Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
LIPID DISORDERS / STATE OF THE ART PAPER
Understanding the link between lipid and ocular disorders for effective therapy
1
Faculty of Pharmacy and Health Sciences, Universiti Kuala Lumpur Royal College of Medicine Perak, Malaysia
2
Department of Biochemistry, Faculty of Medicine, Manipal University College, Malaysia
3
Optometry and Vision Science Programme, Center for Community Health Studies (ReaCH), Faculty of Health Sciences, Universiti Kebangsaan Malaysia, Jalan Raja Muda, Abdul Aziz, Kuala Lumpur, Malaysia
These authors had equal contribution to this work
Submission date: 2025-10-09
Final revision date: 2025-12-24
Acceptance date: 2026-01-04
Online publication date: 2026-04-03
Publication date: 2026-04-30
Corresponding author
Lidawani Lambuk
Optometry and Vision Science Programme Center for Community Health Studies (ReaCH) Faculty of Health Sciences Universiti Kebangsaan Malaysia Jalan Raja Muda Abdul Aziz Kuala Lumpur, Malaysia
Optometry and Vision Science Programme Center for Community Health Studies (ReaCH) Faculty of Health Sciences Universiti Kebangsaan Malaysia Jalan Raja Muda Abdul Aziz Kuala Lumpur, Malaysia
Arch Med Sci 2026;22(2):583-601
KEYWORDS
TOPICS
ABSTRACT
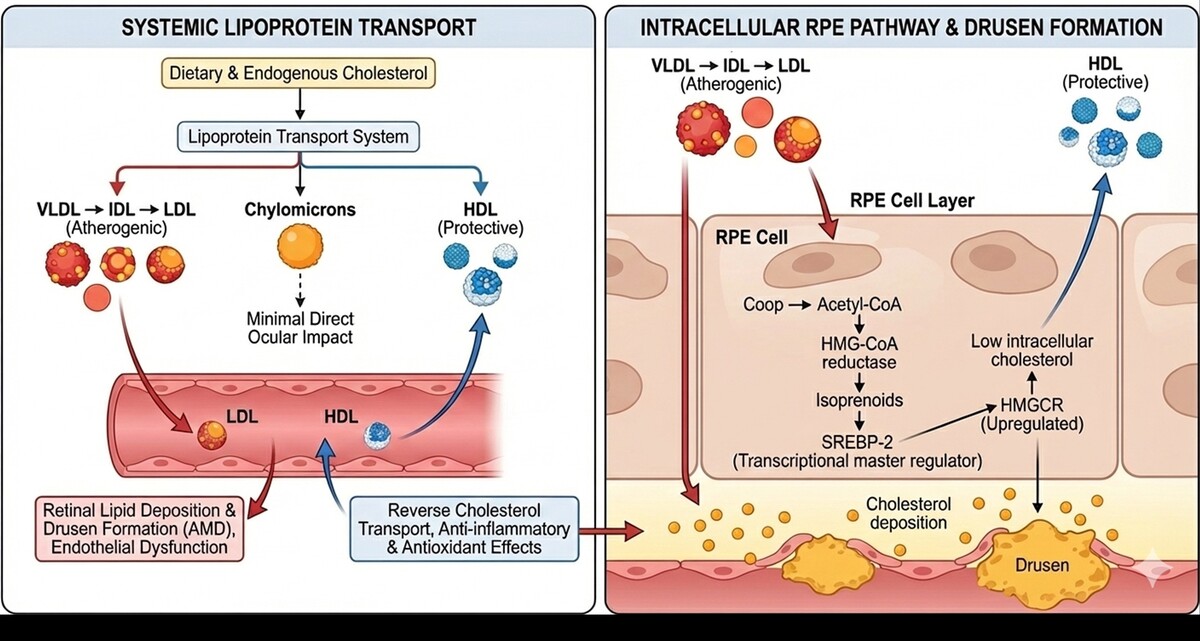
Cholesterol is an essential lipid for cellular integrity and metabolic homeostasis; however, dysregulated cholesterol metabolism is increasingly recognized as a key contributor to multiple ocular diseases. Beyond its established role in cardiovascular pathology, emerging evidence implicates altered lipoprotein balance, impaired cholesterol transport, and lipid-driven inflammation in the pathogenesis of age-related macular degeneration, retinal vascular diseases, cataracts, and glaucoma. This review integrates current knowledge on cholesterol synthesis, transport, and regulation with ocular-specific mechanisms, emphasizing the differential roles of low-density lipoprotein, high-density lipoprotein, and triglyceride-rich lipoproteins. Key molecular pathways, including the mevalonate-SREBP axis, ABCA1/ABCG1-mediated reverse cholesterol transport, VEGF-driven angiogenesis, oxidative stress signaling, and inflammatory cascades, are critically discussed. Clinical and translational implications are highlighted, particularly the potential ocular benefits of lipid-modulating therapies such as statins, fenofibrate, and antioxidant supplementation. Understanding cholesterol as a modifiable determinant of ocular health may enable integrated preventive strategies and guide future therapeutic innovation in vision-threatening diseases.
