Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
CLINICAL RESEARCH
Genetic association of lipid-lowering drugs with functional gastrointestinal disorders: a drug-target Mendelian randomization study
1
Guangdong Provincial Key Laboratory of Gastroenterology, Department of Gastroenterology, Institute of Gastroenterology of Guangdong Province, Nanfang Hospital, Southern Medical University, Guangzhou, China
2
Department of Microbiology, Guangdong Provincial Key Laboratory of Tropical Disease Research, School of Public Health, Southern Medical University, Guangzhou, China
These authors had equal contribution to this work
Submission date: 2025-10-21
Final revision date: 2026-01-14
Acceptance date: 2026-02-11
Online publication date: 2026-05-15
Corresponding author
Hongying Fan
Department of Microbiology, Guangdong Provincial Key Laboratory of Tropical Disease Research, School of Public Health, Southern Medical University, Guangzhou, China
Department of Microbiology, Guangdong Provincial Key Laboratory of Tropical Disease Research, School of Public Health, Southern Medical University, Guangzhou, China
Yang Bai
Guangdong Provincial Key Laboratory of Gastroenterology Department of Gastroenterology Institute of Gastroenterology of Guangdong Province Nanfang Hospital, Southern Medical University Guangzhou, China
Guangdong Provincial Key Laboratory of Gastroenterology Department of Gastroenterology Institute of Gastroenterology of Guangdong Province Nanfang Hospital, Southern Medical University Guangzhou, China
KEYWORDS
TOPICS
ABSTRACT
Introduction:
The effect of lipid-lowering drugs on functional gastrointestinal disorders (FGIDs) remains unclear, and the assessment of their relationship is always confounded by extraneous factors.
Material and methods:
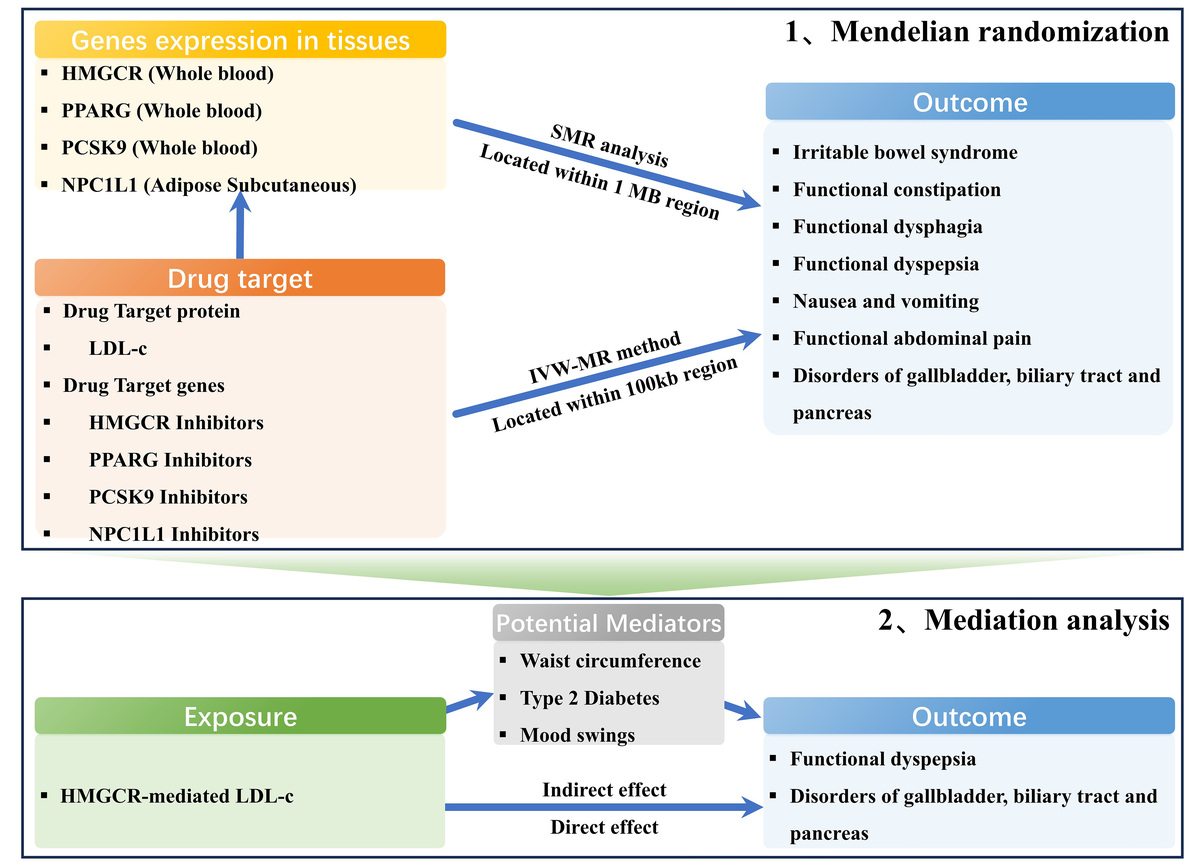
We detected genetic variants linked to LDL cholesterol (LDL-C) levels in or near the lipid-lowering drug targets (HMGCR, PPARG, PCSK9, and NPC1L1), using data from the Global Lipids Genetics Consortium (GLGC) genome-wide association study. Outcome data for FGIDs including seven disorders were extracted from genome-wide association studies (GWAS) datasets. Inverse-variance-weighted Mendelian randomization (IVW-MR) was conducted to identify the causal effect between four genetically proxied lipid-lowering drugs and FGIDs, and mediation Mendelian randomization (MR) was performed to investigate potential mediators. We employed several sensitivity analyses to validate the robustness of the results.
