Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
PULMONOLOGY / BASIC RESEARCH
The plasma metabolite-mediated relationship between inflammatory cytokines and acute respiratory distress syndrome
1
Jinggangshan University Affiliated Hospital, Ji ‘an City, Jiangxi, China
2
Jiangxi Provincial People’s Hospital, The First Affiliated Hospital of Nanchang Medical College, Nanchang, Jiangxi, China
Submission date: 2025-01-02
Final revision date: 2025-02-16
Acceptance date: 2025-02-23
Online publication date: 2025-04-27
Publication date: 2026-04-30
Corresponding author
Zhiying Lin
Jiangxi Provincial People’s Hospital The First Affiliated Hospital of Nanchang Medical College No. 92 Aiguo Road Nanchang, Jiangxi Province, China Phone: 0791-86895550
Jiangxi Provincial People’s Hospital The First Affiliated Hospital of Nanchang Medical College No. 92 Aiguo Road Nanchang, Jiangxi Province, China Phone: 0791-86895550
Arch Med Sci 2026;22(2):1130-1140
KEYWORDS
Mendelian analysisacute respiratory distress syndromeinflammatory cytokinesplasma metabolitesmediation
TOPICS
ABSTRACT
Introduction:
Biomarkers of acute respiratory distress syndrome (ARDS) can provide precise treatment options. ARDS is a clinical syndrome characterized by diffuse lung inflammation and edema, often leading to acute respiratory failure. We used Mendelian randomization (MR) and mediation analysis to assess the potential impact of inflammatory factors and metabolites on ARDS.
Material and methods:
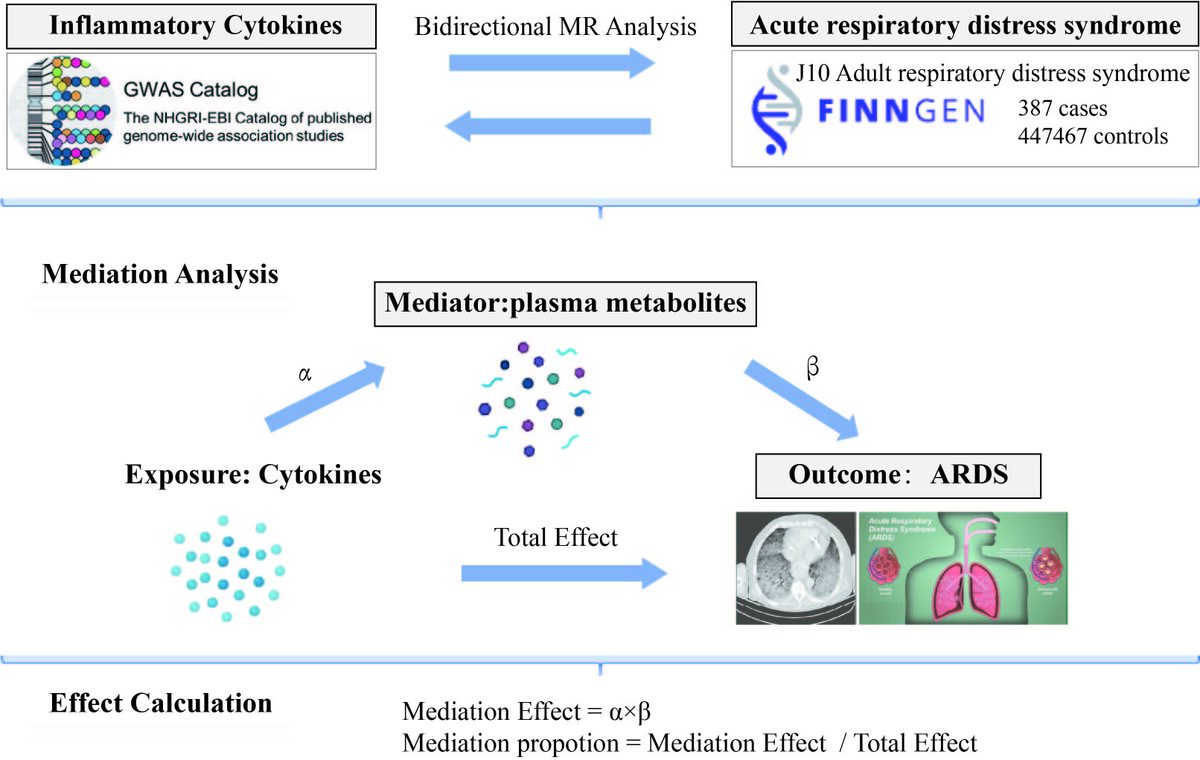
The summary statistics of 1400 plasma metabolite traits and 41 inflammatory cytokine traits were obtained from publicly available genome-wide association studies (GWAS). The inverse variance weighted method was used for bidirectional MR analysis to assess potential causal relationships. Several sensitivity analyses were also used to ensure reliable MR results. Mediation analysis was used to determine the pathway from inflammatory factors to ARDS mediated by plasma metabolism, and the proportion of mediation effects explained by plasma metabolites was estimated.