REFERENCES (139)
1.
Guo J, Chen S, Zhang Y, et al. Cholesterol metabolism: physiological regulation and diseases. MedComm 2024; 5: e476.
2.
Mauricio D, Castelblanco E, Alonso N. Cholesterol and inflammation in atherosclerosis: an immune-metabolic hypothesis. Nutrients 2020; 12: 2444.
3.
Bhale AS, Venkataraman K. Cholesterol transport by various lipoproteins. In: Lipid Analysis in Biology, Health, and Disease. Bhattacharya SK (ed.). Elsevier 2026; 85-103.
4.
Savulescu-Fiedler I, Dorobantu-Lungu LR, Dragosloveanu S, et al. The cross-talk between the peripheral and brain cholesterol metabolisms. Curr Issues Mol Biol 2025; 47: 115.
5.
Ahmed I. Reevaluating low-cholesterol targets: physiological needs vs. cardiovascular risk. (July 11, 2025). Available at SSRN: https://ssrn.com/abstract=5353... or http://dx.doi.org/10.2139/ssrn... .
6.
Cooper RA. Influence of increased membrane cholesterol on membrane fluidity and cell function in human red blood cells. J Supramol Struct 1978; 8: 413-30.
7.
Krause MR, Regen SL. The structural role of cholesterol in cell membranes: from condensed bilayers to lipid rafts. Acc Chem Res 2014; 47: 3512-21.
8.
Subczynski WK, Pasenkiewicz-Gierula M, Widomska J, Mainali L, Raguz M. High cholesterol/low cholesterol: effects in biological membranes: a review. Cell Biochem Biophys 2017; 75: 369-85.
9.
Schade DS, Shey L, Eaton RP. Cholesterol review: a metabolically important molecule. Endocr Pract 2020; 26: 1514-23.
10.
Li Y, Zhao Q, Wang Y, et al. Lipid droplet accumulation in microglia and their potential roles. Lipids Health Dis 2025; 24: 215.
11.
Warda M, Tekin S, Gamal M, Khafaga N, Çelebi F, Tarantino G. Lipid rafts: novel therapeutic targets for metabolic, neurodegenerative, oncological, and cardiovascular diseases. Lipids Health Dis 2025; 24: 147.
12.
Yang Y, Zhang R, Zhang M, et al. Association between disorders of lipid metabolism and oculopathy: an overview. Int J Med Sci 2025; 22: 3878.
13.
Xia Q, Chen Y, Yu Z, et al. Prevalence, awareness, treatment, and control of dyslipidemia in Chinese adults: a systematic review and meta-analysis. Front Cardiovasc Med 2023; 10: 1186330.
14.
Kumar S, Kaur N, Hitaishi P, Ghosh SK, Mithu VS, Scheidt HA. Role of cholesterol in interaction of ionic liquids with model lipid membranes and associated permeability. J Phys Chem B 2024; 128: 5407-18.
15.
Kamil RM, Ghaib ZJ. Chemical structure, classification and clinical significance of steroid hormones: a review article. Al-Salam J Med Sci 2025; 4: 57-66.
16.
Baranowski ES, Arlt W, Idkowiak J. Monogenic disorders of adrenal steroidogenesis. Horm Res Paediatr 2018; 89: 292-310.
17.
