Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
ATHEROSCLEROSIS / RESEARCH PAPER
G protein-coupled estrogen receptor GPR30 exerts vasoprotective effects in apolipoprotein E-deficient mice
1
University Hospital Bonn, Germany
Submission date: 2020-01-30
Final revision date: 2020-08-15
Acceptance date: 2020-09-02
Online publication date: 2021-05-03
KEYWORDS
atherosclerosisreactive oxygen speciesglucose toleranceG-1G protein-coupled receptor 30angiotensin II type 1 receptor
TOPICS
ABSTRACT
Introduction:
GPR30 is an intracellular transmembrane G protein-coupled receptor that mediates non-genomic estrogen signaling. The GPR30 agonist G-1 modulates glucose homeostasis and vascular function. However, its impact on vascular inflammation and atherogenesis has not yet been investigated in the atherosclerotic apolipoprotein E-deficient(ApoE-/-) mouse model.
Material and methods:
ApoE-/- mice were fed a high-cholesterol diet for 7 weeks while being treated with the selective GPR30 agonist G-1 (n=6-7). After the treatment period, vascular relaxation capacity, vascular oxidative stress, and atherosclerotic plaque burden were assessed. In vitro, reactive oxygen species, expression levels of the angiotensin II type1(AT1) receptor, and proliferation rate were quantified in human coronary artery smooth muscle cells(HCASMC).
Results:
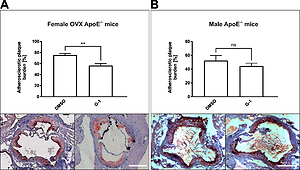
G-1 significantly improved glucose tolerance in vivo (142.2±8.1mg/dl vs. 204.6±13.3mg/dl), G-1 reduced vascular oxidative stress (221±88RLU/s/mg vs.1,983±885RLU/s/mg) and improved endothelium-dependent vasodilation (relaxation to 35.1±4.5% vs.63.0±4.6%). Furthermore, treatment with G-1 significantly reduced the atherosclerotic plaque burden of female ApoE-/- mice (56.5±3.7% vs.75.5±2.9%). In vitro, G-1 provoked a significant downregulation of the AT1 receptor in HCASMC (0.67±0.09-fold). Furthermore, G-1 blunted angiotensin II-induced ROS production by HCASMC (817±7RLU/s/mg vs.1,625±105 RLU/s/mg) and diminished HCASMC proliferation (-26.8±2.7% vs.+50.4±1.7%).
Conclusions:
Selective GPR30 activation improves glucose tolerance in vivo and decreases vascular ROS production in vitro and in vivo. In vitro, the antioxidant effect might be mediated by downregulation of the AT1 receptor. In vivo, the antioxidant effect of G-1 is associated with an improved endothelial function and a reduced atherosclerotic plaque burden in ApoE-deficient mice, indicating beneficial vascular effects of GPR30 activation. GPR30 agonism might thus be a compelling treatment strategy against atherosclerosis.
GPR30 is an intracellular transmembrane G protein-coupled receptor that mediates non-genomic estrogen signaling. The GPR30 agonist G-1 modulates glucose homeostasis and vascular function. However, its impact on vascular inflammation and atherogenesis has not yet been investigated in the atherosclerotic apolipoprotein E-deficient(ApoE-/-) mouse model.
Material and methods:
ApoE-/- mice were fed a high-cholesterol diet for 7 weeks while being treated with the selective GPR30 agonist G-1 (n=6-7). After the treatment period, vascular relaxation capacity, vascular oxidative stress, and atherosclerotic plaque burden were assessed. In vitro, reactive oxygen species, expression levels of the angiotensin II type1(AT1) receptor, and proliferation rate were quantified in human coronary artery smooth muscle cells(HCASMC).
Results:
G-1 significantly improved glucose tolerance in vivo (142.2±8.1mg/dl vs. 204.6±13.3mg/dl), G-1 reduced vascular oxidative stress (221±88RLU/s/mg vs.1,983±885RLU/s/mg) and improved endothelium-dependent vasodilation (relaxation to 35.1±4.5% vs.63.0±4.6%). Furthermore, treatment with G-1 significantly reduced the atherosclerotic plaque burden of female ApoE-/- mice (56.5±3.7% vs.75.5±2.9%). In vitro, G-1 provoked a significant downregulation of the AT1 receptor in HCASMC (0.67±0.09-fold). Furthermore, G-1 blunted angiotensin II-induced ROS production by HCASMC (817±7RLU/s/mg vs.1,625±105 RLU/s/mg) and diminished HCASMC proliferation (-26.8±2.7% vs.+50.4±1.7%).
Conclusions:
Selective GPR30 activation improves glucose tolerance in vivo and decreases vascular ROS production in vitro and in vivo. In vitro, the antioxidant effect might be mediated by downregulation of the AT1 receptor. In vivo, the antioxidant effect of G-1 is associated with an improved endothelial function and a reduced atherosclerotic plaque burden in ApoE-deficient mice, indicating beneficial vascular effects of GPR30 activation. GPR30 agonism might thus be a compelling treatment strategy against atherosclerosis.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.