Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
NEUROLOGY / EXPERIMENTAL RESEARCH
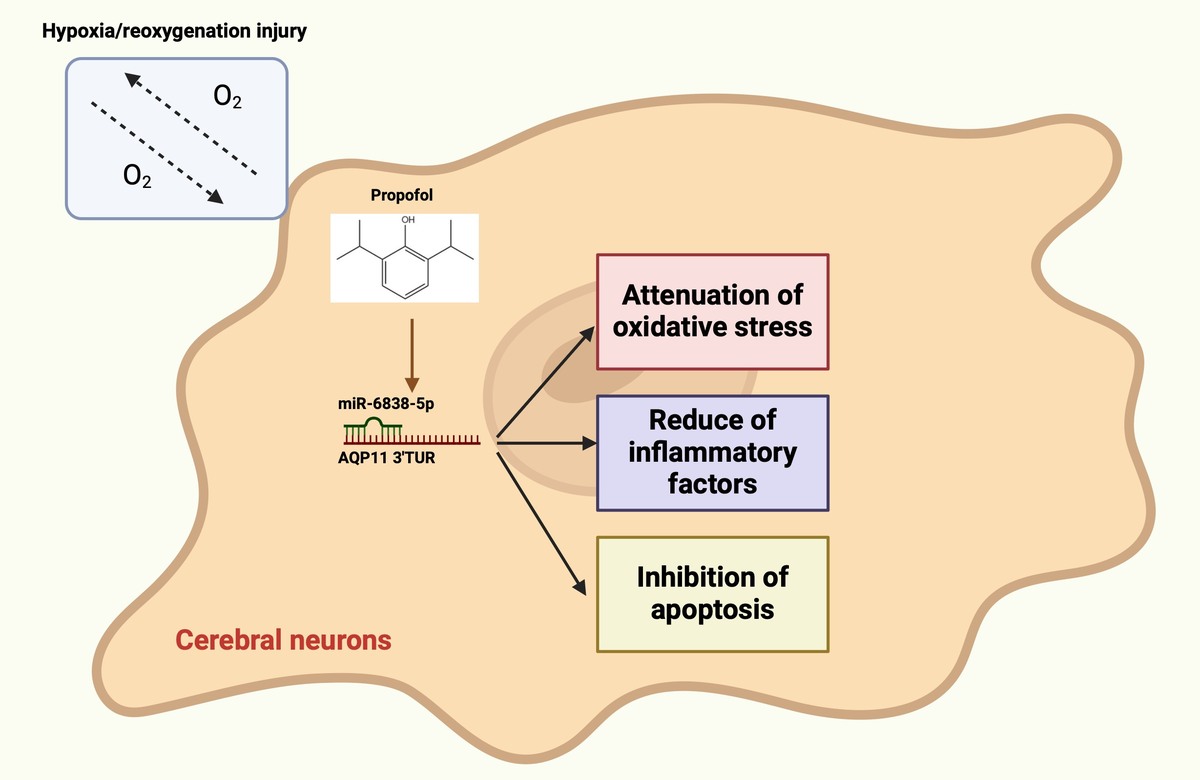
Propofol attenuates cerebral ischemia–reperfusion-mediated neuronal apoptosis and oxidative damage by regulating the miR-6838-5p/AQP11 axis
1
Department of Anesthesiology, Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences, China
2
Department of Anesthesiology, The 960th Hospital of the PLA, China
Submission date: 2024-11-21
Final revision date: 2025-01-23
Acceptance date: 2025-02-10
Online publication date: 2025-04-20
Publication date: 2026-04-30
Corresponding author
XiuQin Wang
Department of Anesthesiology, Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences, 250117, Jinan City, China
Department of Anesthesiology, Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences, 250117, Jinan City, China
Arch Med Sci 2026;22(2):1183-1205
KEYWORDS
TOPICS
ABSTRACT
Introduction:
This study aimed to elucidate the mechanism by which propofol (PPF) exerts its effects in cerebral ischemia–reperfusion injury (CI/RI).
