Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
Editor's Choice
METABOLIC DISORDERS / STATE OF THE ART PAPER
Biomarkers for early identification of metabolic dysfunction-associated steatotic liver disease (MASLD): a narrative review
1
University of Montenegro, Faculty of Medicine, Podgorica, Montenegro
2
Center for Laboratory Diagnostics, Primary Health Care Center, Podgorica, Montenegro
3
Faculty of Medicine, University of Belgrade, Belgrade, Serbia
4
Department of Gastroenterology and Hepatology, Clinic for Internal Medicine, Clinical Hospital Center Zemun, Belgrade, Serbia
5
Clinic for Neurosurgery, Clinical Center of Montenegro, Podgorica, Montenegro
6
Department of Endocrinology and Metabolism, Faculty of Medicine, Düzce University, Düzce, Turkey
7
Department for Medical Biochemistry, University of Belgrade, Faculty of Pharmacy, Belgrade, Serbia
Submission date: 2026-01-22
Final revision date: 2026-03-14
Acceptance date: 2026-03-20
Online publication date: 2026-04-23
Publication date: 2026-04-30
Corresponding author
Aleksandra Klisic
University of Montenegro Faculty of Medicine Podgorica, Montenegro Center for Laboratory Diagnostics Primary Health Care Center Trg Nikole Kovacevica 6 81000 Podgorica Montenegro Phone/fax: +382 20 481 999
University of Montenegro Faculty of Medicine Podgorica, Montenegro Center for Laboratory Diagnostics Primary Health Care Center Trg Nikole Kovacevica 6 81000 Podgorica Montenegro Phone/fax: +382 20 481 999
Arch Med Sci 2026;22(2):602-641
KEYWORDS
metabolic dysfunction-associated steatotic liver diseaseoxidative stressinflammationnon-invasive diagnosticsgenetic markers
TOPICS
ABSTRACT
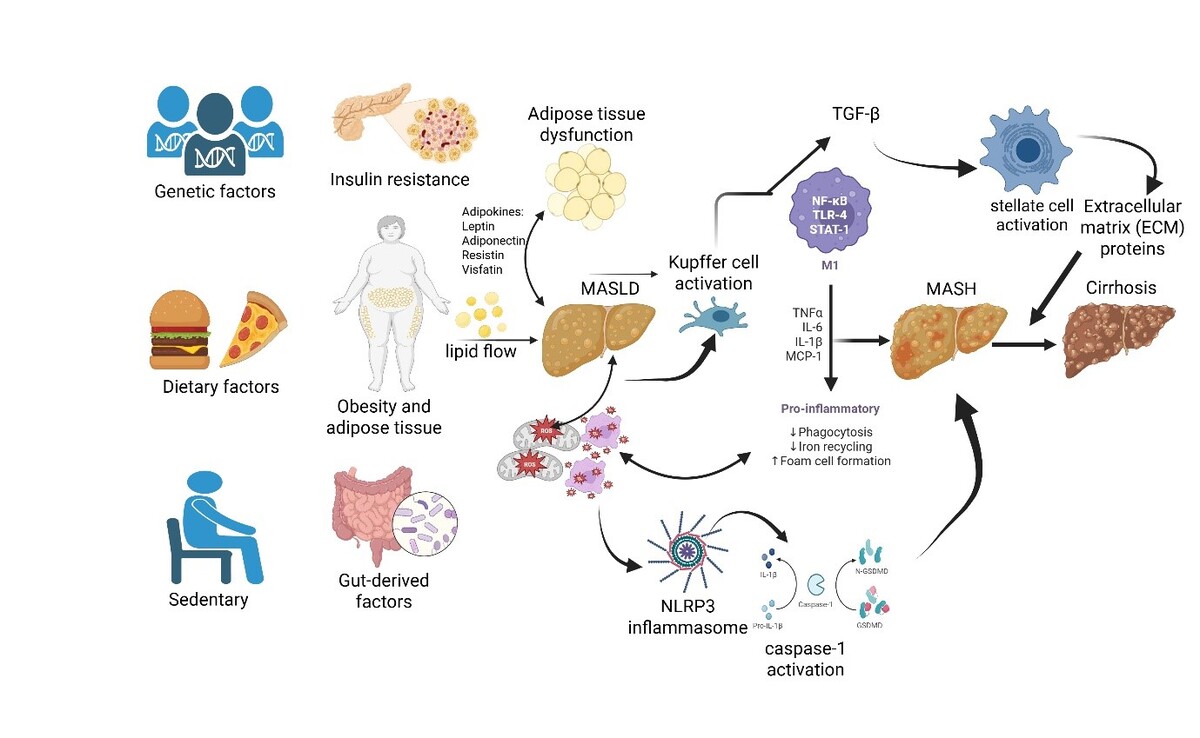
Metabolic dysfunction-associated steatotic liver disease (MASLD) has a globally increasing prevalence. Oxidative stress and inflammation, as interdependent processes, play pivotal roles in the pathogenesis of MASLD. Moreover, heritability has a significant impact on MASLD. Hence, to fully understand the nature of complex and multifactorial diseases such as MASLD, it is important to consider the role of genetic and epigenetic factors as essential aspects of a broader context that includes clinical and environmental influences and their interactions when multiple metabolic pathways are involved. While traditional biomarkers may be insufficient in the early stages of this process, new-generation biomarkers and multi-omic approaches hold the potential to contribute to the improvement of disease diagnosis and prognosis. This review article aims to systematically examine oxidative stress and inflammation biomarkers, as well as genetic and epigenetic factors in MASLD, highlighting current diagnostic advances and future perspectives in the field.
REFERENCES (343)
1.
Puri P, Fuchs M. Population management of nonalcoholic fatty liver disease. Fed Pract 2019; 36: 72-82.
2.
Le MH, Yeo YH, Li X, et al. 2019 Global NAFLD prevalence: a systematic review and meta-analysis. Clin Gastroenterol Hepatol 2022; 20: 2809-17.
3.
Klisic A, Isakovic A, Kocic G, et al. Relationship between oxidative stress, inflammation and dyslipidemia with fatty liver index in patients with type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes 2018; 126: 371-8.
4.
Younossi ZM, Golabi P, de Avila L, et al. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: a systematic review and meta-analysis. J Hepatol 2019; 71: 793-801.
5.
Quek J, Chan KE, Wong ZY, et al. Global prevalence of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in the overweight and obese population: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol 2023; 8: 20-30.
6.
Dulai PS, Singh S, Patel J, et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. Hepatology 2017; 65: 1557-65.
7.
Singh S, Allen AM, Wang Z, Prokop LJ, Murad MH, Loomba R. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin Gastroenterol Hepatol 2015; 13: 643-54.
8.
Lim GEH, Tang A, Ng CH, et al. An observational data meta-analysis on the differences in prevalence and risk factors between MAFLD vs NAFLD. Clin Gastroenterol Hepatol 2023; 21: 619-29.e7.
9.
Koch LK, Yeh MM. Nonalcoholic fatty liver disease (NAFLD): diagnosis, pitfalls, and staging. Ann Diagn Pathol 2018; 37: 83-90.
10.
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease: meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016; 64: 73-84.
11.
Wong RJ, Aguilar M, Cheung R, et al. Nonalcoholic steatohepatitis is the second leading etiology of liver disease among adults awaiting liver transplantation in the United States. Gastroenterology 2015; 148: 547-55.
12.
Golabi P, Paik JM, AlQahtani S, Younossi Y, Tuncer G, Younossi ZM. Burden of non-alcoholic fatty liver disease in Asia, the Middle East and North Africa: data from global burden of disease 2009–2019. J Hepatol 2021; 75: 795-809.
13.
Estes C, Anstee QM, Arias-Loste MT, et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016–2030. J Hepatol 2018; 69: 896-904.
14.
Bae SDW, George J, Qiao L. From MAFLD to hepatocellular carcinoma and everything in between. Chin Med J 2022; 135: 547-56.
15.
Ludwig J, Viggiano TR, McGill DB, Oh BJ. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc 1980; 55: 434-8.
16.
Eslam M, Sanyal AJ, George J. MAFLD: a consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology 2020; 158: 1999-2014.
17.
Eslam M, Newsome PN, Sarin SK, et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J Hepatol 2020; 73: 202-9.
18.
Rinella ME, Lazarus JV, Ratziu V, et al. NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology 2023; 78: 1966-86.
19.
Caldwell SH, Argo CK. Non-alcoholic fatty liver disease. Clinical Dilemmas in Non-Alcoholic Fatty Liver Disease. 1st edition. Wiley-Blackwell; 2016; 1-7.
20.
Kanwal F, Neuschwander-Tetri BA, Loomba R, Rinella ME. Metabolic dysfunction-associated steatotic liver disease: update and impact of new nomenclature on the American Association for the Study of Liver Diseases practice guidance on nonalcoholic fatty liver disease. Hepatology 2024; 79: 1212-9.
21.
Seto WK, Yuen MF. Nonalcoholic fatty liver disease in Asia: emerging perspectives. J Gastroenterol 2017; 52: 164-74.
22.
Musso G, Gambino R, Cassader M, Pagano G. Meta-analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med 2011; 43: 617-49.
23.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012; 55: 2005-23.
24.
Jarvis H, Craig D, Barker R, et al. Metabolic risk factors and incident advanced liver disease in NAFLD: a systematic review and meta-analysis of population-based observational studies. PLoS Med 2020; 17: e1003100.
25.
