy for breast cancer overexpressing human epidermal growth factor receptor type 2: a prospective study. Pharmacol Res 2013; 78: 41-8.
22.
Tan-Chiu E, Yothers G, Romond E, et al. Assessment of cardiac dysfunction in a randomized trial comparing doxorubicin and cyclophosphamide followed by paclitaxel, with or without trastuzumab as adjuvant therapy in node-positive, human epidermal growth factor receptor2-overexpressing breast cancer: NSABP B-31. J Clin Oncol 2005; 23: 7811-9.
23.
Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med 2005; 353: 1673-84.
24.
Russell SD, Blackwell KL, Lawrence J, et al. Independent adjudication of symptomatic heart failure with the use of doxorubicin and cyclophosphamide followed by trastuzumab adjuvant therapy: a combined review of cardiac data from the National Surgical Adjuvant breast and Bowel Project B-31 and the North Central Cancer Treatment Group N9831 clinical trials. J Clin Oncol 2010; 28: 3416-21.
25.
Cameron D, Piccart-Gebhart MJ, Gelber RD, et al. Herceptin Adjuvant (HERA) Trial Study Team. 11 years’ follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive early breast cancer: final analysis of the HERceptin Adjuvant (HERA) trial. Lancet 2017; 389: 1195-205.
26.
Perez EA, Romond EH, Suman VJ, et al. Four-year follow-up of trastuzumab plus adjuvant chemotherapy for operable human epidermal growth factor receptor 2-positive breast cancer: joint analysis of data from NCCTG N9831 and NSABP B-31. J Clin Oncol 2011; 29: 3366-73.
27.
Gibson J, Yao RJ, Davis M et al. The impact of mild left ventricular dysfunction on trastuzumab use and oncologic outcomes in early stage breast cancer therapy. J Clin Oncol 2018; 35: e18148a.
28.
Senkus E, Kyriakides S, Ohno S, et al.; ESMO Guidelines Committee. Primary breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2015; 26 Suppl 5: v8-30.
29.
Earl HM, Hiller L, Vallier AL, et al. on behalf of the Persephone Steering Committee and Trial Investigators. 6 versus 12 months of adjuvant trastuzumab for HER2-positive early breast cancer (Persephone): 4-year disease-free survival results of a randomised phase 3 non-inferiority trial. Lancet 2019; 393: 2599-612.
30.
Pivot X, Romieu G, Debled M, et al. PHARE trial investigators. 6 months versus 12 months of adjuvant trastuzumab in early breast cancer (PHARE): final analysis of a multicentre, open-label, phase 3 randomised trial. Lancet 2019; 393: 2591-8.
31.
Gyawali B, Niraula S. Duration of adjuvant trastuzumab in HER2 positive breast cancer: overall and disease free survival results from meta-analyses of randomized controlled trials. Cancer Treat Rev 2017; 60: 18-23.
32.
Spielmann M, Roche H, Delozier T, et al. Trastuzumab for patients with axillary-node-positive breast cancer: results of the FNCLCC-PACS 04 trial. J Clin Oncol 2009; 27: 6129-34.
33.
Wang SY, Long JB, Hurria A, et al. Cardiovascular events, early discontinuation of trastuzumab, and their impact on survival. Breast Cancer Res Treat 2014; 146: 411-9.
34.
Chen J, Long JB, Hurria A, Owusu C, Steingart RM, Gross CP. Incidence of heart failure or cardiomyopathy after adjuvant trastuzumab therapy for breast cancer. J Am Coll Cardiol 2012; 60: 2504-12.
35.
Penson P, Long DL, Howard G, et al. Associations between cardiovascular disease, cancer, and very low high-density lipoprotein cholesterol in the REasons for Geographical and Racial Differences in Stroke (REGARDS) study. Cardiovasc Res 2019; 115: 204-12.
36.
Ganjali S, Ricciuti B, Pirro M, et al. High-density lipoprotein components and functionality in cancer: state-of-the-art. Trends Endocrinol Metab 2019; 30: 12-24.
37.
Mazidi M, Katsiki N, Mikhailidis DP, et al. Apolipoprotein B/apolipoprotein A-I ratio is a better predictor of cancer mortality compared with C-reactive protein: results from two multi-ethnic US populations. J Clin Med 2020; 9: 170.
38.
Petrelli F, Barni S. Meta-analysis of concomitant compared to sequential adjuvant trastuzumab in breast cancer: the sooner the better. Med Oncol 2012; 29: 503-10.
39.
Gong IY, Verma S, Yan AT, et al. Long-term cardiovascular outcomes and overall survival of early-stage breast cancer patients with early discontinuation of trastuzumab: a population-based study. Breast Cancer Res Treat 2016; 157: 535-44.
40.
Patnaik JL, Byers T, DiGuiseppi C, et al. Cardiovascular disease competes with breast cancer as the leading cause of death for older females diagnosed with breast cancer: a retrospective cohort study. Breast Cancer Res 2011; 13: R64.
41.
Seidman A, Hudis C, Pierri MK, et al. Cardiac dysfunction in the trastuzumab clinical trials experience. J Clin Oncol 2002; 20: 1215-21.
42.
Bowles EA, Wellman R, Delate T, et al. C-B2-01: Cardiotoxic chemotherapy is associated with increased heart failure risk among women with breast cancer in the cancer research network. Clin Med Res 2011; 9: 148.
43.
Du XL, Xia R, Burau K, Liu CC. Cardiac risk associated with the receipt of anthracycline and trastuzumab in a large nationwide cohort of older women with breast cancer, 1998–2005. Med Oncol 2011; 28 Suppl 1: S80-90.
44.
ElZarrad MK, Mukhopadhyay P, Mohan N, et al. Trastuzumab alters the expression of genes essential for cardiac function and induces ultrastructural changes of cardiomyocytes in mice. PLoS One 2013; 8: e79543.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.