Introduction
Postoperative peritoneal adhesions (PPA) are the most common, important, morbid and costly complications of abdominopelvic surgery, with an incidence up to 60–93%, and these complications are associated with intestinal obstructions, chronic pain and secondary infertility. Local inflammatory reaction with fibrinous exudate and fibrin formation caused by peritoneal trauma are the initial steps of peritoneal adhesions [1–3]. The equilibrium between fibrin deposition and degradation is important so as to determine normal peritoneal healing or formation of PPA. Peritoneal wound healing is regulated by various cytokines, mediators and growth factors [4, 5].
Experimental and clinical preventive studies which are required for innovations have commonly been conducted by scientists. Solid, gel or liquid barrier, surgical or non-invasive strategies, cellular strategies, drugs, combination strategies, and also biologic strategies such as activated protein C and neurokinin 1 receptor antagonist have been used to prevent PPA in clinical or experimental studies [6–8].
Platelet-rich plasma (PRP) is an autologous concentration of platelets with various growth factors in concentrated plasma and has been used as a popular biologic treatment method recently for dentistry, orthopedics, dermatology and plastic surgery [9, 10]. Various growth factors contained in PRP have many effects on inflammation, wound or tissue healing [11, 12].
The aim of this study is to reveal the effect of PRP on postoperative peritoneal inflammation and adhesions.
Material and methods
This study was carried out at the Experimental Animal Laboratory of Uludağ University after the approval of the Animal Ethics Committee had been obtained by the researchers. All protocols were in accordance with the regulations concerning the care and use of laboratory animals as in the Declaration of Helsinki.
Animals
Twenty-four Wistar albino outbred female rats (mean weight, 250 ±30 g, mean age 7 months) were divided into 3 groups [13]. Platelet-rich plasma was obtained from the blood of an additional 4 rats, based on the standard protocol described by Okuda et al. [14]. The supernatant containing the concentrated platelets was used as PRP.
Experimental models
After isoflurane inhalation anesthesia, rats were laid in a supine position. After abdominal skin preparation, a midline incision was performed. The cecum was pulled out of the abdomen, and a 1 cm long incision was performed. Stool was seen in the side of the wound and sutured by 3/0 silk one by one. Thereafter, the cecum was returned to the normal position [13].
For the control group (C) none, saline group (S) 3 ml of 0.9% saline solution, and PRP group 3 ml of PRP solution were instilled intraperitoneally. The midline incision was closed with 4/0 polypropylene running sutures. 100 mg/kg of paracetamol was injected subcutaneously for analgesia [13].
Parameters
Four rats from each group were sacrificed at the 3rd and 7th days postoperatively with 100–150 mg/kg high dose sodium thiopental. The peritoneal cavity was opened through a “reverse U” incision [13]. Subsequently, the anterior abdominal wall, peritoneal cavity, small bowels and cecum were examined carefully and assessed according to the staging scale [15] (Table I). After macroscopic evaluation, a cecal segment with neighboring mesenteric root were resected for both histopathologic and biochemical examination. Sacrificed animals were put into the Uludağ University Experimental Animal Laboratory’s medical waste and the study was completed.
Table I
Resected cecal specimens were fixed in 10% formol solution. After histopathologic standard protocols, two slices of 3 μm in thickness were prepared by microtome from each specimen. Hematoxylin and eosin were used for assessing giant cell, lymphocyte/plasmocyte, neutrophil, and histiocyte reaction and Masson’s trichrome was used for fibrosis [13]. Giant cell, lymphocyte/plasmocyte, neutrophil, histiocyte reaction and fibrosis were scored from zero to three [16] (Table I). The pathologist in the study group was blinded during the interpretation of the results.
Cecal samples for biochemical assessment were put into dry tubes and taken to the biochemistry laboratory. After biochemical standard protocols for hydroxyproline, hydroxyproline levels were measured with a spectrophotometer [13].
Statistical analysis
All statistical analyses were performed using a statistical software package (SPSS 16.0). Numerical data were expressed as mean and standard derivation. The Kruskal-Wallis test was used for statistical analysis of giant cell, lymphocyte/plasmocyte, neutrophil, histiocyte reaction and fibrosis scores as their values were nonparametric, and the number of subjects in each group was lower than 30. Statistical significance of hydroxyproline levels was assessed using the ANOVA test because all values obtained from three groups were parametric [13]. Results were expressed with a confidence interval of 95%. P-values below 0.05 were considered as statistically significant.