Chen S, Zeng Q, Cai X, et al. Multiomics analyses decipher intricate changes in the cellular and metabolic landscape of steatotic livers upon dietary restriction and sleeve gastrectomy. Int J Biol Sci 2024; 20: 4438-57.
26.
Ahmadizar F, Younossi ZM. Exploring biomarkers in nonalcoholic fatty liver disease among individuals with type 2 diabetes mellitus. J Clin Gastroenterol 2025; 59: 36-46.
27.
Li K, Mathew B, Saldanha E, et al. New insights into biomarkers and risk stratification to predict hepatocellular cancer. Mol Med 2025; 31: 152.
28.
Tan EY, Muthiah MD, Sanyal AJ. Metabolomics at the cutting edge of risk prediction of MASLD. Cell Rep Med 2024; 5: 101853.
29.
Liu J, Hu S, Chen L, et al. Profiling the genome and proteome of metabolic dysfunction-associated steatotic liver disease identifies potential therapeutic targets. medRxiv 2023: 2023.11.30.23299247.
30.
Mayén AL, Sabra M, Aglago EK, et al. Hepatic steatosis, metabolic dysfunction and risk of mortality: findings from a multinational prospective cohort study. BMC Med 2024; 22: 221.
31.
Denimal D, Ponnaiah M, Phan F, et al. MASLD biomarkers and progression of lower limb arterial calcification in patients with type 2 diabetes: a prospective cohort study. Cardiovasc Diabetol 2025; 24: 176.
32.
Day CP, James OF. Steatohepatitis: a tale of two ‘hits’? Gastroenterology 1998; 114: 842-5.
33.
Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest 2005; 115: 1343-51.
34.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005; 41: 1313-21.
35.
Baselli GA, Jamialahmadi O, Pelusi S, et al. Rare ATG7 genetic variants predispose patients to severe fatty liver disease. J Hepatol 2022; 77: 596-606.
36.
Chang Q, Zhang Y, Zhang T, et al. Healthy lifestyle and the risk of metabolic dysfunction-associated fatty liver disease: a large prospective cohort study. Diabetes Metab J 2024; 48: 971-82.
37.
Targher G, Corey KE, Byrne CD, et al. The complex link between NAFLD and type 2 diabetes mellitus - mechanisms and treatments. Nat Rev Gastroenterol Hepatol 2021; 18: 599-612.
38.
Tilg H, Moschen AR. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology 2010; 52: 1836-46.
39.
Duez H, Pourcet B. Nuclear receptors in the control of the NLRP3 inflammasome pathway. Front Endocrinol 2021; 12: 630536.
40.
Nassir F. NAFLD: mechanisms, treatments, and biomarkers. Biomolecules 2022; 12: 824.
41.
Li J, Cao F, Yin HL, et al. Ferroptosis: past, present and future. Cell Death Dis 2020; 11: 88.
42.
Sookoian S, Pirola CJ. PNPLA3, the history of a gene that was found to be associated with human disease in the post-genome era: from liver to heart. Hum Genet 2022; 141: 709-22.
43.
Hydes T, Alam U, Cuthbertson DJ. The impact of macronutrient intake on non-alcoholic fatty liver disease (NAFLD): too much fat, too much carbohydrate, or just too many calories? Front Nutr 2021; 8: 640-61.
44.
du Plessis J, van Pelt J, Korf H, et al. Association of adipose tissue inflammation with histologic severity of nonalcoholic fatty liver disease. Gastroenterology 2015; 149: 635-48.
45.
Marchesini G, Brizi M, Morselli-Labate AM, et al. Association of nonalcoholic fatty liver disease with insulin resistance. Am J Med 1999; 107: 450-5.
46.
Hotamisligil GS. Inflammation, metaflammation and immunometabolic disorders. Nature 2017; 542: 177-85.
47.
Li S, Tan HY, Wang N, et al. The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci 2015; 16: 26087-124.
48.
Simoes ICM, Amorim R, Teixeira J, et al. The alterations of mitochondrial function during NAFLD progression – an independent effect of mitochondrial ROS production. Int J Mol Sci 2021; 22: 6848.
49.
Smith GI, Shankaran M, Yoshino M, et al. Insulin resistance drives hepatic de novo lipogenesis in nonalcoholic fatty liver disease. J Clin Invest 2020; 130: 1453-60.
50.
Pohl J, Ring A, Hermann T, Stremmel W. Role of FATP in parenchymal cell fatty acid uptake. Biochim Biophys Acta 2004; 1686: 1-6.
51.
Chaldakov GN, Stankulov IS, Hristova M, Ghenev PI. Adipobiology of disease: adipokines and adipokine-targeted pharmacology. Curr Pharm Des 2003; 9: 1023-31.
52.
Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat Rev Microbiol 2021; 19: 55-71.
53.
Sharpton SR, Schnabl B, Knight R, et al. Current concepts, opportunities, and challenges of gut microbiome-based personalized medicine in nonalcoholic fatty liver disease. Cell Metab 2021; 33: 21-32.
54.
Lang S, Demir M, Martin A, et al. Intestinal Virome signature associated with severity of nonalcoholic fatty liver disease. Gastroenterology 2020; 159: 1839-52.
55.
Leung H, Long X, Ni Y, et al. Risk assessment with gut microbiome and metabolite markers in NAFLD development. Sci Transl Med 2022; 14: eabk0855.
56.
Carpino G, Del Ben M, Pastori D, et al. Increased liver localization of lipopolysaccharides in human and experimental NAFLD. Hepatology 2020; 72: 470-85.
57.
Meijnikman AS, Davids M, Herrema H, et al. Microbiome-derived ethanol in nonalcoholic fatty liver disease. Nat Med 2022; 28: 2100-6.
58.
Shin JH, Lee Y, Song EJ, et al. Faecalibacterium prausnitzii prevents hepatic damage in a mouse model of NASH induced by a high-fructose high-fat diet. Front Microbiol 2023; 14: 1123547.
59.
Alexander M, Turnbaugh PJ. Deconstructing mechanisms of diet-microbiome-immune interactions. Immunity 2020; 53: 264-76.
60.
Adams LA, Anstee QM, Tilg H, et al. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 2017; 66: 1138-53.
61.
Ferdous SE, Ferrell JM. Pathophysiological relationship between type 2 diabetes mellitus and metabolic dysfunction-associated steatotic liver disease: novel therapeutic approaches. Int J Mol Sci 2024; 25: 8731.
62.
Targher G, Byrne CD, Tilg H. NAFLD and increased risk of cardiovascular disease: clinical associations, pathophysiological mechanisms and pharmacological implications. Gut 2020; 69: 1691-705.
63.
Chew NWS, Mehta A, Goh RSJ, et al. Cardiovascular-liver-metabolic health: recommendations in screening, diagnosis, and management of metabolic dysfunction-associated steatotic liver disease in cardiovascular disease via modified Delphi approach. Circulation 2025; 151: 98-119.
64.
Gerber L, Otgonsuren M, Mishra A, et al. Non-alcoholic fatty liver disease (NAFLD) is associated with low level of physical activity: a population-based study. Aliment Pharmacol Ther 2012; 36: 772-81.
65.
Younossi Z, Anstee QM, Marietti M, et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol 2018; 151: 11-20.
66.
Klisic A, Abenavoli L, Fagoonee S, Kavaric N, Kocic G, Ninić A. Older age and HDL-cholesterol as independent predictors of liver fibrosis assessed by BARD score. Minerva Med 2019; 110: 191-8.
67.
Zhou XD, Targher G, Byrne CD, et al. An international Multidisciplinary consensus statement on MAFLD and the risk of CVD. Hepatol Int 2023; 17: 773-91.
68.
de Avila L, Henry L, Paik JM, et al. Nonalcoholic fatty liver disease is independently associated with higher all-cause and cause-specific mortality. Clin Gastroenterol Hepatol 2023; 21: 2588-96.
69.
Mantovani A, Petracca G, Csermely A, et al. Non-alcoholic fatty liver disease and risk of new-onset heart failure: an updated meta-analysis of about 11 million individuals. Gut 2023; 72: 372-80.
70.
Tan DJ, Ng CH, Lin SY, et al. Clinical characteristics, surveillance, treatment allocation, and outcomes of non-alcoholic fatty liver disease-related hepatocellular carcinoma: a systematic review and meta-analysis. Lancet Oncol 2022; 23: 521-30.
71.
Mantovani A, Petracca G, Beatrice G, et al. Non-alcoholic fatty liver disease and increased risk of incident extrahepatic cancers: a meta-analysis of observational cohort studies. Gut 2022; 71: 778-88.
72.
Kjaergaard M, Lindvig KP, Thorhauge KH, et al. Using the ELF test, FIB-4 and NAFLD fibrosis score to screen the population for liver disease. J Hepatol 2023; 79: 277-86.
73.
Ozturk A, Olson MC, Samir AE, Venkatesh SK. Liver fibrosis assessment: MR and US elastography. Abdom Radiol 2022; 47: 3037-50.
74.
Zhang X, Zheng MH, Liu D, et al. A blood-based biomarker panel for non-invasive diagnosis of metabolic dysfunction-associated steatohepatitis. Cell Metab 2025; 37: 59-68.e3.
75.
Mak AL, Lee J, van Dijk AM, et al. Systematic review with meta-analysis: diagnostic accuracy of pro-C3 for hepatic fibrosis in patients with non-alcoholic fatty liver disease. Biomedicines 2021; 9: 1920.
76.
Tacke F, Horn P, Wong VWS, et al. EASL–EASD–EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD) J Hepatol 2024; 81: 492-542.
77.