Park GW, Kim H, Won SH, Kim NH, Choi SR. Neurosteroids and neurological disorders. Korean J Physiol Pharmacol 2025; 29: 157-64.
18.
Duan Y, Gong K, Xu S, Zhang F, Meng X, Han J. Regulation of cholesterol homeostasis in health and diseases: from mechanisms to targeted therapeutics. Signal Transd Target Ther 2022; 7: 265.
19.
Shimano H, Sato R. SREBP-regulated lipid metabolism: convergent physiology – divergent pathophysiology. Nat Rev Endocrinol 2017; 13: 710-30.
20.
Lee SH, Lee JH, Im SS. The cellular function of SCAP in metabolic signaling. Exp Mol Med 2020; 52: 724-9.
21.
Madison BB. Srebp2: a master regulator of sterol and fatty acid synthesis. J Lipid Res 2016; 57: 333-5.
22.
Ferré P, Phan F, Foufelle F. SREBP-1c and lipogenesis in the liver: an update. Biochem J 2021; 478: 3723-39.
23.
Sato R. Recent advances in regulating cholesterol and bile acid metabolism. Biosci Biotechnol Biochem 2020; 84: 2185-92.
24.
Jo Y, DeBose-Boyd RA. Post-translational regulation of HMG CoA reductase. Cold Spring Harbor Perspect Biol 2022; 14: a041253.
27.
Lütjohann D, Klör HU, Stellaard F. Measurement of serum low density lipoprotein cholesterol and triglyceride-rich remnant cholesterol as independent predictors of atherosclerotic cardiovascular disease: possibilities and limitations. Nutrients 2023; 15: 2202.
28.
Fan J, Zhu T, Tian X, Wang M, Li Z, Li W. Targeting programmed cell death as an innovative strategy against atherosclerosis: potential of natural compound supplements. Phytother Res 2025; 39: 3664-708.
29.
Baumer Y, Irei J, Boisvert WA. Cholesterol crystals in the pathogenesis of atherosclerosis. Nat Rev Cardiol 2025; 22: 315-32.
30.
Long C, Li Z, Jiang L, et al. Lipid droplet dynamics in type 2 diabetes and its complications: pathophysiological insights and therapeutic options. Lipids Health Dis 2025; 24: 284.
31.
Cochran BJ, Hou L, Manavalan APC, et al. Impact of perturbed pancreatic b-cell cholesterol homeostasis on adipose tissue and skeletal muscle metabolism. Diabetes 2016; 65: 3610-20.
32.
Mordzińska-Rak A, Verdeil G, Hamon Y, Błaszczak E, Trombik T. Dysregulation of cholesterol homeostasis in cancer pathogenesis. Cell Mol Life Sci 2025; 82: 168.
33.
Ramu D, Kim E. Exosomal lipids in cancer progression and metastasis. Cancer Med 2025; 14: e70687.
34.
Wang M, Ge F, Wu C, et al. FASN promotes the stemness of cancer stem cells and protects colorectal cancer cells from ferroptosis by inhibiting the activation of SREBP2. Front Immunol 2025; 16: 1611375.
35.
Brindisi M, Fiorillo M, Frattaruolo L, Sotgia F, Lisanti MP, Cappello AR. Cholesterol and mevalonate: two metabolites involved in breast cancer progression and drug resistance through the ERRa pathway. Cells 2020; 9: 1819.
36.
Mokarram P, Alizadeh J, Razban V, Barazeh M, Solomon C, Kavousipour S. Interconnection of estrogen/testosterone metabolism and mevalonate pathway in breast and prostate cancers. Curr Mol Pharmacol 2017; 10: 86-114.
37.
Mashat RM, Zielinska HA, Holly JM, Perks CM. A role for ER-beta in the effects of low-density lipoprotein cholesterol and 27-hydroxycholesterol on breast cancer progression: involvement of the IGF signalling pathway? Cells 2021; 11: 94.
38.
Vona R, Iessi E, Matarrese P. Role of cholesterol and lipid rafts in cancer signaling: a promising therapeutic opportunity? Front Cell Develop Biol 2021; 9: 622908.
39.
Mohammed HES, Fayed FA, Abbas NA, Mohamed SA. The anticancer potential of statins. Zagazig Univ Med J 2025; 31: 250-4.
40.
