Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
CLINICAL RESEARCH
A study exploring the effects of exercise on cuproptosis targets in Alzheimer’s disease brains based on large-scale data
1
Department of Neurology, The Second Affiliated Hospital of Anhui Medical University, China
2
Department of Neurology, Kunshan Traditional Chinese Medicine Hospital, China
Submission date: 2025-11-07
Final revision date: 2025-12-03
Acceptance date: 2026-01-09
Online publication date: 2026-04-16
Corresponding author
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder marked by memory loss and cognitive decline. Epidemiological studies have linked sedentary behaviour to increased risk of chronic diseases, but the molecular mechanisms connecting prolonged inactivity to AD remain unclear. Copper dyshomeostasis and a newly defined form of copper-induced cell death – cuproptosis – have been implicated in neurodegeneration. This study investigates the role of cuproptosis-related genes in sedentary elderly individuals and AD patients to identify potential biomarkers and therapeutic targets.
Material and methods:
Gene expression profiles from GEO datasets GSE110298 (hippocampus, sedentary AD vs. control) and GSE9103 (skeletal muscle, sedentary vs. active aging) were processed with the limma package (|log2FC| ≥ 1, adj. p < 0.05). Differentially expressed genes (DEGs) were intersected with a curated cuproptosis gene list. KEGG and GO analyses identified enriched pathways. Protein–protein interaction (PPI) networks in Cytoscape highlighted hub genes. Key candidates were validated by quantitative PCR (qPCR) and Western blot in hippocampal and quadriceps tissues of exercise and sedentary mouse models.
Results:
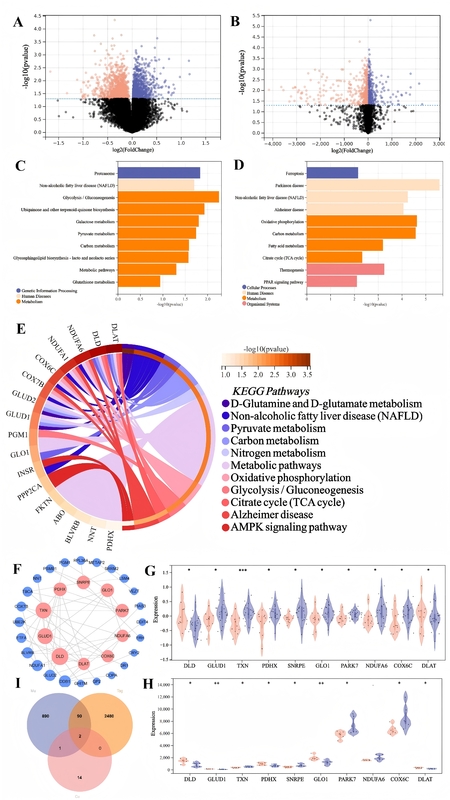
In GSE110298 and GSE9103, 2480 and 890 DEGs were identified, respectively, with 90 overlapping genes enriched in mitochondrial metabolism and neurodegeneration pathways. PPI analysis ranked DLD and DLAT as top hubs; these two remained after intersecting with the cuproptosis gene set. Both DLD and DLAT were significantly upregulated in hippocampi of sedentary AD subjects and in skeletal muscle of sedentary aging individuals. qPCR confirmed 1.6- to 2.8-fold mRNA increases for DLD and DLAT (p < 0.001), and Western blot showed 1.5- to 2.0-fold protein elevation (p < 0.05). Additional modulation of TXN and FDX1 was observed.
Conclusions:
Our integrative analysis identifies DLD and DLAT as key cuproptosis markers dysregulated by sedentary behaviour in AD-relevant tissues. These findings implicate copper-driven cell death in linking prolonged inactivity to neurodegeneration, and highlight DLD/DLAT as promising biomarkers and therapeutic targets. Future work should expand cohort validation and dissect mechanistic roles via multi-omics and functional assays.
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder marked by memory loss and cognitive decline. Epidemiological studies have linked sedentary behaviour to increased risk of chronic diseases, but the molecular mechanisms connecting prolonged inactivity to AD remain unclear. Copper dyshomeostasis and a newly defined form of copper-induced cell death – cuproptosis – have been implicated in neurodegeneration. This study investigates the role of cuproptosis-related genes in sedentary elderly individuals and AD patients to identify potential biomarkers and therapeutic targets.
Material and methods:
Gene expression profiles from GEO datasets GSE110298 (hippocampus, sedentary AD vs. control) and GSE9103 (skeletal muscle, sedentary vs. active aging) were processed with the limma package (|log2FC| ≥ 1, adj. p < 0.05). Differentially expressed genes (DEGs) were intersected with a curated cuproptosis gene list. KEGG and GO analyses identified enriched pathways. Protein–protein interaction (PPI) networks in Cytoscape highlighted hub genes. Key candidates were validated by quantitative PCR (qPCR) and Western blot in hippocampal and quadriceps tissues of exercise and sedentary mouse models.