Abdelhameed F, Kite C, Lagojda L, et al. Non-invasive scores and serum biomarkers for fatty liver in the era of metabolic dysfunction-associated steatotic liver disease (MASLD): a comprehensive review from NAFLD to MAFLD and MASLD. Curr Obes Rep 2024; 13: 510-31.
78.
Feldstein AE, Wieckowska A, Lopez AR, Liu YC, Zein NN, McCullough AJ. Cytokeratin-18 fragment levels as noninvasive biomarkers for nonalcoholic steatohepatitis: a multicenter validation study. Hepatology 2009; 50: 1072-8.
79.
Lichtinghagen R, Pietsch D, Bantel H, Manns MP, Brand K, Bahr MJ. The enhanced liver fibrosis (ELF) score: normal values, influence factors and proposed cut-off values. J Hepatol 2013; 59: 236-42.
80.
Wattacheril JJ, Abdelmalek MF, Lim JK, Sanyal AJ. AGA clinical practice update on the role of noninvasive biomarkers in the evaluation and management of nonalcoholic fatty liver disease: expert review. Gastroenterology 2023; 165: 1080-8.
81.
Daniels SJ, Leeming DJ, Eslam M, et al. ADAPT: an algorithm incorporating PRO-C3 accurately identifies patients with NAFLD and advanced fibrosis. Hepatology 2019; 69: 1075-86.
82.
Liu M, Qiu H, Zhang W, et al. Evaluation of blood-based PRO-C3 testing as a diagnostic marker for staging liver fibrosis: a systematic review and meta-analysis. J Gastroenterol Hepatol 2023; 38: 1018-27.
83.
Jang W, Song JS. Non-invasive imaging methods to evaluate non-alcoholic fatty liver disease with fat quantification: a review. Diagnostics 2023; 13: 1852.
84.
Malik S, Das R, Thongtan T, Thompson K, Dbouk N. AI in hepatology: revolutionizing the diagnosis and management of liver disease. J Clin Med 2024; 13: 7833.
85.
Pugliese N, Bertazzoni A, Hassan C, Schattenberg JM, Aghemo A. Revolutionizing MASLD: how artificial intelligence is shaping the future of liver care. Cancers 2025; 17: 722.
86.
Udompap P, Liu K, Attia IZ, et al. Performance of AI-enabled electrocardiogram in the prediction of metabolic dysfunction-associated steatotic liver disease. Clin Gastroenterol Hepatol 2025; 23: 574-82.e3.
87.
Wakabayashi SI, Kimura T, Tamaki N, et al. AI-based platelet-independent noninvasive test for liver fibrosis in MASLD patients. JGH Open 2025; 9: e70150.
88.
Ratziu V, Hompesch M, Petitjean M, et al. Artificial intelligence-assisted digital pathology for non-alcoholic steatohepatitis: current status and future directions. J Hepatol 2024; 80: 335-51.
89.
Njei B, Osta E, Njei N, Al-Ajlouni YA, Lim JK. An explainable machine learning model for prediction of high-risk nonalcoholic steatohepatitis. Sci Rep 2024; 14: 8589.
90.
Lin B, Ma Y, Wu S. Multi-omics and artificial intelligence-guided data integration in chronic liver disease: prospects and challenges for precision medicine. OMICS 2022; 26: 415-21.
91.
Ballard JL, Wang Z, Li W, Shen L, Long Q. Deep learning-based approaches for multi-omics data integration and analysis. BioData Min 2024; 17: 38.
92.
Harrison SA, Bedossa P, Guy CD, et al. MAESTRO-NASH Investigators. A phase 3, randomized, controlled trial of resmetirom in NASH with liver fibrosis. N Engl J Med 2024; 390: 497-509.
93.
Madrigal Pharmaceuticals Inc. Rezdiffra (resmetirom): Highlights of prescribing information. West Conshohocken, PA: Madrigal Pharmaceuticals, Inc.; 2024.
94.
Souza M, Al-Sharif L, Antunes VLJ, Huang DQ, Loomba R. Comparison of pharmacological therapies in metabolic dysfunction-associated steatohepatitis for fibrosis regression and MASH resolution: systematic review and network meta-analysis. Hepatology 2025; 82: 1523-33.
95.
Devasia AG, Ramasamy A, Leo CH. Current therapeutic landscape for metabolic dysfunction-associated steatohepatitis. Int J Mol Sci 2025; 26: 1778.
96.
Rafaqat S, Gluscevic S, Mercantepe F, Rafaqat S, Klisic A. Interleukins: pathogenesis in non-alcoholic fatty liver disease. Metabolites 2024; 14: 153.
97.
Jiang Z, Chen L, Dou X. Glutathionylation and metabolic dysfunction-associated steatotic liver disease. Biochimie 2025; 234: 10-9.
98.
Begriche K, Massart J, Robin MA, Bonnet F, Fromenty B. Mitochondrial adaptations and dysfunctions in nonalcoholic fatty liver disease. Hepatology 2013; 58: 1497-507.
99.
Rolo AP, Teodoro JS, Palmeira CM. Role of oxidative stress in the pathogenesis of nonalcoholic steatohepatitis. Free Radic Biol Med 2012; 52: 59-69.
100.
García-Ruiz C, Fernández-Checa JC. Mitochondrial oxidative stress and antioxidants balance in fatty liver disease. Hepatol Commun 2018; 2: 1425-39.
101.
Kitada T, Seki S, Iwai S, Yamada T, Sakaguchi H, Wakasa K. In situ detection of oxidative DNA damage, 8-hydroxydeoxyguanosine, in chronic human liver disease. J Hepatol 2001; 35: 613-8.
102.
Gaborit B, Ancel P, Abdullah AE, et al. Effect of empagliflozin on ectopic fat stores and myocardial energetics in type 2 diabetes: the EMPACEF study. Cardiovasc Diabetol 2021; 20: 57.
103.
Radosavljevic T, Brankovic M, Samardzic J, et al. Altered mitochondrial function in MASLD: key features and promising therapeutic approaches. Antioxidants 2024; 13: 906.
104.
Tung C, Varzideh F, Farroni E, et al. Elamipretide: a review of its structure, mechanism of action, and therapeutic potential. Int J Mol Sci 2025; 26: 944.
105.
Fülöp P, Derdák Z, Sheets A, et al. Lack of UCP2 reduces fas-mediated liver injury in Ob/Ob mice and reveals importance of cell-specific UCP2 expression. Hepatology 2006; 44: 592-601.
106.
Plecitá-Hlavatá L, Engstová H, Ježek J, et al. Potential of mitochondria-targeted antioxidants to prevent oxidative stress in pancreatic b-cells. Oxid Med Cell Longev 2019; 2019: 1826303.
107.
Wang K, Tan W, Liu X, et al. New insight and potential therapy for NAFLD: CYP2E1 and flavonoids. Biomed Pharmacother 2021; 137: 111326.
108.
Videla LA, Rodrigo R, Orellana M, et al. Oxidative stress-related parameters in the liver of non-alcoholic fatty liver disease patients. Clin Sci 2004; 106: 261-8.
109.
LeFort KR, Rungratanawanich W, Song BJ. Contributing roles of mitochondrial dysfunction and hepatocyte apoptosis in liver diseases through oxidative stress, post-translational modifications, inflammation, and intestinal barrier dysfunction. Cell Mol Life Sci 2024; 81: 34.
110.
Zeng T, Zhang CL, Zhao N, et al. Impairment of Akt activity by CYP2E1 mediated oxidative stress is involved in chronic ethanol-induced fatty liver. Redox Biol 2018; 14: 295-304.
111.
Seth RK, Das S, Pourhoseini S, et al. M1 polarization bias and subsequent nonalcoholic steatohepatitis progression is attenuated by nitric oxide donor DETA NONOate via inhibition of CYP2E1-induced oxidative stress in obese mices. J Pharmacol Exp Ther 2015; 352: 77-89.
112.
Wang X, Tang Y, Lu J, et al. Characterization of novel cytochrome P450 2E1 knockout rat model generated by CRISPR/Cas9. Biochem Pharmacol 2016; 105: 80-90.
113.
Cammisotto V, Valeriani E, Pignatelli P, Violi F. Nicotinamide adenine dinucleotide phosphate oxidases and metabolic dysfunction-associated steatotic liver disease. Antioxidants 2025; 14: 83.
114.
Allameh A, Niayesh-Mehr R, Aliarab A, Sebastiani G, Pantopoulos K. Oxidative stress in liver pathophysiology and disease. Antioxidants 2023; 12: 1653.
115.
Thannickal VJ, Jandeleit-Dahm K, Szyndralewiez C, Török NJ. Pre-clinical evidence of a dual NADPH oxidase 1/4 inhibitor (Setanaxib) in liver, kidney and lung fibrosis. J Cell Mol Med 2023; 27: 471-81.
116.
Liu L, Hong W, Li M, et al. A cross talk between BRG1 and males absent on the first contributes to reactive oxygen species production in a mouse model of nonalcoholic steatohepatitis. Antioxidants Redox Signal 2019; 30: 1539-52.
117.
Zhong H, Yin H. Role of lipid peroxidation derived 4-hydroxynonenal (4-HNE) in cancer: focusing on mitochondria. Redox Biol 2015; 4: 193-9.
118.
Raza H, John A. 4-hydroxynonenal induces mitochondrial oxidative stress, apoptosis and expression of glutathione S-transferase A4-4 and cytochrome P450 2E1 in PC12 cells. Toxicol Appl Pharmacol 2006; 216: 309-18.
119.