Material and methods:
A rat model of CI/RI was established via middle cerebral artery occlusion/reperfusion (MCAO/R). MCAO/R rats were pre-treated with PPF (10 mg/kg) via intraperitoneal injection. Additionally, 48 h before PPF administration, miR-6838-5p agomir/antagomir and aquaporin-11 (AQP11) lentiviral overexpression vectors were injected into MCAO/R rats. Infarct size was determined using 2,3,5-triphenyl tetrazolium chloride staining. Neurological function was assessed using standardized scoring, and cerebral edema was measured by determining brain water content. Hematoxylin-eosin staining, Nissl staining, and terminal deoxynucleotidyl transferase dUTP nick end labeling staining were performed on brain tissues. Inflammatory and oxidative markers were evaluated. A hypoxia/reoxygenation (H/R) injury model was established in PC12 cells to assess miR-6838-5p and AQP11 expression levels, as well as cell viability and apoptosis.
Results:
The lethal dose 50 (LD50) of PPF in rats was determined to be 22.6 mg/kg, significantly higher than the therapeutic dose. PPF or overexpression of miR-6838-5p resulted in reduced cerebral infarct size, neuronal necrosis, and apoptosis, increased Nissl bodies, and decreased brain edema, apoptosis, tumor necrosis factor-a, interleukin-1b, and malondialdehyde (MDA) levels. Glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD) activities were elevated in MCAO/R rats treated with PPF. These protective effects of PPF were reversed by miR-6838-5p knockdown or AQP11 overexpression. PPF ameliorated H/R-induced neuronal damage, enhanced neuronal activity, reduced apoptosis and MDA production, and increased GSH-Px and SOD levels.
Conclusions:
PPF ameliorates CI/RI by modulating the miR-6838-5p/AQP11 axis.
This study aimed to elucidate the mechanism by which propofol (PPF) exerts its effects in cerebral ischemia–reperfusion injury (CI/RI).
Material and methods:
A rat model of CI/RI was established via middle cerebral artery occlusion/reperfusion (MCAO/R). MCAO/R rats were pre-treated with PPF (10 mg/kg) via intraperitoneal injection. Additionally, 48 h before PPF administration, miR-6838-5p agomir/antagomir and aquaporin-11 (AQP11) lentiviral overexpression vectors were injected into MCAO/R rats. Infarct size was determined using 2,3,5-triphenyl tetrazolium chloride staining. Neurological function was assessed using standardized scoring, and cerebral edema was measured by determining brain water content. Hematoxylin-eosin staining, Nissl staining, and terminal deoxynucleotidyl transferase dUTP nick end labeling staining were performed on brain tissues. Inflammatory and oxidative markers were evaluated. A hypoxia/reoxygenation (H/R) injury model was established in PC12 cells to assess miR-6838-5p and AQP11 expression levels, as well as cell viability and apoptosis.
Results:
The lethal dose 50 (LD50) of PPF in rats was determined to be 22.6 mg/kg, significantly higher than the therapeutic dose. PPF or overexpression of miR-6838-5p resulted in reduced cerebral infarct size, neuronal necrosis, and apoptosis, increased Nissl bodies, and decreased brain edema, apoptosis, tumor necrosis factor-a, interleukin-1b, and malondialdehyde (MDA) levels. Glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD) activities were elevated in MCAO/R rats treated with PPF. These protective effects of PPF were reversed by miR-6838-5p knockdown or AQP11 overexpression. PPF ameliorated H/R-induced neuronal damage, enhanced neuronal activity, reduced apoptosis and MDA production, and increased GSH-Px and SOD levels.
Conclusions:
PPF ameliorates CI/RI by modulating the miR-6838-5p/AQP11 axis.
REFERENCES (53)
1.
Benjamin EJ, Blaha MJ, Chiuve SE, et al. Heart Disease and Stroke Statistics-2017 Update: a report from the American Heart Association. Circulation 2017; 135: e146-603.
2.
