Introduction
N-3 polyunsaturated fatty acids (N-3 PUFA) mainly include a-monolinolenic acid (ALA), eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Different populations have different needs for n-3 PUFA. As an immunonutrient, n-3 PUFA plays a pharmacological role in the prevention and treatment of coronary heart disease, hypertension, arthritis, other inflammatory and autoimmune diseases, and tumors. The fetus receives nutrients from the mother through the placenta [1–4]. PUFAs are important components of biological cell membranes and transmitters of the neuroendocrine and immune systems. They regulate the fluidity of cellular membranes, affect the function of neurotransmitters, and regulate the production and activity of certain hormones. They have very important physiological functions in organisms. Fatty acids obtained from the mother are influenced by the composition of the maternal diet. The placenta also plays an important role in the transport of fatty acids from the mother to the fetus. Preeclampsia is often associated with fetal growth restriction and early placental development defects. The placenta is one of the main sources of lipid peroxidation, and n-3 PUFA plays a potential important regulatory role in lipid metabolism in the placenta and fetus. At the same time, n-3 PUFA, especially DHA, is an important component of the cellular membranes of the retina and the cerebral cortex, and plays a definite role in promoting the development of vision and the central nervous system [5–7]. However, according to the latest protocol from the European Atherosclerosis Society, currently marketed n-3 PUFA reduces neither cardiovascular morbidity nor mortality. Extensive research has been carried out on n-3 PUFA and has shown that it has a wide range of biological characteristics and functions, and plays an important role not only in regulating lipid metabolism but also in inflammation, and it is very important for brain development and function of the brain [8, 9]. Based on reviewing eligible randomized controlled trials (RCTs), the aim of this study was to explore the effect of n-3 PUFA supplementation on pregnancy outcomes.
Material and methods
Search strategy
The data of clinical indexes on the effect of n-3 PUFA supplementation on pregnancy outcomes based on eligible RCTs were obtained from the included studies. In the databases (Cochrane, PubMed, and Embase), all the relevant RCTs before January 2021 were reviewed. The references in eligible RCTs were also reviewed. The key words included fatty acids, omega-3, n-3 polyunsaturated fatty acid, PUFA, n-3 PUFA, a-linolenic acid, fish oil, EPA, DHA, ALA, pregnancy, random, randomized control study, randomized controlled trial, RCT. All the above key words were combined with “AND” or “OR”. Literature was retrieved by two investigators independently. However, when there was disagreement, a third investigator was involved to make a decision.
Following the PICOS (participants, intervention, comparison, outcome, study design) principle, the key search terms included (P, participants) pregnant women; (I, interventions) pregnant women in the treatment group treated with n-3 PUFA, pregnant women in the control group treated with standard care, normal diet, olive oil or placebo; (C/O, comparison/outcome) the comparison of pregnancy outcome; (S, study design) designed as a RCT. All analysis procedures followed the PRISMA principle and guideline.
Study selection criteria
The included studies had to meet all the following inclusion criteria: (1) the study was designed as an RCT; (2) the subjects were pregnant women; (3) patients were treated with n-3 PUFA; 4) articles were published in English or Chinese.
If a study met any of the following exclusion criteria, it was excluded: (1) duplicate articles or similar results; (2) obvious data errors; (3) cohort study, case control study, case series, theoretical research or reviews, guideline, reports, meta-analyses, or other forms of research or comments that were not designed as RCT; (4) data irrelevant to this study.
The studies were reviewed by two investigators independently to determine whether the included studies met the inclusion criteria and did not meet the exclusion criteria. A third investigator was asked to resolve any disagreement.
Data extraction and quality assessment
In all the included RCTs, two categories of data (the basic characteristics of articles and the main clinical indexes) were extracted. The basic characteristics of articles contained authors’ names, publication year, detailed interventions, sample size, age, and gestational age. The main clinical indexes contained preterm delivery (PD), preeclampsia, pregnancy-induced hypertension (PIH), low birthweight, intrauterine growth retardation (IUIG), early PD, anti-hypertensive therapy, gestational diabetes, and body length, body weight and head circumference at birth. The Jadad scoring checklist was used to appraise the quality of the included studies. We evaluated all the RCTs by the following five items: statement of randomization; appropriateness of generating a randomized sequence; use of double blinding; description of double blinding method; details of withdrawals and dropouts. Studies with a score < 3 indicated low quality and high bias risk, studies with a score > 3 indicated high quality. Data were extracted by two investigators independently.
Statistical analysis
All the data analysis was conducted using STATA v10.0 (TX, USA). The heterogeneity of the included RCTs was assessed by χ2 and I2 tests, and the fixed-effects or random-effects models were selected by the above results. When the included RCTs were of high heterogeneity (χ2 p ≤ 0.05 and I2 > 50%), we selected the random-effects model to analyze the indexes. When the included RCTs were of acceptable heterogeneity (χ2 p > 0.05 and I2 ≤ 50%), we selected the fixed-effects model to analyze the indexes. Mean ± standard deviation was used to describe continuous variables, which were then analyzed by the weighted mean difference (WMD). Percentage was used to describe categorical variables, which were analyzed by relative risk (RR). Body length, body weight and head circumference at birth were analyzed by WMD while other indexes were analyzed by RR. Multiple complementary methods (funnel plots, filled funnel plots, Begg’s and Mazumdar’s rank test, and Egger’s test) were used to assess the study quality and risk of bias. Sensitivity analysis was also performed to analyze the indexes.