Schierscher T, Salzmann L, Singh N, et al. An isotope dilution-liquid chromatography-tandem mass spectrometry (ID-LC-MS/MS)-based candidate reference measurement procedure for the quantification of carbamazepine in human serum and plasma. Clin Chem Lab Med 2024; 62: 1301-13.
120.
Sies H. Chapter 1. Oxidative eustress and oxidative distress: introductory remarks. In: Oxidative stress. Sies H (ed.). Academic Press 2020; 3-12.
121.
Valavanidis A, Vlachogianni T, Fiotakis C. 8-hydroxy-2¢-deoxyguanosine (8-OHdG): a critical biomarker of oxidative stress and carcinogenesis. J Environ Sci Heal Part C Environ Carcinog Ecotoxicol Rev 2009; 27: 120-39.
122.
Wu LL, Chiou CC, Chang PY, Wu JT. Urinary 8-OHdG: a marker of oxidative stress to DNA and a risk factor for cancer, atherosclerosis and diabetics. Clin Chim Acta 2004; 339: 1-9.
123.
Kim SH, Weiß C, Hoffmann U, Borggrefe M, Akin I, Behnes M. Advantages and limitations of current biomarker research: from experimental research to clinical application. Curr Pharm Biotechnol 2017; 18: 445-55.
124.
Marrocco I, Altieri F, Peluso I. Measurement and clinical significance of biomarkers of oxidative stress in humans. Oxid Med Cell Longev 2017; 2017: 6501046.
125.
Chatgilialoglu C. Biomarkers of oxidative and radical stress. Biomolecules 2024; 14: 194.
126.
Jakubek P, Kalinowski P, Karkucinska-Wieckowska A, et al. Oxidative stress in metabolic dysfunction-associated steatotic liver disease (MASLD): how does the animal model resemble human disease? FASEB J 2024; 38: e23466.
127.
Masarone M, Rosato V, Dallio M, et al. Role of oxidative stress in pathophysiology of nonalcoholic fatty liver disease. Oxid Med Cell Longev 2018; 2018; 9547613.
128.
Klisic A, Kavaric N, Ninic A, Kotur-Stevuljevic J. Oxidative stress and cardiometabolic biomarkers in patients with non-alcoholic fatty liver disease. Sci Rep 2021; 11: 18455.
129.
Bellanti F, Villani R, Facciorusso A, Vendemiale G, Serviddio G. Lipid oxidation products in the pathogenesis of non-alcoholic steatohepatitis. Free Radic Biol Med 2017; 111: 173-85.
130.
Barrera G, Pizzimenti S, Daga M, et al. Lipid peroxidation-derived aldehydes, 4-hydroxynonenal and malondialdehyde in aging-related disorders. Antioxidants 2018; 7: 102.
131.
Papac-Milicevic N, Busch CJL, Binder CJ. Malondialdehyde epitopes as targets of immunity and the implications for atherosclerosis. Adv Immunol 2016; 131: 1-59.
132.
Park GC, Bang SY, Kim JM, et al. Inhibiting ferroptosis prevents the progression of steatotic liver disease in obese mice. Antioxidants 2024; 13: 1336.
133.
Merino de Paz N, Carrillo-Palau M, Hernández-Camba A, et al. Association of serum malondialdehyde levels with lipid profile and liver function in patients with inflammatory bowel disease. Antioxidants 2024; 13: 1171.
134.
Tratenšek A, Locatelli I, Grabnar I, Drobne D, Vovk T. Oxidative stress-related biomarkers as promising indicators of inflammatory bowel disease activity: a systematic review and meta-analysis. Redox Biol 2024; 77: 103380.
135.
Albano E, Mottaran E, Vidali M, et al. Immune response towards lipid peroxidation products as a predictor of progression of non-alcoholic fatty liver disease to advanced fibrosis. Gut 2005; 54: 987-93.
136.
Korkmaz K, Deger O, Yigit E, Uydu HA, Mercantepe T, Demir S. Effect of propolis extracts on OxLDL and LOX-1 levels in ApoE knockout mice fed a high fat diet. Life 2025; 15: 565.
137.
Yılmaz HK, Türker M, Kutlu EY, et al. Investigation of the effects of white tea on liver fibrosis: an experimental animal model. Food Sci Nutr 2024; 12: 2998-3006.
138.
Yen WJ, Chyau CC, Lee CP, Chu HL, Chang LW, Duh P. Der cytoprotective effect of white tea against H2O2-induced oxidative stress in vitro. Food Chem 2013; 141: 4107-14.
139.
Bilen A, Mercantepe F, Tümkaya L, Yilmaz A, Batcik Ş. The hepatoprotective potential of resveratrol in an experimental model of ruptured abdominal aortic aneurysm via oxidative stress and apoptosis. J Biochem Mol Toxicol 2021; 35: e22836.
140.
Malkoç M, Patan H, Yaman SÖ, et al. L-theanine alleviates liver and kidney dysfunction in septic rats induced by cecal ligation and puncture. Life Sci 2020; 249: 117502.
141.
Chalasani NP, Sanyal AJ, Kowdley KV, et al.; NASH CRN Research Group. Pioglitazone versus vitamin E versus placebo for the treatment of non-diabetic patients with non-alcoholic steatohepatitis: PIVENS trial design. Contemp Clin Trials 2009; 30: 88-96.
142.
Cristani M, Speciale A, Saija A, Gangemi S, Minciullo PL. Circulating advanced oxidation protein products as oxidative stress biomarkers and progression mediators in pathological conditions related to inflammation and immune dysregulation. Curr Med Chem 2016; 23: 3862-82.
143.
Azouaou Toualbi L, Mounir A, Wafa B, et al. Implications of advanced oxidation protein products and vitamin e in atherosclerosis progression. Arch Med Sci Atheroscler Dis 2021; 6: 135-44.
144.
Breitzig M, Bhimineni C, Lockey R, Kolliputi N. 4-hydroxy-2-nonenal: a critical target in oxidative stress? Am J Physiol Cell Physiol 2016; 311: C537-43.
145.
Shoeb M, Ansari N, Srivastava S, Ramana K. 4-hydroxynonenal in the pathogenesis and progression of human diseases. Curr Med Chem 2013; 21: 230-7.
146.
Frijhoff J, Winyard PG, Zarkovic N, et al. Clinical relevance of biomarkers of oxidative stress. Antioxid Redox Signal 2015; 23: 1144-70.
147.
Li Y, Zhao T, Li J, et al. Oxidative stress and 4-hydroxy-2-nonenal (4-HNE): implications in the pathogenesis and treatment of aging-related diseases. J Immunol Res 2022; 2022: 2233906.
148.
Musiek ES, Morrow JD. F2-isoprostanes as markers of oxidant stress: an overview. Curr Protoc Toxicol 2005, Chapter 17: Unit 17.5. doi:10.1002/0471140856.tx1705s24.
149.
Del Ben M, Polimeni L, Carnevale R, et al. NOX2-generated oxidative stress is associated with severity of ultrasound liver steatosis in patients with non-alcoholic fatty liver disease. BMC Gastroenterol 2014; 14: 81.
150.
Yang J, Fernández-Galilea M, Martínez-Fernández L, et al. Oxidative stress and non-alcoholic fatty liver disease: effects of omega-3 fatty acid supplementation. Nutrients 2019; 11: 872.
151.
Maiolino G, Rossitto G, Caielli P, Bisogni V, Rossi GP, Calò LA. The role of oxidized low-density lipoproteins in atherosclerosis: the myths and the facts. Mediators Inflamm 2013; 2013: 714653.
152.
Winklhofer-Roob BM, Faustmann G, Roob JM. Low-density lipoprotein oxidation biomarkers in human health and disease and effects of bioactive compounds. Free Radic Biol Med 2017; 111: 38-86.
153.
Ishigaki Y, Oka Y, Katagiri H. Circulating oxidized LDL: a biomarker and a pathogenic factor. Curr Opin Lipidol 2009; 20: 363-9.
154.
Zhao X, Zhang HW, Xu RX, et al. Oxidized-LDL is a useful marker for predicting the very early coronary artery disease and cardiovascular outcomes. Per Med 2018; 15: 521-9.
155.
Hu Z, Yue H, Jiang N, Qiao L. Diet, oxidative stress and MAFLD: a mini review. Front Nutr 2025; 12: 1539578.
156.
Tan Z, Wu Y, Meng Y, et al. Trends in oxidative balance score and prevalence of metabolic dysfunction-associated steatotic liver disease in the United States: National Health and Nutrition Examination Survey 2001 to 2018. Nutrients 2023; 15: 4931.
157.
Cesarini L, Grignaffini F, Alisi A, Pastore A. Alterations in glutathione redox homeostasis in metabolic dysfunction-associated fatty liver disease: a systematic review. Antioxidants 2024; 13: 1461.
158.
Ore A, Akinloye OA. Oxidative stress and antioxidant biomarkers in clinical and experimental models of non-alcoholic fatty liver disease. Medicina 2019; 55: 26.
159.
Yesilova Z, Yaman H, Oktenli C, et al. Systemic markers of lipid peroxidation and antioxidants in patients with nonalcoholic fatty liver disease. Am J Gastroenterol 2005; 100: 850-5.
160.
Zhang J, Ouyang H, Gu X, et al. Caffeic acid ameliorates metabolic dysfunction-associated steatotic liver disease via alleviating oxidative damage and lipid accumulation in hepatocytes through activating Nrf2 via targeting Keap1. Free Radic Biol Med 2024; 224: 352-65.
161.