Results
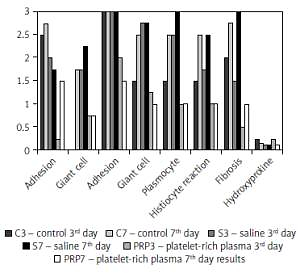
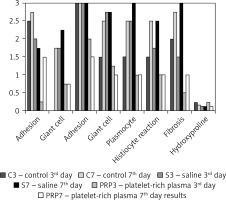
There was no surgical complication or mortality during the study. The results of our study are shown in Table II and Figure 1. Adhesion scores were 2.5 ±0.57 at the 3rd day, 2.75 ±0.95 at the 7th day in the control group, 2.0 ±0.0 at the 3rd day, 1.75 ±0.5 at the 7th day in the saline group, 0.25 ±0.5 at the 3rd day, 1.5 ±0.57 at the 7th day in the PRP group (mean ± standard derivation). Third and 7th day scores were lower in the PRP group but only 3rd day scores were statistically significant (p = 0.01, p = 0.09 respectively).
Table II
Results of macroscopic, histopathologic and biochemistry assessment (mean ± standard derivation, SD)
| Parameter | 3rd day | 7th day | ||||||
|---|---|---|---|---|---|---|---|---|
| Control | Saline | PRP | P-value | Control | Saline | PRP | P-value | |
| Adhesion score | 2.5 ±0.57 | 2.0 ±0.0 | 0.25 ±0.5 | 0.01 | 2.75 ±0.95 | 1.75 ±0.5 | 1.5 ±0.57 | 0.09 |
| Giant cells | 0.0 ±0.0 | 1.75 ±0.5 | 0.75 ±0.5 | 0.12 | 1.75 ±1.5 | 2.25 ±0.95 | 0.75 ±0.5 | 0.185 |
| Neutrophils | 3.0 ±0.0 | 3.0 ±0.0 | 2.0 ±0.0 | 0.004 | 3.0 ±0.0 | 3.0 ±0.0 | 1.5 ±0.57 | 0.005 |
| Lymphocytes | 1.5 ±0.57 | 2.75 ±0.5 | 1.25 ±0.5 | 0.029 | 2.5 ±0.57 | 2.75 ±0.5 | 1.0 ±0.0 | 0.014 |
| Plasmocytes | 1.5 ±0.57 | 2.5 ±0.57 | 1.0 ±0.0 | 0.019 | 2.5 ±0.57 | 3.0 ±0.0 | 1.0 ±0.0 | 0.009 |
| Histiocytes | 1.5 ±0.57 | 1.75 ±0.5 | 1.0 ±0.0 | 0.111 | 2.5 ±0.57 | 2.5 ±0.10 | 1.0 ±0.0 | 0.037 |
| Fibrosis | 2.0 ±1.15 | 1.5 ±1.29 | 0.5 ±0.57 | 0.167 | 2.75 ±0.5 | 3.0 ±0.0 | 1.0 ±0.81 | 0.011 |
| Hydroxyproline* | 0.239 ±0.125 | 0.13 ±0.056 | 0.227 ±0.108 | 0.295 | 0.154 ±0.032 | 0.128 ±0.043 | 0.122 ±0.067 | 0.65 |
Control group scores were 0.0 ±0.0, 3.0 ±0.0, 1.5 ±0.57, 1.5 ±0.57, 1.5 ±0.57, 2.0 ±1.15 at the 3rd day, 1.75 ±1.5, 3.0 ±0.0, 2.5 ±0.57, 2.5 ±0.57, 2.5 ±0.57, 2.75 ±0.5 at the 7th day (mean ± standard derivation; giant cell, neutrophil, lymphocyte, plasmocyte, histiocyte and fibrosis respectively). Saline group scores were 2.0 ±0.0, 1.75 ±0.5, 3.0 ±0.0, 2.75 ±0.5, 2.5 ±0.57, 1.75 ±0.5, 1.75 ±0.5 at the 3rd day, 1.75 ±0.5, 2.25 ±0.95, 3.0 ±0.0, 2.75 ±0.5, 3.0 ±0.0, 2.5 ±0.10, 3.0 ±0.0 at the 7th day (mean ± standard derivation; giant cell, neutrophil, lymphocyte, plasmocyte, histiocyte and fibrosis respectively). Platelet-rich plasma group scores were 0.25 ±0.5, 0.75 ±0.5, 2.0 ±0.0, 1.25 ±0.5, 1.0 ±0.0, 1.0 ±0.0, 0.5 ±0.57 at the 3rd day, 1.5 ±0.57, 0.75 ±0.5, 1.5 ±0.57, 1.0 ±0.0, 1.0 ±0.0, 1.0 ±0.0, 1.0 ±0.81 at the 7th day (mean ± standard derivation; giant cell, neutrophil, lymphocyte, plasmocyte, histiocyte and fibrosis respectively).