Lagunas-Rangel FA, Jonsson J, Jackevica L, Fredriksson R, Dambrova M, Schiöth HB. Statins regulate kinase signaling by causing changes in phosphorylation, rather than through changes in gene expression or direct inhibition: evidence in colorectal cancer. Front Pharmacol 2025; 16: 1653702.
41.
Hendi NN, Nemer GM. SDR42E1 modulates vitamin D absorption and cancer pathogenesis: insights from an in vitro model. Front Endocrinol 2025; 16: 1585859.
42.
Klein R, Myers CE, Lee KE, et al. Oxidized low-density lipoprotein and the incidence of proliferative diabetic retinopathy and clinically significant macular edema determined from fundus photographs. JAMA Ophthalmol 2015; 133: 1054-61.
43.
Ban N, Lee TJ, Sene A, et al. Disrupted cholesterol metabolism promotes age-related photoreceptor neurodegeneration. J Lipid Res 2018; 59: 1414-23.
44.
Storti F, Klee K, Todorova V, et al. Impaired ABCA1/ABCG1-mediated lipid efflux in the mouse retinal pigment epithelium (RPE) leads to retinal degeneration. Elife 2019; 8: e45100.
45.
Jun S, Datta S, Wang L, Pegany R, Cano M, Handa JT. The impact of lipids, lipid oxidation, and inflammation on AMD, and the potential role of miRNAs on lipid metabolism in the RPE. Exp Eye Res 2019; 181: 346-55.
46.
Brewer Jr HB. High-density lipoproteins: a new potential therapeutic target for the prevention of cardiovascular disease. Arterioscler Thromb Vasc Biol 2004; 24: 387-91.
47.
Kushwah N, Bora K, Maurya M, Pavlovich MC, Chen J. Oxidative stress and antioxidants in age-related macular degeneration. Antioxidants 2023; 12: 1379.
48.
Holz FG, Strauss EC, Schmitz-Valckenberg S, van Lookeren Campagne M. Geographic atrophy: clinical features and potential therapeutic approaches. Ophthalmology 2014; 121: 1079-91.
49.
Deschenes M, Kuchel G, Lesk M, Vaucher E. Estradiol treatment increases the Local Retinal Uptake Index (LRUI) of the blood flow tracer 14C–IMP in whole–mount retina from ovariectomized rats. Investig Ophthalmol Visual Sci 2006; 47: 473.
50.
Hammer SS, Dorweiler TF, McFarland D, et al. Cholesterol crystal formation is a unifying pathogenic mechanism in the development of diabetic retinopathy. Diabetologia 2023; 66: 1705-18.
51.
Beatty S, Koh HH, Phil M, Henson D, Boulton M. The role of oxidative stress in the pathogenesis of age-related macular degeneration. Survey Ophthalmol 2000; 45: 115-34.
52.
Pifferi F, Laurent B, Plourde M. Lipid transport and metabolism at the blood-brain interface: implications in health and disease. Front Physiol 2021; 12: 645646.
53.
Abokyi S, To CH, Lam TT, Tse DY. Central role of oxidative stress in age-related macular degeneration: evidence from a review of the molecular mechanisms and animal models. Oxid Med Cell Longev 2020; 2020: 7901270.
54.
Crabb JW. The proteomics of drusen. Cold Spring Harbor Perspect Med 2014; 4: a017194.
55.
Johnson PT, Lewis GP, Talaga KC, et al. Drusen-associated degeneration in the retina. Investig Ophthalmol Visual Sci 2003; 44: 4481-8.
56.
Fleming I. New lipid mediators in retinal angiogenesis and retinopathy. Front Pharmacol 2019; 10: 739.
57.
Lin JB, Halawa OA, Husain D, Miller JW, Vavvas DG. Dyslipidemia in age-related macular degeneration Eye 2022; 36: 312-8.
58.
Roizenblatt M, Naranjit N, Maia M, Gehlbach PL. The question of a role for statins in age-related macular degeneration. Int J Mol Sci 2018; 19: 3688.
59.
Cehofski LJ, Honoré B, Vorum H. A review: proteomics in retinal artery occlusion, retinal vein occlusion, diabetic retinopathy and acquired macular disorders. Int J Mol Sci 2017; 18: 907.