Minetti ET, Hamburg NM, Matsui R. Drivers of cardiovascular disease in metabolic dysfunction-associated steatotic liver disease: the threats of oxidative stress. Front Cardiovasc Med 2024; 11: 1469492.
162.
Nguyen MT, Lian A, Guilford FT, Venketaraman V. A literature review of glutathione therapy in ameliorating hepatic dysfunction in non-alcoholic fatty liver disease. Biomedicines 2025; 13: 644.
163.
Irie M, Sohda T, Anan A, Fukunaga A, Takata K, Tanaka T. Reduced glutathione suppresses oxidative stress in nonalcoholic fatty liver disease. Euroasian J Hepatogastroenterol 2016; 6: 13-8.
164.
Vincent A, Thauvin M, Quévrain E, et al. Evaluation of the compounds commonly known as superoxide dismutase and catalase mimics in cellular models. J Inorg Biochem 2021; 219: 3-12.
165.
Hwang I, Uddin MJ, Pak ES, et al. The impaired redox balance in peroxisomes of catalase knockout mice accelerates nonalcoholic fatty liver disease through endoplasmic reticulum stress. Free Radic Biol Med 2020; 148: 22-32.
166.
Uchiyama S, Shimizu T, Shirasawa T. CuZn-SOD deficiency causes ApoB degradation and induces hepatic lipid accumulation by impaired lipoprotein secretion in mice. J Biol Chem 2006; 281: 31713-9.
167.
Svobodová G, Šadibolová M, Velecká E, et al. Metabolic dysfunction-associated steatotic liver disease is accompanied by increased activities of superoxide dismutase, catalase, and carbonyl reductase 1 and levels of miR-200b-3p in mouse models. Antioxidants 2024; 13: 1371.
168.
Svobodová G, Horní M, Velecká E, Boušová I. Metabolic dysfunction-associated steatotic liver disease-induced changes in the antioxidant system: a review. Arch Toxicol 2024; 99: 1-22.
169.
Xu X, Poulsen KL, Wu L, et al. Targeted therapeutics and novel signaling pathways in non-alcohol-associated fatty liver/steatohepatitis (NAFL/NASH). Signal Transduct Target Ther 2022; 7: 287.
170.
Zare Z, Hosseinzadeh M, Sharuni F, et al. The effect of the dietary approaches to stop hypertension diet on total antioxidant capacity, superoxide dismutase, catalase, and body composition in patients with non-alcoholic fatty liver disease: a randomized controlled trial. Front Nutr 2023; 10: 1163516.
171.
Boll M, Weber LWD, Becker E, Stampfl A. Mechanism of carbon tetrachloride-induced hepatotoxicity. Hepatocellular damage by reactive carbon tetrachloride metabolites. Z Naturforsch C J Biosci 2001; 56: 649-59.
172.
Vrentzos E, Ikonomidis I, Pavlidis G, et al. Six-month supplementation with high dose coenzyme Q10 improves liver steatosis, endothelial, vascular and myocardial function in patients with metabolic-dysfunction associated steatotic liver disease: a randomized double-blind, placebo-controlled trial. Cardiovasc Diabetol 2024; 23: 245.
173.
Dludla PV, Orlando P, Silvestri S, et al. Coenzyme Q10 supplementation improves adipokine levels and alleviates inflammation and lipid peroxidation in conditions of metabolic syndrome: a meta-analysis of randomized controlled trials. Int J Mol Sci 2020; 21: 3247.
174.
Zhu M, Dagah OMA, Silaa BB, Lu J. Thioredoxin/glutaredoxin systems and gut microbiota in NAFLD: interplay, mechanism, and therapeutical potential. Antioxidants 2023; 12: 1680.
175.
Jiang Y, Han T, Zhang ZG, et al. The significance of 8-hydroxy-deoxyguanosine acid in the diagnosis of nonalcoholic steatohepatitis. Zhonghua Nei Ke Za Zhi 2017; 56: 34-8.
176.
AbuBakr HO, Aljuaydi SH, Abou-Zeid SM, El-Bahrawy A. Burn-induced multiple organ injury and protective effect of lutein in rats. Inflammation 2018; 41: 760-72.
177.
Martínez-Orgado J, Martínez-Vega M, Silva L, et al. Protein carbonylation as a biomarker of oxidative stress and a therapeutic target in neonatal brain damage. Antioxidants 2023; 12: 1839.
178.
Fedorova M, Bollineni RC, Hoffmann R. Protein carbonylation as a major hallmark of oxidative damage: update of analytical strategies. Mass Spectrom Rev 2014; 33: 79-97.
179.
Zhou M, Lv J, Chen X, Shi Y, Chao G, Zhang S. From gut to liver: exploring the crosstalk between gut-liver axis and oxidative stress in metabolic dysfunction-associated steatotic liver disease. Ann Hepatol 2025; 30: 101777.
180.
Klisic A, Malenica M, Kostadinovic J, Kocic G, Ninic A. Malondialdehyde as an independent predictor of body mass index in adolescent girls. J Med Biochem 2023; 42: 224-31.
181.
Cordeiro A, Costa R, Andrade N, et al. Does adipose tissue inflammation drive the development of non-alcoholic fatty liver disease in obesity? Clin Res Hepatol Gastroenterol 2020; 44: 394-402.
182.
Liu M, Cao B, Luo Q, Song Y, Liu K, Wu D. Association between serum uric acid-to-high-density lipoprotein cholesterol ratio and metabolic dysfunction-associated steatotic liver disease among chinese children with obesity. Front Endocrinol 2024; 15: 1474384.
183.
Zhao K, Zhang H, Ding W, et al. Adipokines regulate the development and progression of MASLD through organellar oxidative stress. Hepatol Commun 2025; 9: e0639.
184.
Mogna-Peláez P, Riezu-Boj JI, Milagro FI, et al. Inflammatory markers as diagnostic and precision nutrition tools for metabolic dysfunction-associated steatotic liver disease: results from the fatty liver in obesity trial. Clin Nutr 2024; 43: 1770-81.
185.
Torres S, Segalés P, García-Ruiz C, Fernandez-Checa JC. Mitochondria and the NLRP3 inflammasome in alcoholic and nonalcoholic steatohepatitis. Cells 2022; 11: 1475.
186.
Chen KQ, Ke BY, Cheng L, Yu XQ, Wang ZB, Wang SZ. Research and progress of inflammasomes in nonalcoholic fatty liver disease. Int Immunopharmacol 2023; 118: 110013.
187.
Ramos-Tovar E, Muriel P. NLRP3 inflammasome in hepatic diseases: a pharmacological target. Biochem Pharmacol 2023; 217: 115861.
188.
Hu H, Wang S, Chen C. Pathophysiological role and potential drug target of NLRP3 inflammasome in the metabolic disorders. Cell Signal 2024; 122: 111320.
189.
Vachliotis ID, Polyzos SA. The role of tumor necrosis factor-alpha in the pathogenesis and treatment of nonalcoholic fatty liver disease. Curr Obes Rep 2023; 12: 191-206.
190.
Burger K, Jung F, Baumann A, et al. TNFa is a key trigger of inflammation in diet-induced non-obese MASLD in mice. Redox Biol 2023; 66: 102870.
191.
Schramm C, Schneider A, Marx A, Lohse AW. Adalimumab could suppress the activity of non alcoholic steatohepatitis (NASH). Z Gastroenterol 2008; 46: 1369-71.
192.
Ubiaga CI, García Porrúa C, Pinto Tasende JA, Iñiguez Ubiaga LP, Sánchez-Andrade Fernandez A. Liver fibrosis 4 score: use in the evaluation of non-alcoholic fatty liver disease in patients with psoriatic arthritis. Reumatol Clin 2023: S2173-5743(23)00093-X.
193.
Mas E, Danjoux M, Garcia V, Carpentier S, Ségul B, Levade T. IL-6 deficiency attenuates murine diet-induced non-alcoholic steatohepatitis. PLoS One 2009; 4: e7929.
194.
Bucarey JL, Trujillo-González I, Paules EM, Espinosa A. Myokines and their potential protective role against oxidative stress in metabolic dysfunction-associated steatotic liver disease (MASLD). Antioxidants 2024; 13: 1363.
195.
Yamaguchi K, Itoh Y, Yokomizo C, et al. Blockade of interleukin-6 signaling enhances hepatic steatosis but improves liver injury in methionine choline-deficient diet-fed mice. Lab Investig 2010; 90: 1169-78.
196.
Fan X, Wang Y, Wang Y, et al. Dapagliflozin attenuates metabolic dysfunction-associated steatotic liver disease by inhibiting lipid accumulation, inflammation and liver fibrosis. BMC Pharmacol Toxicol 2025; 26: 59.
197.
Mueller L, von Seggern L, Schumacher J, et al. TNF-a similarly induces IL-6 and MCP-1 in fibroblasts from colorectal liver metastases and normal liver fibroblasts. Biochem Biophys Res Commun 2010; 397: 586-91.
198.
Liang B, Qiu X, Huang J, et al. Nonlinear associations of the hs-CRP/HDL-C index with metabolic dysfunction-associated steatotic liver disease and advanced liver fibrosis in US adults: insights from NHANES 2017-2018. Sci Rep 2025; 15: 4029.
199.
Ding Z, Wei Y, Peng J, Wang S, Chen G, Sun J. The potential role of c-reactive protein in metabolic-dysfunction-associated fatty liver disease and aging. Biomedicines 2023; 11: 2711.
200.