Results:
MR analysis revealed the causal effects of 5 inflammatory cytokines and 18 plasma metabolites on ARDS. Reverse MR analysis showed that ARDS has no effect on these 5 inflammatory cytokines. In addition, we identified 18 pathogenic metabolites associated with ARDS. Mediation analysis suggested that ceramide levels and the a-tocopherol to sulfate ratio may mediate the causal pathway from inflammatory factors to ARDS, with mediation ratios of 14.1% and 17.7%, respectively (p < 0.05).
Conclusions:
Elevated ceramide levels and the a-tocopherol to sulfate ratio may reduce the risk of ARDS. Our research provides further insights into the complex interactions between inflammatory cytokines and metabolites in the development of ARDS, supporting innovative strategies for ARDS prevention and treatment.
Biomarkers of acute respiratory distress syndrome (ARDS) can provide precise treatment options. ARDS is a clinical syndrome characterized by diffuse lung inflammation and edema, often leading to acute respiratory failure. We used Mendelian randomization (MR) and mediation analysis to assess the potential impact of inflammatory factors and metabolites on ARDS.
Material and methods:
The summary statistics of 1400 plasma metabolite traits and 41 inflammatory cytokine traits were obtained from publicly available genome-wide association studies (GWAS). The inverse variance weighted method was used for bidirectional MR analysis to assess potential causal relationships. Several sensitivity analyses were also used to ensure reliable MR results. Mediation analysis was used to determine the pathway from inflammatory factors to ARDS mediated by plasma metabolism, and the proportion of mediation effects explained by plasma metabolites was estimated.
Results:
MR analysis revealed the causal effects of 5 inflammatory cytokines and 18 plasma metabolites on ARDS. Reverse MR analysis showed that ARDS has no effect on these 5 inflammatory cytokines. In addition, we identified 18 pathogenic metabolites associated with ARDS. Mediation analysis suggested that ceramide levels and the a-tocopherol to sulfate ratio may mediate the causal pathway from inflammatory factors to ARDS, with mediation ratios of 14.1% and 17.7%, respectively (p < 0.05).
Conclusions:
Elevated ceramide levels and the a-tocopherol to sulfate ratio may reduce the risk of ARDS. Our research provides further insights into the complex interactions between inflammatory cytokines and metabolites in the development of ARDS, supporting innovative strategies for ARDS prevention and treatment.
REFERENCES (38)
1.
Bos LDJ, Ware LB. Acute respiratory distress syndrome: causes, pathophysiology, and phenotypes. Lancet 2022; 400: 1145-56.
2.
Osorio-Valencia S, Zhou B. Roles of macrophages and endothelial cells and their crosstalk in acute lung injury. Biomedicines 2024; 12: 632.
3.
Meyer NJ, Gattinoni L, Calfee CS. Acute respiratory distress syndrome. Lancet 2021; 398: 622-37.
4.
Ashbaugh D, Boyd Bigelow D, Petty T, Levine B. Acute respiratory distress in adults. Lancet 1967; 290: 319-23.
5.
Calfee CS, Delucchi K, Parsons PE, Thompson BT, Ware LB, Matthay MA. Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir Med 2014; 2: 611-20.
6.
Calfee CS, Delucchi KL, Sinha P, et al. Acute respiratory distress syndrome subphenotypes and differential response to simvastatin: secondary analysis of a randomised controlled trial. Lancet Respir Med 2018; 6: 691-8.
7.
The ARDS Definition Task Force. Acute respiratory distress syndrome: the Berlin definition. JAMA 2012; 307: 2526-33.
8.
Xiong S, Hong Z, Huang LS, et al. IL-1b suppression of VE-cadherin transcription underlies sepsis-induced inflammatory lung injury. J Clin Invest 2020; 130: 3684-98.
9.
Dolinay T, Kim YS, Howrylak J, et al. Inflammasome-regulated cytokines are critical mediators of acute lung injury. Am J Respir Crit Care Med 2012; 185: 1225-34.
10.
Mehta P, Samanta R, Wick K, et al. Elevated ferritin, mediated by IL-18 is associated with systemic inflammation and mortality in acute respiratory distress syndrome (ARDS). Thorax 2024; 79: 227-35.
11.
Rogers AJ, Guan J, Trtchounian A, et al. Association of elevated plasma interleukin-18 level with increased mortality in a clinical trial of statin treatment for acute respiratory distress syndrome. Crit Care Med 2019; 47: 1089-96.
12.
Xu H, Sheng S, Luo W, Xu X, Zhang Z. Acute respiratory distress syndrome heterogeneity and the septic ARDS subgroup. Front Immunol 2023; 14: 1277161.
13.
Johnson CH, Ivanisevic J, Siuzdak G. Metabolomics: beyond biomarkers and towards mechanisms. Nat Rev Mol Cell Biol 2016; 17: 451-9.
14.