Only giant cell 3rd day scores were lower in the control group rather than other scores and all scores were statistically significant (p = 0.012). Neutrophil scores were higher and similar in control and saline groups both at the 3rd and the 7th day and statistically significant (p = 0.04, p = 0.05 respectively). Both 3rd and 7th day lower lymphocyte, plasmocyte scores in PRP group were statistically significant (p < 0.05). Histiocyte reaction and fibrosis scores were lower in the PRP group both at the 3rd and the 7th day but only 7th day scores were statistically significant (p = 0.037, p = 0.011 respectively).
Hydroxyproline levels were measured as 0.239 ±0.125, 0.154 ±0.032 in the control group, 0.13 ±0.056, 0.128 ±0.043 in the saline group, 0.227 ±0.108, 0.122 ±0.067 in the PRP group (mg proline/g protein ± standard derivation, 3rd and 7th days respectively).
The difference of hydroxyproline levels were not statistically significant both at the 3rd and 7th days (p = 0.295, p = 0.65 respectively).
Discussion
Postoperative peritoneal adhesions are a significant cause of morbidity with 60–70% of small bowel obstruction, secondary infertility, chronic pelvic pain, requirement of reoperations after abdominopelvic surgery [17]. Postoperative peritoneal adhesions also have major financial implications; e.g. adhesion-related health care costs in the United States were estimated at $1.3 billion and in the United Kingdom were estimated at £12 million [18].
Postoperative peritoneal adhesions start with contacting of injured peritoneal or visceral surfaces after surgery [19]. Vascular permeability increases after peritoneal trauma initially, followed by exudation of inflammatory cells and subsequently formation of fibrin matrix. This matrix is progressively organized and replaced by tissue fibroblasts, macrophages and giant cells. Then, connections occur between injured tissue or peritoneum by modeling fibrin bands. Destruction of fibrin bands by the fibrinolytic system provided peritoneal healing, while reducing the fibrinolytic system results in persistence of fibrin bands at this stage. The fibroblasts deposit extracellular matrix (ECM) containing collagen and fibronectin leading to adhesion formation. Adhesion tissue is formed by macrophages, eosinophils, red blood cells, tissue debris, mast cells and fibroblast mixture [20, 21].
Solid, gel and liquid barriers such as carboxymethyl cellulose + hyaluronic acid, oxide regenere cellulose, and 4% icodextrin provide adhesion prevention with mechanical separation. Drugs such as anticoagulants, fibrinolytics, thromboxane A2 receptor inhibitors, non-steroid anti-inflammatory drugs, antihistaminics, growth factor inhibitors or modulators, epidermal growth factor or matrix metalloproteinase provide antiadhesive effects with targeting of the steps of adhesion formation [6].
Platelet-rich plasma has been a famous non-surgical, biologic treatment modality for the last two decades. Tendon, cartilage, bone or joint healing in orthopedics, periodontal or gingival healing in dentistry, and chronic wound healing in dermatology are the most common application sites of PRP. Although not exactly the same, the peritoneal healing mechanism is similar to that of other application areas [9–12, 22].
Platelet-rich plasma shows its effects according to peritoneal adhesion formation stages as described below.
Separation of the damaged peritoneal and visceral surfaces are critical for starting peritoneal healing and subsequent peritoneal adhesion formation. Solid, gel or liquid barriers have been developed primarily for separation of injured surfaces during the 5–7 day required for peritoneal re-epithelialization [2, 6]. The liquid form of PRP can provide separation of damaged surfaces.