Wang JJ, Zheng Z, Zhang Y. Association of hematological biomarkers of inflammation with 10-year major adverse cardiovascular events and all-cause mortality in patients with metabolic dysfunction-associated steatotic liver disease: the ARIC study. J Inflamm Res 2024; 17: 4247-56.
201.
Baek SU, Yoon JH. High-sensitivity C-reactive protein levels in metabolic dysfunction-associated steatotic liver disease (MASLD), metabolic alcohol-associated liver disease (MetALD), and alcoholic liver disease (ALD) with metabolic dysfunction. Biomolecules 2024; 14: 1468.
202.
Gensluckner S, Wernly B, Datz C. Iron, oxidative stress, and metabolic dysfunction-associated steatotic liver disease. Antioxidants 2024, 13: 2.
203.
Peleman C, Francque S, Berghe TV. Emerging role of ferroptosis in metabolic dysfunction-associated steatotic liver disease: revisiting hepatic lipid peroxidation. eBioMedicine 2024; 102: 105088.
204.
Fujita K, Nozaki Y, Wada K, et al. Dysfunctional very-low-density lipoprotein synthesis and release is a key factor in nonalcoholic steatohepatitis pathogenesis. Hepatology 2009; 50: 772-80.
205.
Anastasopoulos NA, Barbouti A, Goussia AC, Christodoulou DK, Glantzounis GK. Exploring the role of metabolic hyperferritinaemia (MHF) in steatotic liver disease (SLD) and hepatocellular carcinoma (HCC). Cancers 2025; 17: 842.
206.
Nanizawa E, Tamaki Y, Yakura T, Otsuka S, Hatayama N, Naito M. The impact of a short-term high-fat diet on coagulation function in a mouse model and its role in exacerbating concanavalin A-induced liver injury. BMC Nutr 2024; 10: 158.
207.
Harrison SA, Gawrieh S, Roberts K, et al. Prospective evaluation of the prevalence of non-alcoholic fatty liver disease and steatohepatitis in a large middle-aged US cohort. J Hepatol 2021; 75: 284-91.
208.
Papatheodoridi M, De Ledinghen V, Lupsor-Platon M, et al. Agile scores in MASLD and ALD: external validation and their utility in clinical algorithms. J Hepatol 2024; 81: 590-9.
209.
Li SJ, Ding ST, Mersmann HJ, Chu CH, Hsu CD, Chen CY. A nutritional nonalcoholic steatohepatitis minipig model. J Nutr Biochem 2016; 28: 51-60.
210.
Jiang B, Wang D, Hu Y, et al. Serum amyloid A1 exacerbates hepatic steatosis via TLR4-mediated NF-KB signaling pathway. Mol Metab 2022; 59: 101462.
211.
Misiak B, Kowalski K, Stańczykiewicz B, et al. Appetite-regulating hormones in bipolar disorder: a systematic review and meta-analysis. Front Neuroendocrinol 2022; 67: 101013.
212.
Kořínková L, Pražienková V, Černá L, et al. Pathophysiology of NAFLD and NASH in experimental models: the role of food intake regulating peptides. Front Endocrinol 2020; 11: 597583.
213.
Corrado A, Colia R, Rotondo C, Sanpaolo E, Cantatore FP. Changes in serum adipokines profile and insulin resistance in patients with rheumatoid arthritis treated with anti-TNF-a. Curr Med Res Opin 2019; 35: 2197-205.
214.
Cantley J. The control of insulin secretion by adipokines: current evidence for adipocyte-beta cell endocrine signalling in metabolic homeostasis. Mamm Genome 2014; 25: 442-54.
215.
de Boom F, Talib A, Roebroek YGM, et al. Non-invasive assessment of metabolic dysfunction-associated steatotic liver disease in adolescents with severe obesity; patient characteristics and association with leptin – a cross-sectional overview of baseline data from a RCT. Children 2024; 11: 965.
216.
Mercantepe F, Baydur Sahin S, Cumhur Cure M, Karadag Z. Relationship between serum endocan levels and other predictors of endothelial dysfunction in obese women. Angiology 2023; 74: 948-57.
217.
Younossi ZM, Jarrar M, Nugent C, et al. A novel diagnostic biomarker panel for obesity-related nonalcoholic steatohepatitis (NASH). Obes Surg 2008; 18: 1430-7.
218.
Barbalho SM, Méndez-Sánchez N, Fornari Laurindo L. AdipoRon and ADP355, adiponectin receptor agonists, in metabolic-associated fatty liver disease (MAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Biochem Pharmacol 2023; 218: 115871.
219.
Ismaiel A, Ciornolutchii V, Herrera TE, et al. Adiponectin as a biomarker in liver cirrhosis – a systematic review and meta-analysis. Eur J Clin Invest 2025; 55: e14328.
220.
Younossi ZM, Page S, Rafiq N, et al. A biomarker panel for non-alcoholic steatohepatitis (NASH) and NASH-related fibrosis. Obes Surg 2011; 21: 431-9.
221.
Ismaiel A, Leucuta DC, Popa SL, Dumitrascu DL. Serum visfatin levels in nonalcoholic fatty liver disease and liver fibrosis: systematic review and meta-analysis. J Clin Med 2021; 10: 3029.
222.
Ku NO, Strnad P, Bantel H, Omary MB. Keratins: biomarkers and modulators of apoptotic and necrotic cell death in the liver. Hepatology 2016; 64: 966-76.
223.
Hempel F, Roderfeld M, Müntnich LJ, et al. Caspase-cleaved keratin 18 measurements identified ongoing liver injury after bariatric surgery. J Clin Med 2021; 10: 1233.
224.
Ugonabo O, Udoh UAS, Rajan PK, et al. The current status of the liver liquid biopsy in MASH related HCC: overview and future directions. Biomolecules 2023; 13: 1369.
225.
Sirniö P, Väyrynen JP, Mutt SJ, et al. Systemic inflammation is associated with circulating cell death released keratin 18 fragments in colorectal cancer. Oncoimmunology 2020; 9: 1783046.
226.
Charoensuk L, Thongpon P, Sitthirach C, et al. High-fat/high-fructose diet and opisthorchis viverrini infection promote metabolic dysfunction-associated steatotic liver disease via inflammation, fibrogenesis, and metabolic dysfunction. Acta Trop 2025; 261: 107491.
227.
Tamber SS, Bansal P, Sharma S, Singh RB, Sharma R. Biomarkers of liver diseases. Mol Biol Rep 2023; 50: 7815-23.
228.
Tang M, Jia H, Chen S, et al. Significance of MR/OPN/HMGB1 axis in NAFLD-associated hepatic fibrogenesis. Life Sci 2021; 264: 118619.
229.
Ai S, Pan J, Liu Q, et al. OTUB1 mediates PARP1 deubiquitination to alleviate NAFLD by regulating HMGB1. Exp Cell Res 2025; 445: 114425.
230.
Muraki M. Soluble Fas ligand, soluble Fas receptor, and decoy receptor 3 as disease biomarkers for clinical applications: a review. AIMS Med Sci 2022; 9: 98-267.
231.
Alkhouri N, Alisi A, Okwu V, et al. Circulating soluble Fas and Fas ligand levels are elevated in children with nonalcoholic steatohepatitis. Dig Dis Sci 2015; 60: 2353-9.
232.
Li Y, Yang P, Ye J, Xu Q, Wu J, Wang Y. Updated mechanisms of MASLD pathogenesis. Lipids Health Dis 2024; 23: 117.
233.
Niu L, Sulek K, Vasilopoulou CG, et al. Defining NASH from a multi-omics systems biology perspective. J Clin Med 2021; 10: 4673.
234.
Pirola CJ, Sookoian S. Multiomics biomarkers for the prediction of nonalcoholic fatty liver disease severity. World J Gastroenterol 2018; 24: 1601-15.
235.
Gallego-Durán R, Ampuero J, Maya-Miles D, et al. Fibroblast growth factor 21 is a hepatokine involved in MASLD progression. United Eur Gastroenterol J 2024; 12: 1056-68.
236.
Yilmaz Y, Eren F, Yonal O, et al. Increased serum FGF21 levels in patients with nonalcoholic fatty liver disease. Eur J Clin Invest 2010; 40: 887-92.
237.
Liu WY, Huang S, Shi KQ, et al. The role of fibroblast growth factor 21 in the pathogenesis of liver disease: a novel predictor and therapeutic target. Expert Opin Ther Targets 2014; 18: 1305-13.
238.
Sarkar S, Chen S, Spencer B, et al. Non-alcoholic steatohepatitis severity associates with FGF21 level and kidney glucose uptake. Metab Syndr Relat Disord 2021; 19: 491-7.
239.
Zhang H, Li Q, Chen H, et al. First in human study on tolerability, pharmacokinetics, and pharmacodynamics of single escalating doses of HEC88473, a novel dual GLP 1 and FGF21 receptor agonist in healthy and obese Chinese subjects. BioDrugs 2025; 39: 477-86.
240.
Flores-Guerrero JL, Post A, van Dijk PR, et al. Circulating trimethylamine-N-oxide is associated with all-cause mortality in subjects with nonalcoholic fatty liver disease. Liver Int 2021; 41: 2371-82.
241.
Termite F, Archilei S, D’Ambrosio F, et al. Gut microbiota at the crossroad of hepatic oxidative stress and MASLD. Antioxidants 2025; 14: 56.
242.
Nian F, Chen Y, Xia Q, Zhu C, Wu L, Lu X. Gut microbiota metabolite trimethylamine N-oxide promoted NAFLD progression by exacerbating intestinal barrier disruption and intrahepatic cellular imbalance. Int Immunopharmacol 2024; 142: 113173.
