Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
DIABETOLOGY / RESEARCH PAPER
FTO promotes ferroptosis through m6A-dependent downregulation of HSPG2 expression in endothelial cell metabolic memory and diabetic nephropathy
1
Department of Nephrology, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, China
2
Department of Nephrology, Kashgar Prefecture Second People's Hospital, China
Submission date: 2025-04-14
Final revision date: 2026-01-13
Acceptance date: 2026-02-05
Online publication date: 2026-05-01
Corresponding author
Man Yang
Department of Nephrology, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, 200080, China
Department of Nephrology, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, 200080, China
KEYWORDS
TOPICS
ABSTRACT
Introduction:
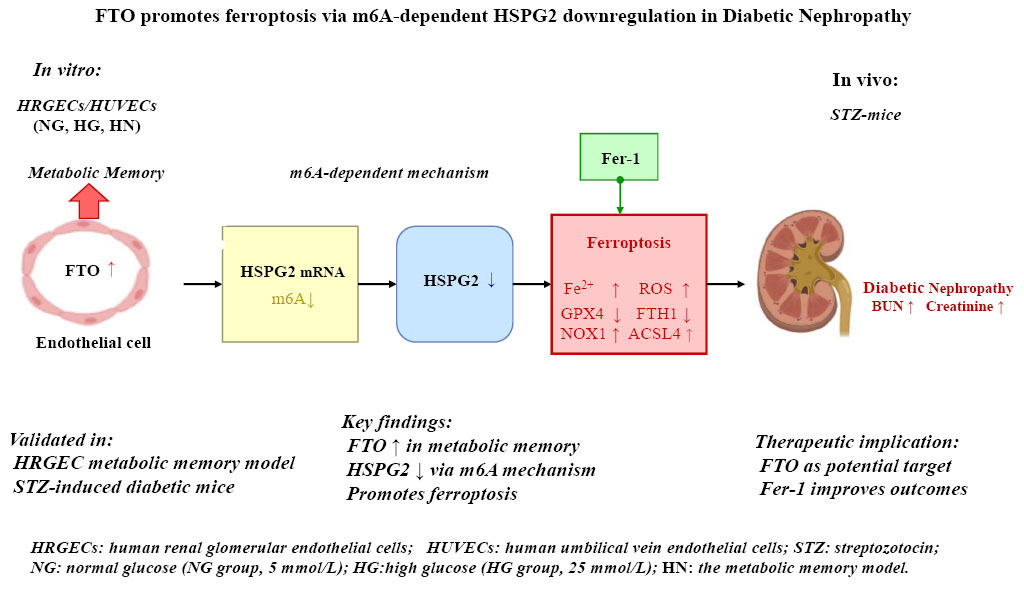
Metabolic memory represents a major challenge in diabetic nephropathy (DN), yet the involvement of m6A RNA modification and ferroptosis remains unclear.
Material and methods:
Human endothelial cell lines (HUVECs and HRGECs) were used to establish an in vitro model of metabolic memory, alongside a murine model of DN induced by streptozotocin. MeRIP-seq was performed to profile m6A modification in total RNA, and MeRIP-qPCR was conducted to validate m6A enrichment in HSPG2. In HRGECs, gain- and loss-of-function studies for FTO and HSPG2 were carried out prior to induction of metabolic memory. The expression of ferroptosis-related proteins, FTO, and HSPG2 was assessed using real-time PCR, western blotting, and cellular immunofluorescence. Iron dyshomeostasis and oxidative stress were evaluated using commercial kits. Serum samples were analyzed for blood glucose and renal function.
Results:
Dysregulation of RNA m6A modification was observed in the endothelial cell metabolic memory model, with a significant downregulation of m6A methylation in HSPG2. Both in vitro and in vivo models exhibited reduced m6A levels and decreased mRNA and protein expression of HSPG2. In contrast, FTO expression was markedly upregulated. Functional analyses demonstrated that FTO suppressed HSPG2 expression in an m6A-dependent manner, thereby promoting ferroptosis. In the mouse model of DN, ferroptosis inhibitor Fer-1 blocked ferroptosis and improved blood glucose and kidney function parameters.
Conclusions:
FTO promotes ferroptosis by downregulating HSPG2 expression in an m6A-dependent mechanism and is critically involved in the pathogenesis of endothelial cell metabolic memory and DN. These findings highlight FTO as a potential therapeutic target for DN.
Metabolic memory represents a major challenge in diabetic nephropathy (DN), yet the involvement of m6A RNA modification and ferroptosis remains unclear.
Material and methods:
Human endothelial cell lines (HUVECs and HRGECs) were used to establish an in vitro model of metabolic memory, alongside a murine model of DN induced by streptozotocin. MeRIP-seq was performed to profile m6A modification in total RNA, and MeRIP-qPCR was conducted to validate m6A enrichment in HSPG2. In HRGECs, gain- and loss-of-function studies for FTO and HSPG2 were carried out prior to induction of metabolic memory. The expression of ferroptosis-related proteins, FTO, and HSPG2 was assessed using real-time PCR, western blotting, and cellular immunofluorescence. Iron dyshomeostasis and oxidative stress were evaluated using commercial kits. Serum samples were analyzed for blood glucose and renal function.
Results:
Dysregulation of RNA m6A modification was observed in the endothelial cell metabolic memory model, with a significant downregulation of m6A methylation in HSPG2. Both in vitro and in vivo models exhibited reduced m6A levels and decreased mRNA and protein expression of HSPG2. In contrast, FTO expression was markedly upregulated. Functional analyses demonstrated that FTO suppressed HSPG2 expression in an m6A-dependent manner, thereby promoting ferroptosis. In the mouse model of DN, ferroptosis inhibitor Fer-1 blocked ferroptosis and improved blood glucose and kidney function parameters.
Conclusions:
FTO promotes ferroptosis by downregulating HSPG2 expression in an m6A-dependent mechanism and is critically involved in the pathogenesis of endothelial cell metabolic memory and DN. These findings highlight FTO as a potential therapeutic target for DN.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.