Inflammation is the initial stage of peritoneal adhesion formation [20]. Platelet-rich plasma has an anti-inflammatory effect associated with interleukin 1 (IL-1), IL-6, IL-10 and tumor necrosis factor-α (TNF-α). These cytokines have an important role in the formation of peritoneal adhesions such as other anti-inflammatory cytokines or growth factors, e.g. hepatocyte growth factor (HGF), IL-8, TGF-β [11, 12, 23].
Tissue plasminogen activator and urokinase-like plasminogen activator are plasminogen activators that convert plasminogen to plasmin that provides degradation of fibrin in the fibrinolytic system [20, 21]. Baeten et al. reported that platelets, especially in platelet-rich plasma, enhanced single chain urokinase type plasminogen activator and increased activation of urokinase type plasminogen activator by serine protease [24].
Matrix metalloproteinases (MMP) are a large family of enzymes that degrade various components of ECM, and are inhibited by tissue-derived inhibitors of matrix metalloproteinase (TIMP). Parietal peritoneum and serosal surface of intraperitoneal organs express MMP and TIMP. Increase in MMP and decrease in TIMP reduce peritoneal adhesions [20, 25]. De Mos et al. reported that PRP showed upregulation of MMP1 and MMP3 [26].
Saline is the most common abdominal cleansing liquid in abdominal surgery and is often used for supplying comparison peritoneal adhesion prevention strategies. Cwalinski et al. reported that normal saline solution may accelerate formation of the peritoneal adhesions [27]. In our study adhesion scores were higher on the 7th day than the 3rd day in both control and PRP groups, but saline groups’ scores were lower on the 7th day than the 3rd day. Platelet-rich plasma had lower adhesion scores compared to the other two groups both on the 3rd and 7th days, but it was not statistically significant. Platelet-rich plasma has a preventive effect on peritoneal adhesions on both 3rd and 7th days.
Fibrosis score is another indicator of peritoneal adhesions and is the most frequently used parameter to evaluate the effectiveness of adhesion preventive strategies [28]. A low fibrosis score indicates weak adhesions. Our study demonstrated that 3rd day scores were lower than 7th day scores in all groups but 7th day scores were significantly lower in the PRP group. The lowest fibrosis scores of PRP showed that PRP reduced peritoneal adhesions.
Neutrophils are the initial inflammatory cells that occur in the early period of peritoneal healing. Decreasing neutrophils and phagocytosis reduced peritoneal adhesions [28]. In our study the lowest neutrophil scores of the PRP group showed reduced PPA.
Histiocytes are important cells of chronic inflammation and a major component of the monocyte–phagocytic system and tissue consolidating mature macrophages [13]. Macrophages have an important role in adhesion formation related to inflammation and plasminogen activators. Decreased macrophages are associated with adhesion formation [20]. In our study, PRP had the lowest histiocyte scores both on the 3rd and 7th days but only 7th day scores were statistically significant. Platelet-rich plasma decreased adhesion formation in the early and late periods.
Additionally, plasmocytes and lymphocytes play a crucial role in chronic inflammation, triggering the subsequent adhesion maintenance [29]. In our study both plasmocyte/lymphocyte scores were statistically significantly lower in the PRP group both at the 3rd and 7th days. The control group has the most significant difference between 3rd and 7th days. Platelet-rich plasma decreased inflammation and adhesion in early and late periods of adhesion formation.
Giant cells are seen in granulomatous inflammation, which is a different pattern of chronic inflammatory reaction and generally occurs due to foreign bodies [13]. The 3rd day giant cell score was the lowest in the control group but the 7th day score was the lowest in the PRP group. Platelet-rich plasma demonstrated low foreign body reaction during the 7th day.
Hydroxyproline is the basic amino acid of collagen structure [13]. 7th day levels of all groups were lower than 3rd day but a statistically significant difference was not noted. Saline group hydroxyproline levels were lowest at the 3rd day. However, PRP group hydroxyproline levels were lowest at the 7th day. Platelet-rich plasma showed reducing effects in the late period of peritoneal adhesion formation.
In conclusion, PRP were used in orthopedic, craniofacial or dermatological healing with presence of various growth factors but effects on peritoneal healing have not been studied yet. A recent study demonstrated that PRP reduced the postoperative peritoneal adhesions with separation of injured tissues, and affecting inflammation, matrix metalloproteinase, plasminogen activation or the other stages of adhesion formation. Long-term studies are needed to evaluate the further effects of PRP on postoperative peritoneal inflammation and adhesions.