Serkova NJ, Van Rheen Z, Tobias M, Pitzer JE, Wilkinson JE, Stringer KA. Utility of magnetic resonance imaging and nuclear magnetic resonance-based metabolomics for quantification of inflammatory lung injury. Am J Physiol Lung Cell Mol Physiol 2008; 295: L152-61.
15.
Stringer KA, McKay RT, Karnovsky A, Quémerais B, Lacy P. Metabolomics and its application to acute lung diseases. Front Immunol 2016; 7: 44.
16.
Lin WC, Fessler MB. Regulatory mechanisms of neutrophil migration from the circulation to the airspace. Cell Mol Life Sci 2021; 78: 4095-124.
17.
Pietzner M, Stewart ID, Raffler J, et al. Plasma metabolites to profile pathways in noncommunicable disease multimorbidity. Nat Med 2021; 27: 471-9.
18.
Tan T, Yang F, Wang Z, Gao F, Sun L. Mediated Mendelian randomization analysis to determine the role of immune cells in regulating the effects of plasma metabolites on childhood asthma. Medicine (Baltimore) 2024; 103: e38957.
19.
Li Y, Wang X, Zhang Z, Shi L, Cheng L, Zhang X. Effect of the gut microbiome, plasma metabolome, peripheral cells, and inflammatory cytokines on obesity: a bidirectional two-sample Mendelian randomization study and mediation analysis. Front Immunol 2024; 15: 1348347.
20.
Zhao JH, Stacey D, Eriksson N, et al. Genetics of circulating inflammatory proteins identifies drivers of immune-mediated disease risk and therapeutic targets. Nat Immunol 2023; 24: 1540-51.
21.
Chen Y, Lu T, Pettersson-Kymmer U, et al. Genomic atlas of the plasma metabolome prioritizes metabolites implicated in human diseases. Nat Genet 2023; 55: 44-53.
22.
Burgess S, Small DS, Thompson SG. A review of instrumental variable estimators for Mendelian randomization. Stat Methods Med Res 2017; 26: 2333-55.
23.
Ponsford MJ, Gkatzionis A, Walker VM, et al. Cardiometabolic traits, sepsis, and severe COVID-19: a Mendelian randomization investigation. Circulation 2020; 142: 1791-3.
24.
Burgess S, Butterworth A, Thompson SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol 2013; 37: 658-65.
25.
Sanderson E. Multivariable Mendelian randomization and mediation. Cold Spring Harb Perspect Med 2021; 11: a038984.
26.
Cohen JF, Chalumeau M, Cohen R, Korevaar DA, Khoshnood B, Bossuyt PMM. Cochran’s Q test was useful to assess heterogeneity in likelihood ratios in studies of diagnostic accuracy. J Clin Epidemiol 2015; 68: 299-306.
27.
Burgess S, Thompson SG. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol 2017; 32: 377-89.
28.
Pandolfi L, Fossali T, Frangipane V, et al. Broncho-alveolar inflammation in COVID-19 patients: a correlation with clinical outcome. BMC Pulm Med 2020; 20: 301.
29.
Matthay MA, Zemans RL, Zimmerman GA, et al. Acute respiratory distress syndrome. Nat Rev Dis Primers 2019; 5: 18.
30.
Wishart DS. Metabolomics for investigating physiological and pathophysiological processes. Physiol Rev 2019; 99: 1819-75.
31.
Grassin-Delyle S, Roquencourt C, Moine P, et al: Metabolomics of exhaled breath in critically ill COVID-19 patients: a pilot study. EBioMedicine 2021; 63: 103154.
32.
Salazar-Puerta AI, Rincon-Benavides MA, Cuellar-Gaviria TZ, et al. Engineered extracellular vesicles derived from dermal fibroblasts attenuate inflammation in a murine model of acute lung injury. Adv Mater 2023; 35: e2210579.
33.
Lu Y, Wu Y, Huang M, et al. Fuzhengjiedu formula exerts protective effect against LPS-induced acute lung injury via gut-lung axis. Phytomedicine 2024; 123: 155190.
34.
Hepokoski M, Wang J, Li K, et al. Altered lung metabolism and mitochondrial DAMPs in lung injury due to acute kidney injury. Am J Physiol Lung Cell Mol Physiol 2021; 320: L821-31.
35.
Emdin CA, Khera AV, Kathiresan S. Mendelian randomization. JAMA 2017; 318: 1925-6.
36.
Carter AR, Sanderson E, Hammerton G, et al. Mendelian randomisation for mediation analysis: current methods and challenges for implementation. Eur J Epidemiol 2021; 36: 465-78.
37.
Huijuan O, Yi W, Jie W, Yanli J. Mechanisms of pulmonary microvascular endothelial cells barrier dysfunction induced by LPS: the roles of ceramides and the Txnip/NLRP3 inflammasome. Microvasc Res 2023; 147(0).
38.
Hu M, Yang J, Xu Y. Effect of a-tocopherol in alleviating the lipopolysaccharide-induced acute lung injury via inhibiting nuclear factor kappa-B signaling pathways. Bioengineered 2022, 13: 3958-68.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.