60.
Sivaprasad S, Gupta B, Crosby-Nwaobi R, Evans J. Prevalence of diabetic retinopathy in various ethnic groups: a worldwide perspective. Survey Ophthalmol 2012; 57: 347-70.
61.
Hammer SS, Busik JV. The role of dyslipidemia in diabetic retinopathy. Vision Res 2017; 139: 228-36.
62.
Madsen-Bouterse SA, Kowluru RA. Oxidative stress and diabetic retinopathy: pathophysiological mechanisms and treatment perspectives. Rev Endocr Metab Disord 2008; 9: 315-27.
63.
Chang YC, Wu WC. Dyslipidemia and diabetic retinopathy. The review of diabetic studies: RDS 2013; 10: 121-32.
64.
Jeng CJ, Hsieh YT, Yang CM, Yang CH, Lin CL, Wang IJ. Diabetic retinopathy in patients with dyslipidemia: development and progression. Ophthalmol Retina 2018; 2: 38-45.
65.
Higashi Y. Endothelial function in dyslipidemia: roles of LDL-cholesterol, HDL-cholesterol and triglycerides. Cells 2023; 12: 1293.
66.
Nita M, Grzybowski A. The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxid Med Cell Longev 2016; 2016: 3164734.
67.
Chatterjee S. Oxidative stress, inflammation, and disease. In: Oxidative Stress and Biomaterials. Dziubla T, Butterfield DA (eds.). Elsevier 2016; 35-58.
68.
Grambergs R, Mondal K, Mandal N. Inflammatory ocular diseases and sphingolipid signaling. Bioact Ceramides Health Dis Intertwined Roles Enigmatic Lipids 2019; 1159: 139-52.
69.
Wen Z, He X, Wang J, et al. Hyperlipidemia induces proinflammatory responses by activating STING pathway through IRE1α-XBP1 in retinal endothelial cells. J Nutr Biochem 2023; 112: 109213.
70.
Noonan JE, Jenkins AJ, Ma JX, Keech AC, Wang JJ, Lamoureux EL. An update on the molecular actions of fenofibrate and its clinical effects on diabetic retinopathy and other microvascular end points in patients with diabetes. Diabetes 2013; 62: 3968-75.
71.
Farooqui AA, Farooqui T. Phospholipids, sphingolipids, and cholesterol-derived lipid mediators and their role in neurological disorders. Int J Mol Sci 2024; 25: 10672.
72.
Busik JV. Lipid metabolism dysregulation in diabetic retinopathy. J Lipid Res 2021; 62: 100017.
73.
Kang EYC, Chen TH, Garg SJ, et al. Association of statin therapy with prevention of vision-threatening diabetic retinopathy. JAMA Ophthalmol 2019; 137: 363-71.
74.
Nicholson L, Talks SJ, Amoaku W, Talks K, Sivaprasad S. Retinal vein occlusion (RVO) guideline: executive summary. Eye 2022; 36: 909-12.
75.
Zheng C, Lin Y, Jiang B, et al. Plasma lipid levels and risk of retinal vascular occlusion: a genetic study using Mendelian randomization. Front Endocrinol 2022; 13: 954453.
76.
Pikuleva IA, Curcio CA. Cholesterol in the retina: the best is yet to come. Prog Retin Eye Res 2014; 41: 64-89.
77.
Kim J, Lim DH, Han K, et al. Retinal vein occlusion is associated with low blood high-density lipoprotein cholesterol: a nationwide cohort study. Am J Ophthalmol 2019; 205: 35-42.
78.
Cui MY, Yi X, Zhu DX, Wu J. Identification of differentially expressed genes related to the lipid metabolism of esophageal squamous cell carcinoma by integrated bioinformatics analysis. Curr Oncol 2022; 30: 1-18.
79.
Mason RP, Jacob RF. Characterization of cholesterol crystalline domains in model and biological membranes using X-ray diffraction. Adv Exp Med Biol 2015; 842: 231-45.
80.
Guo Z, Ma X, Zhang RX, Yan H. Oxidative stress, epigenetic regulation and pathological processes of lens epithelial cells underlying diabetic cataract. Adv Ophthalmol Pract Res 2023; 3: 180-6.
81.