Results
Overview of the included studies
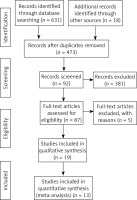
Finally, 649 articles were identified through search using initial keywords. After reviewing the titles and abstracts, we excluded 562 articles. The remaining 87 articles were evaluated by reading the full text, and then 74 articles were excluded using the selection criteria. The studies were excluded for the following reasons: no qualified studies (21), lack of clinical outcomes (27), review or theory research (8), and non-RCT studies (18). Finally, 13 studies [8, 10–22] with 9069 pregnant women that met the eligibility criteria were included in this meta-analysis. The article screening process is presented in Figure 1. The basic information of each study is summarized in Table I. The main Jadad score of the included studies was 4.46, and the main score was higher than 3, indicating that the 13 included RCTs were of high quality.
Table I
Basic characteristics of included studies
Pregnancy outcomes
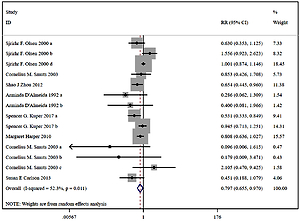
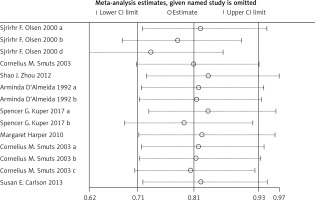
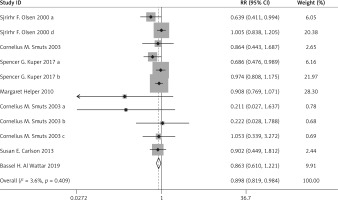
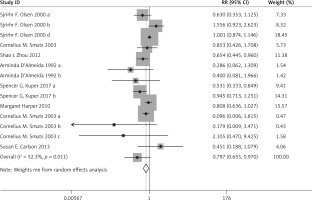
Compared with the control group, n-3 PUFA significantly decreased the incidence of PD (RR = 0.898, 95% CI: 0.819–0.984), and low birthweight (RR = 0.797, 95% CI: 0.655–0.970). Sensitivity analysis of low birthweight is presented in Figure 2.
Compared with the control group, n-3 PUFA significantly increased the body weight (WMD = 99.340, 95% CI: 10.503–188.177) and body length at birth (WMD = 0.449, 95% CI: 0.236–0.663).
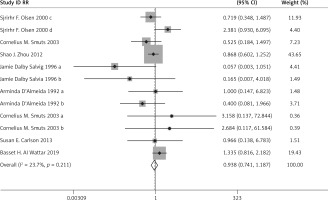
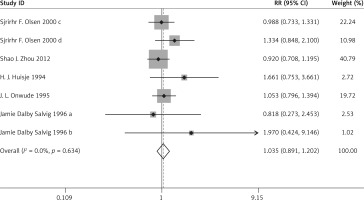
There was no significant difference in the incidence of PIH (RR = 1.035, 95% CI: 0.891–1.202), preeclampsia (RR = 0.938, 95% CI: 0.741–1.187), IUIG (RR = 1.131, 95% CI: 0.957–1.337), early PD (RR = 0.603, 95% CI: 0.263–1.383), the rate of anti-hypertensive therapy (RR = 1.070, 95% CI: 0.518–2.210), gestational diabetes (RR = 0.869, 95% CI: 0.731–1.033), or head circumference at birth (WMD = 0.295, 95% CI: –0.005–0.595) between the two groups.
The above results are presented in Figures 3–6 and Table II.