243.
Barrea L, Annunziata G, Muscogiuri G, et al. Trimethylamine-N-oxide (TMAO) as novel potential biomarker of early predictors of metabolic syndrome. Nutrients 2018; 10: 1971.
244.
Gątarek P, Kałużna-Czaplińska J. Trimethylamine N-oxide (TMAO) in human health. EXCLI J 2021; 20: 301-19.
245.
Janeiro MH, Ramírez MJ, Milagro FI, Martínez JA, Solas M. Implication of trimethylamine N-oxide (TMAO) in disease: potential biomarker or new therapeutic target. Nutrients 2018; 10: 1398.
246.
Farooqui N, Elhence A, Shalimar A. Current understanding of bile acids in chronic liver disease. J Clin Exp Hepatol 2022; 12: 155-73.
247.
Fang J, Yu CH, Li XJ, et al. Gut dysbiosis in nonalcoholic fatty liver disease: pathogenesis, diagnosis, and therapeutic implications. Front Cell Infect Microbiol 2022; 12: 997018.
248.
Liu Q, Liu S, Chen L, et al. Role and effective therapeutic target of gut microbiota in NAFLD/NASH (review). Exp Ther Med 2019; 18: 1935-44.
249.
Tan X, Liu Y, Long J, et al. Trimethylamine N-oxide aggravates liver steatosis through modulation of bile acid metabolism and inhibition of farnesoid X receptor signaling in nonalcoholic fatty liver disease. Mol Nutr Food Res 2019; 63: e1900257.
250.
Xie C, Halegoua-Demarzio D. Role of probiotics in non-alcoholic fatty liver disease: does gut microbiota matter? Nutrients 2019; 11: 2837.
251.
Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med 2018; 24: 908-22.
252.
Lodhi IJ, Semenkovich CF. Peroxisomes: a nexus for lipid metabolism and cellular signaling. Cell Metab 2014; 19: 380-92.
253.
Kazankov K, Barrera F, Møller HJ, et al. The macrophage activation marker sCD163 is associated with morphological disease stages in patients with non-alcoholic fatty liver disease. Liver Int 2016; 36: 1549-57. Erratum in: Liver Int 2017; 37: 1745.
254.
Jayakumar S, Harrison SA, Loomba R. Noninvasive markers of fibrosis and inflammation in nonalcoholic fatty liver disease. Curr Hepatol Rep 2016; 15: 86-95.
255.
Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest 2003; 112: 1796-808.
256.
Park HS, Park JY, Yu R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-alpha and IL-6. Diabetes Res Clin Pract 2005; 69: 29-35.
257.
Sookoian S, Rotman Y, Valenti L. Genetics of metabolic dysfunction-associated steatotic liver disease: the state of the art update. Clin Gastroenterol Hepatol 2024; 22: 2177-87.e3.
258.
Arechederra M, Recalde M, Gárate-Rascón M, Fernández-Barrena MG, Ávila MA, Berasain C. Epigenetic biomarkers for the diagnosis and treatment of liver disease. Cancers 2021; 13: 1265.
259.
Ha S, Wong VW, Zhang X, Yu J. Interplay between gut microbiome, host genetic and epigenetic modifications in MASLD and MASLD-related hepatocellular carcinoma. Gut 2025; 74: 141-52.
260.
Pingitore P, Romeo S. The role of PNPLA3 in health and disease. Biochim Biophys Acta Mol Cell Biol Lipids 2019; 1864: 900-6.
261.
Trépo E, Romeo S, Zucman-Rossi J, Nahon P. PNPLA3 gene in liver diseases. J Hepatol 2016; 65: 399-412.
262.
Romeo S, Kozlitina J, Xing C, et al. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat Genet 2008; 40: 1461-5.
263.
Salari N, Darvishi N, Mansouri K, et al. Association between PNPLA3 rs738409 polymorphism and nonalcoholic fatty liver disease: a systematic review and meta-analysis. BMC Endocr Disord 2021; 21: 125.
264.
Romeo S, Valenti L, Trépo E, Moreno C, Nahon P, Deltenre P. Potential role of oxidative DNA damage in the impact of PNPLA3 variant (rs 738409 C> G) in hepatocellular carcinoma risk. Hepatology 2014; 60: 1111-2.
265.
Donati B, Motta BM, Pingitore P, et al. The rs2294918 E434K variant modulates patatin-like phospholipase domain-containing 3 expression and liver damage. Hepatology 2016; 63: 787-98.
266.
Mahdessian H, Taxiarchis A, Popov S, et al. TM6SF2 is a regulator of liver fat metabolism influencing triglyceride secretion and hepatic lipid droplet content. Proc Natl Acad Sci 2014; 111: 8913-8.
267.
Newberry EP, Hall Z, Xie Y, et al. Liver-specific deletion of mouse Tm6sf2 promotes steatosis, fibrosis, and hepatocellular cancer. Hepatology 2021; 74: 1203-19.
268.
Holmen OL, Zhang HE, Fan Y, et al. Systematic evaluation of coding variation identifies a candidate causal variant in TM6SF2 influencing total cholesterol and myocardial infarction risk. Nat Genet 2014; 46: 345-51.
269.
Sookoian S, Castaño GO, Scian R, et al. Genetic variation in transmembrane 6 superfamily member 2 and the risk of nonalcoholic fatty liver disease and histological disease severity. Hepatology 2015; 61: 515-25.
270.
O’Hare EA, Yang R, Yerges-Armstrong LM, et al. TM6SF2 rs58542926 impacts lipid processing in liver and small intestine. Hepatology 2017; 65: 1526-42.
271.
Sun B, Ding X, Tan J, et al. TM6SF2 E167K variant decreases PNPLA3-mediated PUFA transfer to promote hepatic steatosis and injury in MASLD. Clin Mol Hepatol 2024; 30: 863.
272.
Varadharajan V, Massey WJ, Brown JM. Membrane-bound O-acyltransferase 7 (MBOAT7)-driven phosphatidylinositol remodeling in advanced liver disease. J Lipid Res 2022; 63: 100234.
273.
Mancina RM, Dongiovanni P, Petta S, et al. The MBOAT7-TMC4 variant rs641738 increases risk of nonalcoholic fatty liver disease in individuals of European descent. Gastroenterology 2016; 150: 1219-30.
274.
Varadharajan V, Ramachandiran I, Massey WJ, et al. Membrane-bound O-acyltransferase 7 (MBOAT7) shapes lysosomal lipid homeostasis and function to control alcohol-associated liver injury. Elife 2024; 12: RP92243.
275.
Teo K, Abeysekera KW, Adams L, et al. rs641738C> T near MBOAT7 is associated with liver fat, ALT and fibrosis in NAFLD: a meta-analysis. J Hepatol 2021; 74: 20-30.
276.
Alharthi J, Bayoumi A, Thabet K, et al. A metabolic associated fatty liver disease risk variant in MBOAT7 regulates toll like receptor induced outcomes. Nat Commun 2022; 13: 7430.
277.
Krawczyk M, Rau M, Schattenberg JM, et al.; NAFLD Clinical Study Group. Combined effects of the PNPLA3 rs738409, TM6SF2 rs58542926, and MBOAT7 rs641738 variants on NAFLD severity: a multicenter biopsy-based study. J Lipid Res 2017; 58: 247-55.
278.
Di Costanzo A, Belardinilli F, Bailetti D, et al. Evaluation of polygenic determinants of non-alcoholic fatty liver disease (NAFLD) by a candidate genes resequencing strategy. Sci Rep 2018; 8: 3702.
279.
Kanda T, Sasaki-Tanaka R, Abe H, et al. Polygenic risk score for metabolic dysfunction-associated steatotic liver disease and steatohepatitis: a narrative review. Int J Mol Sci 2025; 26: 5164.
280.
Chen VL, Du X, Oliveri A, et al. Genetic risk accentuates dietary effects on hepatic steatosis, inflammation and fibrosis in a population-based cohort. J Hepatol 2024; 81: 379-88.
281.
Longo M, Meroni M, Paolini E, et al. TM6SF2/PNPLA3/MBOAT7 loss-of-function genetic variants impact on NAFLD development and progression both in patients and in in vitro models. Cell Mol Gastroenterol Hepatol 2022; 13: 759-88.
282.
O’Brien J, Hayder H, Zayed Y, Peng C. Overview of microRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol 2018; 9: 402.
283.
Kloosterman WP, Plasterk RH. The diverse functions of microRNAs in animal development and disease. Develop Cell 2006; 11: 441-50.
284.
Valinezhad Orang A, Safaralizadeh R, Kazemzadeh-Bavili M. Mechanisms of miRNA-mediated gene regulation from common downregulation to mRNA-specific upregulation. Int J Genom 2014; 2014: 970607.
285.
Gallo A, Tandon M, Alevizos I, Illei GG. The majority of microRNAs detectable in serum and saliva is concentrated in exosomes. PLoS One 2012; 7: e30679.
286.
Murakami Y, Yasuda T, Saigo K, et al. Comprehensive analysis of microRNA expression patterns in hepatocellular carcinoma and non-tumorous tissues. Oncogene 2006; 25: 2537-45.
287.
Valdmanis PN, Kim HK, Chu K, et al. miR-122 removal in the liver activates imprinted microRNAs and enables more effective microRNA-mediated gene repression. Nat Commun 2018; 9: 5321.
288.
Jopling C. Liver-specific microRNA-122: biogenesis and function. RNA Biol 2012; 9: 137-42.
289.