Results:
Seven variants were identified as proxies for LDL-C lowering through HMGCR inhibitors, 3 for PPARG inhibitors, 12 for PCSK9 inhibitors, and 3 for NPC1L1 inhibitors. IVW-MR analysis revealed that genetically proxied HMGCR-mediated LDL-C elevation significantly increased the risk of several gastrointestinal outcomes. These included functional constipation (OR = 1.258, 95% CI: 1.043–1.517), functional dyspepsia (OR = 1.438, 95% CI: 1.014–2.036), nausea and vomiting (OR = 1.536, 95% CI: 1.070–2.207), and disorders of the gallbladder, biliary tract and pancreas (OR = 1.664, 95% CI: 1.392–1.989). Furthermore, mediation analysis identified mood swings as a significant mediator. It accounted for 6.23% of the total effect on functional dyspepsia (OR = 1.023, 95% CI: 1.001–1.058) and for 2.62% of the effect on disorders of the gallbladder, biliary tract, and pancreas (OR = 1.013, 95% CI: 1.001–1.033).
Conclusions:
Our study suggested a potential protective effect of HMGCR inhibitors on FGIDs, which may be mediated by a reduced risk of mood swings.
The effect of lipid-lowering drugs on functional gastrointestinal disorders (FGIDs) remains unclear, and the assessment of their relationship is always confounded by extraneous factors.
Material and methods:
We detected genetic variants linked to LDL cholesterol (LDL-C) levels in or near the lipid-lowering drug targets (HMGCR, PPARG, PCSK9, and NPC1L1), using data from the Global Lipids Genetics Consortium (GLGC) genome-wide association study. Outcome data for FGIDs including seven disorders were extracted from genome-wide association studies (GWAS) datasets. Inverse-variance-weighted Mendelian randomization (IVW-MR) was conducted to identify the causal effect between four genetically proxied lipid-lowering drugs and FGIDs, and mediation Mendelian randomization (MR) was performed to investigate potential mediators. We employed several sensitivity analyses to validate the robustness of the results.