Adu-Agyeiwaah, Y. Oral administration of alpha-cyclodextrin corrects diabetes induced retinopathy and bone marrow pathology by modulation of the gut-retina axis. The University of Alabama at Birmingham 2024.
82.
Babizhayev MA. Mitochondria induce oxidative stress, generation of reactive oxygen species and redox state unbalance of the eye lens leading to human cataract formation: disruption of redox lens organization by phospholipid hydroperoxides as a common basis for cataract disease. Cell Biochem Funct 2011; 29: 183-206.
83.
Bang CN, Greve AM, La Cour M, et al. Effect of randomized lipid lowering with simvastatin and ezetimibe on cataract development (from the Simvastatin and Ezetimibe in Aortic Stenosis Study). Am J Cardiol 2015; 116: 1840-4.
84.
Erie JC, Pueringer MR, Brue SM, Chamberlain AM, Hodge DO. Statin use and incident cataract surgery: a case-control study. Ophthal Epidemiol 2016; 23: 40-5.
85.
Shin S, Zhou H, He C, et al. Qki activates Srebp2-mediated cholesterol biosynthesis for maintenance of eye lens transparency. Nat Commun 2021; 12: 3005.
86.
Reyes LP, Reyes TC, Duenas Z, Duran D, Perdomo S, Avila MY. Expression of oxysterols in human lenses: implications of the sterol pathway in age-related cataracts. J Steroid Biochem Mol Biol 2023; 225: 106200.
87.
Khadka NK, Hazen P, Akinola O, Pu X, Mainali L. Lipid and cholesterol peroxidation leads to a-crystallin membrane aggregation and cataract formation. Investig Ophthalmol Visual Sci 2025; 66: 8.
88.
Aleo MD, Doshna CM, Baltrukonis D, et al. Lens cholesterol biosynthesis inhibition: a common mechanism of cataract formation in laboratory animals by pharmaceutical products. J Appl Toxicol 2019; 39: 1348-61.
90.
Wang F, Gou X, Wu F, Qiao H, Zhao D. Quantitative assessment of the associations between ABCA1 gene polymorphism and glaucoma risk, evidence from a meta-analysis. Medicine 2024; 103: e40427.
91.
Joshi RS, Adatiya VH. Study of the relationship between serum lipid levels and primary open-angle glaucoma. Indian J Ophthalmol 2023; 71: 1948-52.
92.
Madjedi KM, Stuart KV, Chua SY, et al. The association between serum lipids and intraocular pressure in 2 large United Kingdom cohorts. Ophthalmology 2022; 129: 986-96.
93.
Wang T, Kimmel HR, Park C, et al. Regulatory role of cholesterol in modulating actin dynamics and cell adhesive interactions in the trabecular meshwork. BioRxiv 2024.
94.
Ferreira-Guerra M, Coleto-Alcudia V, Mora-García S, Caño-Delgado AI. The evolutionary journey of sterol synthesis pathways in eukaryotes. Plant Cell Physiol 2025; pcaf016.
95.
DeBose-Boyd RA, Ye J. SREBPs in lipid metabolism, insulin signaling, and beyond. Trends Biochem Sci 2018; 43: 358-68.
96.
Yan R, Cao P, Song W, et al. Structural basis for sterol sensing by Scap and Insig. Cell Rep 2021; 35: 109299.
97.
El-Mulki OS, Berni A, Kastner J, et al. The macular burden of calcified drusen and the onset of large choroidal hypertransmission defects in intermediate AMD. Am J Ophthalmol 2025; 278: 402-12.
98.
Storti F, Grimm C. Active cholesterol efflux in the retina and retinal pigment epithelium. In: Retinal Degenerative Diseases: Mechanisms and Experimental Therapy. Rickman CB, Grimm C, Anderson RE, et al. (eds.). Springer 2019; 51-5.
99.
Zhang X, Wang K, Zhu L, Wang Q. Reverse cholesterol transport pathway and cholesterol efflux in diabetic retinopathy. J Diab Res 2021; 2021: 8746114.
100.
Yu XH, Tang CK. ABCA1, ABCG1, and cholesterol homeostasis. Adv Exp Med Biol 2022; 1377: 95-107.
101.