Lo WL, Mao YR, Li L, et al. Prospective clinical study of rehabilitation interventions with multisensory interactive training in patients with cerebral infarction: study protocol for a randomised controlled trial. Trials 2017; 18: 173.
4.
Obadia N, Lessa MA, Daliry A, et al. Cerebral microvascular dysfunction in metabolic syndrome is exacerbated by ischemia-reperfusion injury. BMC Neurosci 2017; 18: 67.
5.
Xue X, Wang H, Su J. Inhibition of MiR-122 decreases cerebral ischemia-reperfusion injury by upregulating DJ-1-phosphatase and tensin homologue deleted on chromosome 10 (PTEN)/phosphonosinol-3 kinase (PI3K)/AKT. Med Sci Monit 2020; 26: e915825.
6.
Zhou X, Wang Z, Xu B, et al. Long non-coding RNA NORAD protects against cerebral ischemia/reperfusion injury induced brain damage, cell apoptosis, oxidative stress and inflammation by regulating miR-30a-5p/YWHAG. Bioengineered 2021; 12: 9174-88.
7.
Wu MY, Yiang GT, Liao WT, et al. Current mechanistic concepts in ischemia and reperfusion injury. Cell Physiol Biochem 2018; 46: 1650-67.
8.
Herpich F, Rincon F. Management of acute ischemic stroke. Crit Care Med 2020; 48: 1654-63.
9.
Amani H, Mostafavi E, Alebouyeh MR, et al. Would colloidal gold nanocarriers present an effective diagnosis or treatment for ischemic stroke? Int J Nanomed 2019; 14: 8013-31.
10.
Zhu XN, Li J, Qiu GL, et al. Propofol exerts anti-anhedonia effects via inhibiting the dopamine transporter. Neuron 2023; 111: 1626-36 e6.
11.
Guo XN, Ma X. The effects of propofol on autophagy. DNA Cell Biol 2020; 39: 197-209.
12.
Fan W, Zhu X, Wu L, et al. Propofol: an anesthetic possessing neuroprotective effects. Eur Rev Med Pharmacol Sci 2015; 19: 1520-9.
13.
Tao W, Zhang X, Ding J, et al. The effect of propofol on hypoxia- and TNF-a-mediated BDNF/TrkB pathway dysregulation in primary rat hippocampal neurons. CNS Neurosci Ther 2022; 28: 761-74.
14.
Engelhard K, Werner C, Eberspächer E, et al. Influence of propofol on neuronal damage and apoptotic factors after incomplete cerebral ischemia and reperfusion in rats: a long-term observation. Anesthesiology 2004; 101: 912-7.
15.
Huang L, Ding L, Yu S, Huang X, Ren Q. Propofol postconditioning alleviates diabetic myocardial ischemia‑reperfusion injury via the miR‑200c‑3p/AdipoR2/STAT3 signaling pathway. Mol Med Rep 2022; 25: 137.
16.
Zhou R, Yang Z, Tang X, Tan Y, Wu X, Liu F. Propofol protects against focal cerebral ischemia via inhibition of microglia-mediated proinflammatory cytokines in a rat model of experimental stroke. PLoS One 2013; 8: e82729.
17.
Gao X, Mi Y, Guo N, et al. The mechanism of propofol in cancer development: an updated review. Asia Pac J Clin Oncol 2020; 16: e3-11.
18.
Feng L, Sun ZG, Liu QW, et al. Propofol inhibits the expression of Abelson nonreceptor tyrosine kinase without affecting learning or memory function in neonatal rats. Brain Behav 2020; 10: e01810.
19.
Romuk EB, Szczurek W, Nowak PG, Hudziec E, Chwalińska E, Birkner E. Effects of propofol on the liver oxidative-antioxidant balance in a rat model of Parkinson’s disease. Adv Clin Exp Med 2016; 25: 815-20.
20.
Yu W, Gao D, Jin W, Liu S, Qi S. Propofol prevents oxidative stress by decreasing the ischemic accumulation of succinate in focal cerebral ischemia-reperfusion injury. Neurochem Res 2018; 43: 420-9.