Results:
Seven variants were identified as proxies for LDL-C lowering through HMGCR inhibitors, 3 for PPARG inhibitors, 12 for PCSK9 inhibitors, and 3 for NPC1L1 inhibitors. IVW-MR analysis revealed that genetically proxied HMGCR-mediated LDL-C elevation significantly increased the risk of several gastrointestinal outcomes. These included functional constipation (OR = 1.258, 95% CI: 1.043–1.517), functional dyspepsia (OR = 1.438, 95% CI: 1.014–2.036), nausea and vomiting (OR = 1.536, 95% CI: 1.070–2.207), and disorders of the gallbladder, biliary tract and pancreas (OR = 1.664, 95% CI: 1.392–1.989). Furthermore, mediation analysis identified mood swings as a significant mediator. It accounted for 6.23% of the total effect on functional dyspepsia (OR = 1.023, 95% CI: 1.001–1.058) and for 2.62% of the effect on disorders of the gallbladder, biliary tract, and pancreas (OR = 1.013, 95% CI: 1.001–1.033).
Conclusions:
Our study suggested a potential protective effect of HMGCR inhibitors on FGIDs, which may be mediated by a reduced risk of mood swings.
REFERENCES (52)
1.
Sperber AD, Bangdiwala SI, Drossman DA, et al. Worldwide prevalence and burden of functional gastrointestinal disorders, results of Rome Foundation Global Study. Gastroenterology 2021; 160: 99-114.
2.
Drossman DA. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features and Rome IV. Gastroenterology 2016; S0016-5085(16)00223-7.
3.
Holtmann GJ, Ford AC, Talley NJ. Pathophysiology of irritable bowel syndrome. Lancet Gastroenterol Hepatol 2016; 1: 133-46.
4.
Chiou E, Nurko S. Functional abdominal pain and irritable bowel syndrome in children and adolescents. Therapy 2011; 8: 315-31.
5.
Pierno S, Musumeci O. Pharmacotherapy of the lipid-lowering drugs: update on efficacy and risk. Int J Mol Sci 2023; 24: 996.
6.
Zhang X, Liu Y, Ou Y, et al. Gender-specific association between the regular use of statins and the risk of irritable bowel syndrome: a population-based prospective cohort study. Front Pharmacol 2022; 13: 1044542.
7.
Vieira-Silva S, Falony G, Belda E, et al. Statin therapy is associated with lower prevalence of gut microbiota dysbiosis. Nature 2020; 581: 310-5.
8.
Bissonnette S, Habib R, Sampalis F, Boukas S, Sampalis JS. Efficacy and tolerability of ezetimibe 10 mg/day coadministered with statins in patients with primary hypercholesterolemia who do not achieve target LDL-C while on statin monotherapy: A Canadian, multicentre, prospective study – the Ezetrol Add-On Study. Can J Cardiol 2006; 22: 1035-44.
9.
Boccuzzi SJ, Bocanegra TS, Walker JF, Shapiro DR, Keegan ME. Long-term safety and efficacy profile of simvastatin. Am J Cardiol 1991; 68: 1127-31.
10.
Kasliwal R, Wilton LV, Cornelius V, Aurich-Barrera B, Shakir SA. Safety profile of rosuvastatin: results of a prescription-event monitoring study of 11,680 patients. Drug Saf 2007; 30: 157-70.
11.
Suchartlikitwong S, Laoveeravat P, Teerakanok J, et al. Meta-analysis comparing the effects of statins on the risk of Clostridium difficile diarrhea. Proc (Bayl Univ Med Cent) 2018; 31: 447-52.
12.
Pearlman M, Covin Y, Schmidt R, Mortensen EM, Mansi IA. Statins and lower gastrointestinal conditions: a retrospective cohort study. J Clin Pharmacol 2017; 57: 1053-63.
13.
Gottlieb K, Wacher V, Sliman J, Pimentel M. Review article: inhibition of methanogenic archaea by statins as a targeted management strategy for constipation and related disorders. Aliment Pharmacol Ther 2016; 43: 197-212.