Wang B, Tontonoz P. Liver X receptors in lipid signalling and membrane homeostasis. Nat Rev Endocrinol 2018; 14: 452-63.
102.
Domínguez-Avila JA. Dietary phenolic compounds exert some of their health-promoting bioactivities by targeting liver x receptor (Lxr) and retinoid x receptor (Rxr). Foods 2023; 12: 4205.
103.
Wang N, Westerterp M. ABC transporters, cholesterol efflux, and implications for cardiovascular diseases. Adv Exp Med Biol 2020; 1276: 67-83.
104.
Sharma A, Mannan A, Singh TG. Unraveling novel mechanisms of ATP-binding cassette (ABC) transporter in insulin resistance-induced amyloidogenesis. Metab Brain Dis 2025; 40: 218.
105.
Vieira CP, Fortmann SD, Hossain M, et al. Selective LXR agonist DMHCA corrects retinal and bone marrow dysfunction in type 2 diabetes. JCI Insight 2020; 5: e137230.
106.
Shah AA, Kamal MA, Akhtar S. Tumor angiogenesis and VEGFR-2: mechanism, pathways and current biological therapeutic interventions. Curr Drug Metab 2021; 22: 50-9.
107.
Ahmad A, Nawaz MI. Molecular mechanism of VEGF and its role in pathological angiogenesis. J Cell Biochem 2022; 123: 1938-65.
108.
Marchesi N, Capierri M, Pascale A, Barbieri A. Different therapeutic approaches for dry and wet AMD. Int J Mol Sci 2024; 25: 13053.
109.
Zhang J, Zhang J, Zhang C, et al. Diabetic macular edema: current understanding, molecular mechanisms and therapeutic implications. Cells 2022; 11: 3362.
110.
Almalki SG, Agrawal DK. ERK signaling is required for VEGF-A/VEGFR2-induced differentiation of porcine adipose-derived mesenchymal stem cells into endothelial cells. Stem Cell Res Ther 2017; 8: 113.
111.
Formica ML, Awde Alfonso HG, Palma SD. Biological drug therapy for ocular angiogenesis: anti-VEGF agents and novel strategies based on nanotechnology. Pharmacol Res Perspect 2021; 9: e00723.
112.
Shinojima A, Lee D, Tsubota K, Negishi K, Kurihara T. Retinal diseases regulated by hypoxia – basic and clinical perspectives: a comprehensive review. J Clin Med 2021; 10: 5496.
113.
Stanigut AM, Pana C, Enciu M, Deacu M, Cimpineanu B, Tuta LA. Hypoxia-inducible factors and diabetic kidney disease – how deep can we go? Int J Mol Sci 2022; 23: 10413.
114.
Hu S, Hang X, Wei Y, Wang H, Zhang L, Zhao L. Crosstalk among podocytes, glomerular endothelial cells and mesangial cells in diabetic kidney disease: an updated review. Cell Commun Signal 2024; 22: 136.
115.
Lewis-Luján LM, Guerrero-Magaña DE, Iloki-Lewis AP, et al. Advances in the molecular pathophysiology and emerging therapeutic strategies for diabetic retinopathy. Preprints 2025. DOI:10.20944/preprints202509.2271.v1.
116.
Wang J, Li M, Geng Z, et al. Role of oxidative stress in retinal disease and the early intervention strategies: a review. Oxid Med Cell Longev 2022; 2022: 7836828.
117.
Tu W, Wang H, Li S, Liu Q, Sha H. The anti-inflammatory and anti-oxidant mechanisms of the Keap1/Nrf2/ARE signaling pathway in chronic diseases. Aging Dis 2019; 10: 637.
118.
Matsumaru D, Motohashi H. The KEAP1-NRF2 system in healthy aging and longevity. Antioxidants 2021; 10: 1929.
119.
Testa G, Giannelli S, Sottero B, et al. 24-Hydroxycholesterol induces Tau proteasome-dependent degradation via the SIRT1/PGC1a/Nrf2 pathway: a potential mechanism to counteract Alzheimer’s disease. Antioxidants 2023; 12: 631.
120.
Zhang J, Zhang T, Zeng S, et al. The role of Nrf2/sMAF signalling in retina ageing and retinal diseases. Biomedicines 2023; 11: 1512.