Table II
Other results of meta-analysis
| Index | N (case/control) | ES (95%CI) | P* | I2 | P# | P-value | |
|---|---|---|---|---|---|---|---|
| Begg’s | Egger’s | ||||||
| Intrauterine growth retardation IUIG | 685/689 | 1.131 (0.957, 1.337)a | 0.835 | 0.0% | 0.150 | 0.999 | – |
| Early PD | 394/403 | 0.603 (0.263, 1.383)a | 0.101 | 62.7% | 0.232 | 0.999 | – |
| Anti-hypertensive therapy | 550/574 | 1.070 (0.518, 2.210)a | 0.038 | 64.3% | 0.855 | 0.999 | 0.704 |
| Gestational diabetes | 2108/2137 | 0.869 (0.731, 1.033)a | 0.122 | 38.6% | 0.112 | 0.999 | 0.324 |
| Birth weight [g] | 2189/2207 | 99.340 (10.503, 188.177)b | < 0.001 | 96.0% | 0.028 | 0.350 | 0.374 |
| Birth length [cm] | 1572/1574 | 0.449 (0.236, 0.663)b | 0.823 | 0.0% | < 0.001 | 0.999 | 0.063 |
| Birth head circumference [cm] | 1572/1574 | 0.295 (–0.005, 0.595)b | 0.052 | 54.3% | 0.054 | 0.999 | 0.508 |
Quality and bias assessment
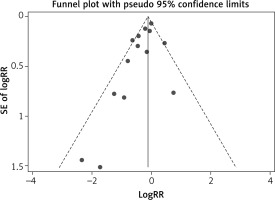
Multiple complementary methods (funnel plots, Begg’s and Mazumdar’s rank test, and Egger’s test) were used to assess the study quality and risk of bias. The funnel plot was based on the log RR funnel plot for low birthweight for all these studies (Figure 7), which showed a clear symmetry, indicating a low publication bias. There still was no significant bias risk of the included studies by Begg’s and Mazumdar’s rank test (Z = 0.99, p = 0.024) or Egger’s test (p = 0.042). The filled funnel plot was based on the log RR funnel plot for low birthweight for all these studies (Figure 8), which also showed a clear symmetry, indicating a low publication bias (Q = 26.828, p = 0.013).
Discussion
Compared with the control group, n-3 PUFA significantly decreased the incidence of PD and low birthweight while increasing birth weight and birth length. In addition, these significant efficacies were all associated with neonatal health status. Maternal deficiency of n-3PUFA during pregnancy can lead to fetal growth retardation in utero, which is closely related to low birth weight. Body weight, body length and head circumference are important indicators of infant physical development. Weight can reflect recent nutritional status, body length reflects the long-term nutritional status, and head circumference reflects the development of the brain and skull. If the growth of the physical development index is slow, it indicates that the nutrition and energy needed for growth and development may be insufficient. The incidence of eclampsia was reduced, and this difference was considered to be related to a reduced inflammatory response by n-3 PUFA. However, there was no significant difference in PIH, preeclampsia, IUIG, early PD, anti-hypertensive therapy, gestational diabetes, or head circumference at birth between the two groups. These results were all related to maternal pregnancy outcomes, so there still need to be more high-quality studies to confirm the efficacy of n-3 PUFA in maternal pregnancy outcomes.
N-3 PUFA contains a variety of polyunsaturated fatty acids starting from the methyl end and having the first unsaturated double bond between the third and fourth carbon atoms. It mainly includes α-linolenic acid, EPA and DHA. α-linolenic acid is mainly found in vegetable oils, such as flaxseed oil and chory oil. EPA and DHA, on the other hand, are found mainly in fish oils. DHA is the most abundant polyunsaturated fatty acid in retinal photoreceptors, which is necessary to maintain the normal function of rhodopsin, and also promotes fetal brain development. EPA can reduce cholesterols and triglycerides, reduce blood viscosity, and prevent atherosclerosis and other cardiovascular diseases. Since n-3 PUFA cannot be synthesized in the human body, it must be obtained from food, and pregnancy is a period when the demand for various trace elements increases significantly, so we should eat more fish, especially deep-sea fish, to obtain sufficient n-3 PUFA.
Considering the effect of n-3 PUFA on blood lipid and cardiovascular diseases, as well as its ability to reduce inflammation, patients with diabetes are also recommended to eat more fish to increase the intake of n-3 PUFA. The Chinese Medical Nutrition Guidelines for Diabetes Treatment recommends that n-3 PUFA in the vegetable oil in the diet should be increased. Daily intake of 3.5 g n-3 PUFA can significantly reduce the level of TG in diabetes patients. There is no evidence to recommend whether pregnant women should take supplements of n-3 PUFA or at which specific doses, and there is some debate about the effects of n-3 supplementation. Pregnancy induces changes in the cardiovascular system in order to meet the increased metabolic demands of the mother and fetus, but there are no recommendations about the prevention and management of lipid disorders [23]. Some meta-analyses [24, 25] found that statin therapy would not increase the incidence of birth defects, and an increasing number of pregnant women at high CVD risk are in need of statin therapy. One study [26] also suggested that vitamin D supplementation may be useful in preventing preeclampsia. In the present study, n-3 PUFA had no significant influence on the incidence of PIH, anti-hypertensive therapy and gestational diabetes compared with the control group. Finally, the meta-analysis showed no significant advantage of n-3 PUFA in reducing the incidence of gestational diabetes mellitus and hypertension.
However, the present meta-analysis has some limitations; for example, the number of included RCTs was limited. The exclusion and inclusion criteria were different in different RCTs. Moreover, the results of the study were influenced by multiple factors such as the active ingredient of n-3 PUFA preparation, dosage, duration of intervention and the supplementation of other nutrients in pregnant women’s daily life. Finally, our analysis was based on secondary data as the original data were not available, which precluded in-depth analyses.
In conclusion, this meta-analysis shows that n-3 PUFA is not beneficial in reducing the incidence of maternal pregnancy outcomes such as gestational diabetes mellitus and hypertension, but it is beneficial in neonatal health status.