Results:
In GSE110298 and GSE9103, 2480 and 890 DEGs were identified, respectively, with 90 overlapping genes enriched in mitochondrial metabolism and neurodegeneration pathways. PPI analysis ranked DLD and DLAT as top hubs; these two remained after intersecting with the cuproptosis gene set. Both DLD and DLAT were significantly upregulated in hippocampi of sedentary AD subjects and in skeletal muscle of sedentary aging individuals. qPCR confirmed 1.6- to 2.8-fold mRNA increases for DLD and DLAT (p < 0.001), and Western blot showed 1.5- to 2.0-fold protein elevation (p < 0.05). Additional modulation of TXN and FDX1 was observed.
Conclusions:
Our integrative analysis identifies DLD and DLAT as key cuproptosis markers dysregulated by sedentary behaviour in AD-relevant tissues. These findings implicate copper-driven cell death in linking prolonged inactivity to neurodegeneration, and highlight DLD/DLAT as promising biomarkers and therapeutic targets. Future work should expand cohort validation and dissect mechanistic roles via multi-omics and functional assays.
REFERENCES (22)
1.
Zhang XX, Tian Y, Wang ZT, Ma YH, Tan L, Yu JT. The epidemiology of Alzheimer’s disease modifiable risk factors and prevention. J Prev Alzheimers Dis 2021; 8: 313-21,.
2.
Lin M, Zhou Y, Liang P, et al. Identification of Alzheimer’s disease biomarkers and their immune function characterization. Arch Med Sci 2025; 21: 233-57.
4.
Wallace LMK, Theou O, Godin J, Andrew MK, Bennett DA, Rockwood K. Investigation of frailty as a moderator of the relationship between neuropathology and dementia in Alzheimer’s disease: a cross-sectional analysis of data from the rush memory and aging project. Lancet Neurol 2019; 18: 177-84.
5.
Meng D, Luo G, Liu P. Copper metabolism and cuproptosis in Alzheimer’s disease: mechanisms and therapeutic potential. Biomed Pharmacother 2025; 190: 118354.
6.
Sensi SL, Granzotto A, Siotto M, Squitti R. Copper and zinc dysregulation in Alzheimer’s disease. Trends Pharmacol Sci 2018; 39: 1049-63.
7.
Scheiber IF, Mercer JFB, Dringen R. Metabolism and Functions of copper in brain. Prog Neurobiol 2014; 116: 33-57.
8.
Roy RG, Mandal PK, Maroon JC. Oxidative stress occurs prior to amyloid Ab plaque formation and tau phosphorylation in Alzheimer’s disease: role of glutathione and metal ions. ACS Chem Neurosci 2023; 14: 2944-54.
9.
Chen L, Min J, Wang F. Copper homeostasis and cuproptosis in health and disease. Signal Transduct Target Ther 2022; 7: 378.
10.
The Molecular Mechanisms of Cuproptosis and Its Relevance to Cardiovascular Disease-All Databases Available online: https://webofscience.clarivate... (accessed on 28 April 2025).
11.
Home - GEO - NCBI Available online: https://www.ncbi.nlm.nih.gov/g... (accessed on 28 April 2025).
12.
Kanehisa, M, Furumichi, M, Sato, Y, Kawashima, M, Ishiguro-Watanabe, M. KEGG for taxonomy-based analysis of pathways and genomes. Nucleic Acids Res 2023; 51: D587-92.
13.
Everaert C, Luypaert M, Maag JLV, et al. Benchmarking of RNA-sequencing analysis workflows using whole transcriptome RT-qPCR expression data. Sci Rep 2017; 7: 1559.
14.
Pillai-Kastoori L, Schutz-Geschwender AR, Harford JA. A systematic approach to quantitative western blot analysis. Anal Biochem 2020; 593: 113608.
15.
Zhang E, Dai F, Chen T, Liu S, Xiao C, Shen X. Diagnostic models and predictive drugs associated with cuproptosis hub genes in Alzheimer’s disease. Front Neurol 2023; 13: 1064639,.
16.
Ahmad W, Ebert PR. Suppression of a Core metabolic enzyme dihydrolipoamide dehydrogenase (Dld) protects against amyloid beta toxicity in C. elegans model of Alzheimer’s disease. Genes Dis 2021; 8: 849-66.
17.
Brown AM, Gordon D, Lee H, et al. Testing for linkage and association across the dihydrolipoyl dehydrogenase gene region with Alzheimer’s disease in three sample populations. Neurochem Res 2007; 32: 857-69.
18.
Chen G, Xi E, Gu X, Wang H, Tang Q. The study on cuproptosis in Alzheimer’s disease based on the cuproptosis key gene FDX1. Front Aging Neurosci 2024; 16: 1480332.
19.
Copper induces cognitive impairment in mice via modulation of cuproptosis and CREB signaling-All Databases Available online: https://webofscience.clarivate... (accessed on 28 April 2025).
20.
Agarwal R, Kushwaha SS, Tripathi CB, Singh N, Chhillar N. Serum copper in Alzheimer’s disease and vascular dementia. Indian J Clin Biochem 2008; 23: 369-74.
21.
AIBL research group; Rembach A, Doecke JD, Roberts BR, et al. Longitudinal analysis of serum copper and ceruloplasmin in Alzheimer’s disease. J Alzheimers Dis 2013; 34: 171-82.
22.
Rakhra G, Masih D, Vats A, et al. Effect of physical activity and age on plasma copper, zinc, iron, and magnesium concentration in physically active healthy males. Nutrition 2017; 43-44: 75-82.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.