121.
Camelo S, Latil M, Veillet S, Dilda PJ, Lafont R. Beyond AREDS formulations, what is next for intermediate age-related macular degeneration (iAMD) treatment? Potential benefits of antioxidant and anti-inflammatory apocarotenoids as neuroprotectors. Oxid Med Cell Longev 2020; 2020: 4984927.
122.
Cheng J, Liang J, Qi J. Role of nuclear factor (erythroid-derived 2)-like 2 in the age-resistant properties of the glaucoma trabecular meshwork. Exp Ther Med 2017; 14: 791-6.
123.
Xie T, Chen X, Chen W, et al. Curcumin is a potential adjuvant to alleviates diabetic retinal injury via reducing oxidative stress and maintaining Nrf2 pathway homeostasis. Front Pharmacol 2021; 12: 796565.
124.
Yuan D, Xu Y, Xue L, Zhang W, Gu L, Liu Q. Resveratrol protects against diabetic retinal ganglion cell damage by activating the Nrf2 signaling pathway. Heliyon 2024; 10: e30786.
125.
Wang J, Niu S, Hu X, et al. Trans-Golgi network tethering factors regulate TBK1 trafficking and promote the STING-IFN-I pathway. Cell Discov 2025; 11: 23.
126.
Liu T, Zhang L, Joo D, Sun SC. NF-kB signaling in inflammation. Signal Transd Target Ther 2017; 2: 17023.
127.
Li W, Jin K, Luo J, et al. NF-kB and its crosstalk with endoplasmic reticulum stress in atherosclerosis. Front Cardiovasc Med 2022; 9: 988266.
128.
Hu P, Li K, Peng X, et al. Nuclear receptor PPARa as a therapeutic target in diseases associated with lipid metabolism disorders. Nutrients 2023; 15: 4772.
129.
Botta M, Audano M, Sahebkar A, Sirtori CR, Mitro N, Ruscica M. PPAR agonists and metabolic syndrome: an established role? Int J Mol Sci 2018; 19: 1197.
130.
Liu M, Lim ST, Song W, Coffman TM, Wang X. Beyond lipids: fenofibrate in diabetic retinopathy and nephropathy. Trends Pharmacol Sci 2026; 47: 325-41.
131.
Ngah NF, Muhamad NA, Aziz RAA, et al. Fenofibrate for the prevention of progression of non-proliferative diabetic retinopathy: review, consensus recommendations and guidance for clinical practice. Int J Ophthalmol 2022; 15: 2001-8.
132.
Zhao J, Geng Y, Hua H, et al. Fenofibrate inhibits the expression of VEGFC and VEGFR-3 in retinal pigmental epithelial cells exposed to hypoxia. Exp Ther Med 2015; 10: 1404-12.
133.
Varet J, Vincent L, Mirshahi P, et al. Fenofibrate inhibits angiogenesis in vitro and in vivo. Cell Mol Life Sci 2003; 60: 810-9.
134.
Lymperopoulou C, Kandarakis SA, Tzanaki I, Mylona I, Xanthos T, Agouridis AP. The effect of statins on ocular disorders: a systematic review of randomized controlled trials. Pharmaceuticals 2023; 16: 711.
135.
Varughese MS, Nayak AU, Jacob S. Fenofibrate therapy in reducing the progression of diabetic retinopathy: revisiting the FIELD and ACCORD-EYE studies through the LENS trial. Eye 2025; 39: 15-7.
136.
Hang A, Feldman S, Amin AP, Ochoa JAR, Park SS. Intravitreal anti-vascular endothelial growth factor therapies for retinal disorders. Pharmaceuticals 2023; 16: 1140.
137.
Hobbs RP, Bernstein PS. Nutrient supplementation for age-related macular degeneration, cataract, and dry eye. J Ophthal Vision Res 2014; 9: 487.
138.
Shirian JD, Shukla P, Singh RP. Exploring new horizons in neovascular age-related macular degeneration: novel mechanisms of action and future therapeutic avenues. Eye 2025; 39: 40-4.
139.
Ana RD, Gliszczyńska A, Sanchez-Lopez E, et al. Precision medicines for retinal lipid metabolism-related pathologies. J Personal Med 2023; 13: 635.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.