Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
EXPERIMENTAL RESEARCH
The effect of Angelica dahurica extract on breast carcinogenesis in mouse
1
Obstetric Department, Qilu Hospital, Shandong University, China
Submission date: 2020-03-17
Final revision date: 2020-06-10
Acceptance date: 2020-06-25
Online publication date: 2020-11-27
Publication date: 2026-04-12
Arch Med Sci 2026;22(3)
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Angelica dahurica extract (ADE) has been demonstrated to possess functions of immune response modulation, breast injury and fibrosis amelioration, breast regeneration regulation, and suppression of breast cancer.
Material and methods:
To explore the effect of the ADE on breast cancer generation and epithelial-mesenchymal transition (EMT) in vivo, a breast cancer mouse model was established. Chinese traditional medicine ADE was administered to mice bearing breast cancer, which were randomly divided into a negative group, ADE group (two doses), and model group. The breast tissue of the mouse model was fixed and H&E staining was performed for subsequent histological observation. Immunofluorescence assay and immunohistochemical staining were performed to examine the breast cancer related proteins, SNCG and BCRP in breast tissue; Western blotting and quantitative real-time polymerase chain reaction (PCR) were performed to detect proteins and gene expression of epithelial-mesenchymal transition (EMT)-related markers, E-cadherin, N-cadherin, vimentin, transforming growth factor (TGF-), and SNAIL, as well as signals in the MAPK and JAK/STAT signaling pathways.
Results:
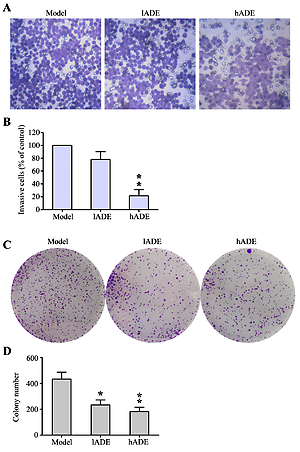
Pathological manifestation of breast tissues in ADE-treated mouse was clearly reduced in both visual inspection and H&E staining. Breast cancer specific SNCG and BCRP expression significantly decreased after ADE administration. Furthermore, increased E-cadherin and decreased N-cadherin, vimentin, SNAIL and TGF- expression might be associated with the anti-EMT property of ADE. Both MAPK and JAK/STAT signaling pathways were observed to be inactivated after ADE treatment.
Conclusions:
Collectively, the evidence showed that ADE restrained breast cancer development in vivo through blocking the EMT process, probably by down-regulation of MAPK and JAK/STAT signaling pathways.
Angelica dahurica extract (ADE) has been demonstrated to possess functions of immune response modulation, breast injury and fibrosis amelioration, breast regeneration regulation, and suppression of breast cancer.
Material and methods:
To explore the effect of the ADE on breast cancer generation and epithelial-mesenchymal transition (EMT) in vivo, a breast cancer mouse model was established. Chinese traditional medicine ADE was administered to mice bearing breast cancer, which were randomly divided into a negative group, ADE group (two doses), and model group. The breast tissue of the mouse model was fixed and H&E staining was performed for subsequent histological observation. Immunofluorescence assay and immunohistochemical staining were performed to examine the breast cancer related proteins, SNCG and BCRP in breast tissue; Western blotting and quantitative real-time polymerase chain reaction (PCR) were performed to detect proteins and gene expression of epithelial-mesenchymal transition (EMT)-related markers, E-cadherin, N-cadherin, vimentin, transforming growth factor (TGF-), and SNAIL, as well as signals in the MAPK and JAK/STAT signaling pathways.
Results:
Pathological manifestation of breast tissues in ADE-treated mouse was clearly reduced in both visual inspection and H&E staining. Breast cancer specific SNCG and BCRP expression significantly decreased after ADE administration. Furthermore, increased E-cadherin and decreased N-cadherin, vimentin, SNAIL and TGF- expression might be associated with the anti-EMT property of ADE. Both MAPK and JAK/STAT signaling pathways were observed to be inactivated after ADE treatment.
Conclusions:
Collectively, the evidence showed that ADE restrained breast cancer development in vivo through blocking the EMT process, probably by down-regulation of MAPK and JAK/STAT signaling pathways.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.