14.
Van Oudenhove L, Crowell MD, Drossman DA, et al. Biopsychosocial Aspects of Functional Gastrointestinal Disorders. Gastroenterology 2016; S0016-5085(16)00218-3.
15.
Hay R, Cullen B, Graham N, et al. Genetic analysis of the PCSK9 locus in psychological, psychiatric, metabolic and cardiovascular traits in UK Biobank. Eur J Hum Genet 2022; 30: 1380-90.
16.
Schmidt AF, Finan C, Gordillo-Maranon M, et al. Genetic drug target validation using Mendelian randomisation. Nat Commun 2020; 11: 3255.
17.
Li XH, Pang WW, Zhang Y, et al. A Mendelian randomization study for drug repurposing reveals bezafibrate and fenofibric acid as potential osteoporosis treatments. Front Pharmacol 2023; 14: 1211302.
18.
Willer CJ, Schmidt EM, Sengupta S, et al. Discovery and refinement of loci associated with lipid levels. Nat Genet 2013; 45: 1274-83.
19.
Hemani G, Zheng J, Elsworth B, et al. The MR-Base platform supports systematic causal inference across the human phenome. Elife 2018; 7: e34408.
20.
Sudlow C, Gallacher J, Allen N, et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. Plos Med 2015; 12: e1001779.
21.
Kurki MI, Karjalainen J, Palta P, et al. FinnGen provides genetic insights from a well-phenotyped isolated population. NATURE 2023; 613: 508-18.
22.
Zhu Z, Zhang F, Hu H, et al. Integration of summary data from GWAS and eQTL studies predicts complex trait gene targets. Nat Genet 2016; 48: 481-7.
23.
Burgess S, Butterworth A, Thompson SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol 2013; 37: 658-65.
24.
Burgess S, Foley CN, Allara E, Staley JR, Howson J. A robust and efficient method for Mendelian randomization with hundreds of genetic variants. Nat Commun 2020; 11: 376.
25.
Bowden J, Del GMF, Minelli C, Davey SG, Sheehan NA, Thompson JR. Assessing the suitability of summary data for two-sample Mendelian randomization analyses using MR-Egger regression: the role of the I2 statistic. Int J Epidemiol 2016; 45: 1961-74.
26.
Bowden J, Davey SG, Haycock PC, Burgess S. Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol 2016; 40: 304-14.
27.
Bowden J, Davey SG, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 2015; 44: 512-25.
28.
Verbanck M, Chen CY, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet 2018; 50: 693-8.
29.
Feldman M, Schiller LR. Disorders of gastrointestinal motility associated with diabetes mellitus. Ann Intern Med 1983; 98: 378-84.
30.
Eijsbouts C, Zheng T, Kennedy NA, et al. Genome-wide analysis of 53,400 people with irritable bowel syndrome highlights shared genetic pathways with mood and anxiety disorders. Nat Genet 2021; 53: 1543-52.
31.
Lee CG, Lee JK, Kang YS, et al. Visceral abdominal obesity is associated with an increased risk of irritable bowel syndrome. Am J Gastroenterol 2015; 110: 310-9.
32.
Woolf B, Zagkos L, Gill D. TwoStepCisMR: A novel method and R package for attenuating bias in cis-Mendelian randomization analyses. Genes (Basel) 2022; 13: 1541.
33.
Sanderson E. Multivariable Mendelian randomization and mediation. Cold Spring Harb Perspect Med 2021; 11: a038984.
34.
Chen L, Yang H, Li H, He C, Yang L, Lv G. Insights into modifiable risk factors of cholelithiasis: A Mendelian randomization study. Hepatology 2022; 75: 785-96.
35.
Mancini GB, Baker S, Bergeron J, et al. Diagnosis, prevention, and management of statin adverse effects and intolerance: Canadian Consensus Working Group update (2016). Can J Cardiol 2016; 32: S35-65.