21.
Jia L, Wang F, Gu X, et al. Propofol postconditioning attenuates hippocampus ischemia-reperfusion injury via modulating JAK2/STAT3 pathway in rats after autogenous orthotropic liver transplantation. Brain Res 2017; 1657: 202-7.
22.
Chen Y, Li Z. Protective effects of propofol on rats with cerebral ischemia-reperfusion injury via the PI3K/Akt pathway. J Mol Neurosci 2021; 71: 810-20.
23.
Hausburg MA, Banton KL, Roman PE, et al. Effects of propofol on ischemia-reperfusion and traumatic brain injury. J Crit Care 2020; 56: 281-7.
24.
Zhang Y, Yuan X, Xu J, Gu H. CircRBM33 induces endothelial dysfunction by targeting the miR-6838-5p/PDCD4 axis affecting blood-brain barrier in mice with cerebral ischemia-reperfusion injury. Clin Hemorheol Microcirc 2023; 85: 355-70.
25.
Morishita Y, Matsuzaki T, Hara-chikuma M, et al. Disruption of aquaporin-11 produces polycystic kidneys following vacuolization of the proximal tubule. Mol Cell Biol 2005; 25: 7770-9.
26.
Gorelick DA, Praetorius J, Tsunenari T, et al. Aquaporin-11: a channel protein lacking apparent transport function expressed in brain. BMC Biochem 2006; 7: 14.
27.
Zuo ML, Wang AP, Song GL, Yang ZB. miR-652 protects rats from cerebral ischemia/reperfusion oxidative stress injury by directly targeting NOX2. Biomed Pharmacother 2020; 124: 109860.
28.
Sun B, Ou H, Ren F, Guan Y, Huan Y, Cai H. Propofol protects against cerebral ischemia/reperfusion injury by down-regulating long noncoding RNA SNHG14. ACS Chem Neurosci 2021; 12: 3002-14.
29.
Liang C, Cang J, Wang H, Xue Z. Propofol attenuates cerebral ischemia/reperfusion injury partially using heme oxygenase-1. J Neurosurg Anesthesiol 2013; 25: 311-6.
30.
Sun B, Ou H, Ren F, et al. Propofol protects against cerebral ischemia/reperfusion injury by down-regulating long noncoding RNA SNHG14. ACS Chem Neurosci 2021; 12: 3002-14.
31.
Lu Y, Han Y, He J, Zhou B, Fang P, Li X. LncRNA FOXD3-AS1 knockdown protects against cerebral ischemia/reperfusion injury via miR-765/BCL2L13 axis. Biomed Pharmacother 2020; 132: 110778.
32.
Wang C, Hu F. Long noncoding RNA SOX2OT silencing alleviates cerebral ischemia-reperfusion injury via miR-135a-5p-mediated NR3C2 inhibition. Brain Res Bull 2021; 173: 193-202.
33.
Yang B, Zang LE, Cui JW, Zhang MY, Ma X, Wei LL. Melatonin plays a protective role by regulating miR-26a-5p-NRSF and JAK2-STAT3 pathway to improve autophagy, inflammation and oxidative stress of cerebral ischemia-reperfusion injury. Drug Des Devel Ther 2020; 14: 3177-88.
34.
Hu Y, Ye C, Cheng S, Chen J. Propofol downregulates lncRNA MALAT1 to alleviate cerebral ischemia-reperfusion injury. Inflammation 2021; 44: 2580-91.
35.
Chen C, Chang X, Zhang S, Zhao Q, Lei C. CircRNA CTNNB1 (circCTNNB1) ameliorates cerebral ischemia/reperfusion injury by sponging miR-96-5p to up-regulate scavenger receptor class B type 1 (SRB1) expression. Bioengineered 2022; 13: 10258-73.
36.