36.
Finegold JA, Manisty CH, Goldacre B, Barron AJ, Francis DP. What proportion of symptomatic side effects in patients taking statins are genuinely caused by the drug? Systematic review of randomized placebo-controlled trials to aid individual patient choice. Eur J Prev Cardiol 2014; 21: 464-74.
37.
Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med 2008; 359: 2195-207.
38.
Martin D, Schmidt R, Mortensen EM, Mansi I. Association of statin therapy and risks of cholelithiasis, biliary tract diseases, and gallbladder procedures: retrospective cohort analysis of a US population. Ann Pharmacother 2016; 50: 161-71.
39.
Nolan JA, Skuse P, Govindarajan K, et al. The influence of rosuvastatin on the gastrointestinal microbiota and host gene expression profiles. Am J Physiol Gastrointest Liver Physiol 2017; 312: G488-97.
40.
Caparros-Martin JA, Lareu RR, Ramsay JP, et al. Statin therapy causes gut dysbiosis in mice through a PXR-dependent mechanism. Microbiome 2017; 5: 95.
41.
Catry E, Pachikian BD, Salazar N, Neyrinck AM, Cani PD, Delzenne NM. Ezetimibe and simvastatin modulate gut microbiota and expression of genes related to cholesterol metabolism. Life Sci 2015; 132: 77-84.
42.
Han JX, Tao ZH, Wang JL, et al. Microbiota-derived tryptophan catabolites mediate the chemopreventive effects of statins on colorectal cancer. Nat Microbiol 2023; 8: 919-33.
43.
Peng Y, Ma Y, Luo Z, Jiang Y, Xu Z, Yu R. Lactobacillus reuteri in digestive system diseases: focus on clinical trials and mechanisms. Front Cell Infect Microbiol 2023; 13: 1254198.
44.
Kubota M, Ito K, Tomimoto K, et al. Lactobacillus reuteri DSM 17938 and magnesium oxide in children with functional chronic constipation: a double-blind and randomized clinical trial. Nutrients 2020; 12: 225.
45.
Wang G, Huang S, Cai S, et al. Lactobacillus reuteri ameliorates intestinal inflammation and modulates gut microbiota and metabolic disorders in dextran sulfate sodium-induced colitis in mice. Nutrients 2020; 12: 2298.
46.
Vasant DH, Ford AC. Functional gastrointestinal disorders in inflammatory bowel disease: Time for a paradigm shift? World J Gastroenterol 2020; 26: 3712-9.
47.
Kim KO. Functional gastrointestinal disorders in patients with inflammatory bowel disease. Korean J Gastroenterol 2022; 79: 4-11.
48.
Lochhead P, Khalili H, Sachs MC, Chan AT, Olen O, Ludvigsson JF. Association between statin use and inflammatory bowel diseases: results from a Swedish, nationwide, population-based case-control study. J Crohns Colitis 2021; 15: 757-65.
49.
Sasaki M, Bharwani S, Jordan P, et al. The 3-hydroxy-3-methylglutaryl-CoA reductase inhibitor pravastatin reduces disease activity and inflammation in dextran-sulfate induced colitis. J Pharmacol Exp Ther 2003; 305: 78-85.
50.
Walker AJ, Kim Y, Borissiouk I, et al. Statins: neurobiological underpinnings and mechanisms in mood disorders. Neurosci Biobehav Rev 2021; 128: 693-708.
51.
Sommer IE, Gangadin SS, de Witte LD, et al. Simvastatin augmentation for patients with early-phase schizophrenia-spectrum disorders: a double-blind, randomized placebo-controlled trial. Schizophr Bull 2021; 47: 1108-15.
52.
Kohler-Forsberg O, Gasse C, Berk M, Ostergaard SD. Do statins have antidepressant effects? CNS Drugs 2017; 31: 335-43.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.