Li J, He W, Wang Y, Zhao J, Zhao X. miR-103a-3p alleviates oxidative stress, apoptosis, and immune disorder in oxygen-glucose deprivation-treated BV2 microglial cells and rats with cerebral ischemia-reperfusion injury by targeting high mobility group box 1. Ann Transl Med 2020; 8: 1296.
37.
Kuai F, Zhou L, Zhou J, Sun X, Dong W. Long non-coding RNA THRIL inhibits miRNA-24-3p to upregulate neuropilin-1 to aggravate cerebral ischemia-reperfusion injury through regulating the nuclear factor kB p65 signaling. Aging 2021; 13: 9071-84.
38.
Wang B, Chen S, Yang J, Yang L, Liu J, Zhang W. ET-26 hydrochloride (ET-26 HCl) has similar hemodynamic stability to that of etomidate in normal and uncontrolled hemorrhagic shock (UHS) rats. PLoS One 2017; 12: e0183439.
39.
Liu H, Wu X, Luo J, et al. Adiponectin peptide alleviates oxidative stress and NLRP3 inflammasome activation after cerebral ischemia-reperfusion injury by regulating AMPK/GSK-3b. Exp Neurol 2020; 329: 113302.
40.
Liang Q, Yang J, He J, et al. Stigmasterol alleviates cerebral ischemia/reperfusion injury by attenuating inflammation and improving antioxidant defenses in rats. Biosci Rep 2020; 40: BSR20192133.
41.
Leech T, Chattipakorn N, Chattipakorn SC. The beneficial roles of metformin on the brain with cerebral ischaemia/reperfusion injury. Pharmacol Res 2019; 146: 104261.
43.
Xing F, Liu Y, Dong R, Cheng Y. miR-374 improves cerebral ischemia reperfusion injury by targeting Wnt5a. Exp Anim 2021; 70: 126-36.
44.
Lu MY, Wu JR, Liang RB, et al. Upregulation of miR-219a-5p decreases cerebral ischemia/reperfusion injury in vitro by targeting Pde4d. J Stroke Cerebrovasc Dis 2020; 29: 104801.
45.
Jiang F, Liu X, Wang X, Hu J, Chang S, Cui X. LncRNA FGD5-AS1 accelerates intracerebral hemorrhage injury in mice by adsorbing miR-6838-5p to target VEGFA. Brain Res 2022; 1776: 147751.
46.
Zhang G, Ding L, Sun G, et al. LncRNA AZIN1-AS1 ameliorates myocardial ischemia-reperfusion injury by targeting miR-6838-5p/WNT3A axis to activate Wnt-b/catenin signaling pathway. In Vitro Cell Dev Biol Anim 2022; 58: 54-68.
47.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116: 281-97.
48.
Koike S, Tanaka Y, Matsuzaki T, Morishita Y, Ishibashi K. Aquaporin-11 (AQP11) expression in the mouse brain. Int J Mol Sci 2016; 17: 861.
49.
Xi T, Jin F, Zhu Y, et al. miR-27a-3p protects against blood-brain barrier disruption and brain injury after intracerebral hemorrhage by targeting endothelial aquaporin-11. J Biol Chem 2018; 293: 20041-50.
50.
Su G, Qu Y, Li G, Deng M. Sevoflurane protects against cerebral ischemia/reperfusion injury via microrna-30c-5p modulating homeodomain-interacting protein kinase 1. Bioengineered 2021; 12: 11858-71.
51.
Yan K, Hu C, Liu C, et al. LncRNA CERS6-AS1, sponging miR-6838-5p, promotes proliferation and invasion in cervical carcinoma cells by upregulating FOXP2. Histol Histopathol 2023; 38: 823-35.
52.
Zhao X, Zhu X, Xiao C, Hu Z. LncRNA SH3BP5-AS1 promotes hepatocellular carcinoma progression by sponging miR-6838-5p and activation of PTPN4. Aging 2024; 16: 8511-23.
53.
Krajčová A, Waldauf P, Anděl M, Duška F. Propofol infusion syndrome: a structured review of experimental studies and 153 published case reports. Crit Care 2015; 19: 398.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.