Laudadio I, Manfroid I, Achouri Y, et al. A feedback loop between the liver-enriched transcription factor network and miR-122 controls hepatocyte differentiation. Gastroenterology 2012; 142: 119-29.
290.
Hochreuter MY, Dall M, Treebak JT, Barrès R. MicroRNAs in non-alcoholic fatty liver disease: progress and perspectives. Mol Metab 2022; 65: 101581.
291.
Long JK, Dai W, Zheng YW, Zhao SP. miR-122 promotes hepatic lipogenesis via inhibiting the LKB1/AMPK pathway by targeting Sirt1 in non-alcoholic fatty liver disease. Mol Med 2019; 25: 26.
292.
Wu GY, Rui C, Chen JQ, et al. MicroRNA-122 inhibits lipid droplet formation and hepatic triglyceride accumulation via Yin Yang 1. Cell Physiol Biochem 2017; 44: 1651-64.
293.
Juanola O, Martínez-López S, Francés R, Gómez-Hurtado I. Non-alcoholic fatty liver disease: metabolic, genetic, epigenetic and environmental risk factors. Int J Environ Res Public Health 2021; 18: 5227.
294.
Carpi S, Daniele S, de Almeida JF, Gabbia D. Recent advances in miRNA-based therapy for MASLD/MASH and MASH-associated HCC. Int J Mol Sci 2024; 25: 12229.
295.
Hsu KH, Wei CW, Su YR, et al. Upregulation of RelB in the miR-122 knockout mice contributes to increased levels of proinflammatory chemokines/cytokines in the liver and macrophages. Immunol Letters 2020; 226: 22-30.
296.
Castro RE, Ferreira DM, Afonso MB, et al. miR-34a/SIRT1/p53 is suppressed by ursodeoxycholic acid in the rat liver and activated by disease severity in human non-alcoholic fatty liver disease. J Hepatol 2013; 58: 119-25.
297.
Su Q, Kumar V, Sud N, Mahato RI. MicroRNAs in the pathogenesis and treatment of progressive liver injury in NAFLD and liver fibrosis. Adv Drug Deliv Rev 2018; 129: 54-63.
298.
Xu Y, Zalzala M, Xu J, Li Y, Yin L, Zhang Y. A metabolic stress-inducible miR-34a-HNF4a pathway regulates lipid and lipoprotein metabolism. Nat Commun 2015; 6: 7466.
299.
Luebke-Wheeler J, Zhang K, Battle M, et al. Hepatocyte nuclear factor 4a is implicated in endoplasmic reticulum stress–induced acute phase response by regulating expression of cyclic adenosine monophosphate responsive element binding protein H. Hepatology 2008; 48: 1242-50.
300.
Ding J, Li M, Wan X, et al. Effect of miR-34a in regulating steatosis by targeting PPARa expression in nonalcoholic fatty liver disease. Sci Rep 2015; 5: 13729.
301.
Choi SE, Fu T, Seok S, et al. Elevated micro RNA-34a in obesity reduces NAD+ levels and SIRT 1 activity by directly targeting NAMPT. Aging Cell 2013; 12: 1062-72.
302.
Shan W, Gao L, Zeng W, et al. Activation of the SIRT1/p66shc antiapoptosis pathway via carnosic acid-induced inhibition of miR-34a protects rats against nonalcoholic fatty liver disease. Cell Death Dis 2015; 6: e1833.
303.
Zhu H, Lin Y, Liu Y. miR-34a increases inflammation and oxidative stress levels in patients with necrotizing enterocolitis by downregulating SIRT1 expression. Mol Med Rep 2021; 24: 664.
304.
Caviglia JM, Yan J, Jang MK, et al. MicroRNA-21 and Dicer are dispensable for hepatic stellate cell activation and the development of liver fibrosis. Hepatology 2018; 67: 2414-29.
305.
Loyer X, Paradis V, Hénique C, et al. Liver microRNA-21 is overexpressed in non-alcoholic steatohepatitis and contributes to the disease in experimental models by inhibiting PPARa expression. Gut 2016; 65: 1882-94.
306.
Calo N, Ramadori P, Sobolewski C, et al. Stress-activated miR-21/miR-21* in hepatocytes promotes lipid and glucose metabolic disorders associated with high-fat diet consumption. Gut 2016; 65: 1871-81.
307.
López-Sánchez GN, Dóminguez-Pérez M, Uribe M, Chávez-Tapia NC, Nuño-Lámbarri N. Non-alcoholic fatty liver disease and microRNAs expression, how it affects the development and progression of the disease. Ann Hepatol 2021; 21: 100212.
308.
Ahn J, Lee H, Jung CH, Ha T. Lycopene inhibits hepatic steatosis via micro RNA-21-induced downregulation of fatty acid-binding protein 7 in mice fed a high-fat diet. Mol Nutr Food Res 2012; 56: 1665-74.
309.
Sun C, Huang F, Liu X, et al. miR-21 regulates triglyceride and cholesterol metabolism in non-alcoholic fatty liver disease by targeting HMGCR. Int J Mol Med 2015; 35: 847-53.
310.
Lu X, Yu Y, Tan S. The role of the miR-21-5p-mediated inflammatory pathway in ulcerative colitis. Exp Ther Med 2020; 19: 981-9.
311.
De Melo P, Alvarez AR, Serezani CH. Macrophage-derived miR-21 drives overwhelming glycolytic and inflammatory response during sepsis via repression of the PGE2/IL-10 axis. bioRxiv 2020.05.09.086421.
312.
Erceg S, Munjas J, Sopić M, et al. Expression analysis of circulating miR-21, miR-34a and miR-122 and redox status markers in metabolic dysfunction-associated steatotic liver disease patients with and without type 2 diabetes. Int J Mol Sci 2025; 26: 2392.
313.
Ren FJ, Yao Y, Cai XY, Fang GY. Emerging role of MiR-192-5p in human diseases. Front Pharmacol 2021; 12: 614068.
314.
Ragab HM, Ezzat WM, Hassan EM, et al. Significance of MiRNA-34a and MiRNA-192 as a risk factor for nonalcoholic fatty liver disease. J Genet Engin Biotechnol 2023; 21: 13.
315.
Lin Y, Ding D, Huang Q, et al. Downregulation of miR-192 causes hepatic steatosis and lipid accumulation by inducing SREBF1: novel mechanism for bisphenol A-triggered non-alcoholic fatty liver disease. Biochim Biophys Acta Mol Cell Biol Lipids 2017; 1862: 869-82.
316.
Liu XL, Cao HX, Wang BC, et al. miR-192-5p regulates lipid synthesis in non-alcoholic fatty liver disease through SCD-1. World J Gastroenterol 2017; 23: 8140.
317.
Paton CM, Ntambi JM. Biochemical and physiological function of stearoyl-CoA desaturase. Am J Physiol Endocrinol Metabol 2009; 297: E28-37.
318.
Ma L, Song H, Zhang CY, Hou D. MiR-192-5p ameliorates hepatic lipid metabolism in non-alcoholic fatty liver disease by targeting Yy1. Biomolecules 2023; 14: 34.
319.
Jaeger A, Zollinger L, Saely CH, et al. Circulating micro-RNAs-192 and-194 are associated with the presence and incidence of diabetes mellitus. Sci Rep 2018; 8: 14274.
320.
Csak T, Bala S, Lippai D, et al. MicroRNA-155 deficiency attenuates liver steatosis and fibrosis without reducing inflammation in a mouse model of steatohepatitis. PLoS One 2015; 10: e0129251.
321.
Wang L, Zhang N, Wang Z, Ai DM, Cao ZY, Pan HP. Decreased MiR-155 level in the peripheral blood of non-alcoholic fatty liver disease patients may serve as a biomarker and may influence LXR activity. Cell Physiol Biochem 2016; 39: 2239-48.
322.
Hu J, Chen X. miR-155 modulates fatty acid accumulation by targeting C/EBPb in free fatty acid-induced steatosis in HepG2 cells. Non-coding RNA Investig 2019; 3.
323.
Klisic A, Vujacic IR, Munjas J, Ninic A, Kotur-Stevuljevic J. Micro-ribonucleic acid modulation with oxidative stress and inflammation in patients with type 2 diabetes mellitus–a review article. Arch Med Sci 2022; 18: 870.
324.
Lei L, Zhou C, Yang X, Li L. Down-regulation of micro RNA-375 regulates adipokines and inhibits inflammatory cytokines by targeting AdipoR2 in non-alcoholic fatty liver disease. Clin Exp Pharmacol Physiol 2018; 45: 819-31.
325.
Lee J, Hong I, Lee C, Kim D, Kim S, Lee Y. SNPs in microRNA seed region and impact of miR-375 in concurrent regulation of multiple lipid accumulation-related genes. Sci Rep 2024; 14: 10924.
326.
Luo H, Zhao L, Dong B, Liu Y. MiR-375 inhibitor alleviates inflammation and oxidative stress by upregulating the GPR39 expression in atherosclerosis. Int Heart J 2024; 65: 135-45.
327.
Ando Y, Yamazaki M, Yamada H, et al. Association of circulating miR-20a, miR-27a, and miR-126 with non-alcoholic fatty liver disease in general population. Sci Rep 2019; 9: 18856.
328.
Tobaruela-Resola AL, Milagro FI, Elorz M, et al. Circulating miR-122-5p, miR-151a-3p, miR-126-5p and miR-21-5p as potential predictive biomarkers for metabolic dysfunction-associated steatotic liver disease assessment. J Physiol Biochem 2025; 81: 1275-88.
329.
Chu M, Zhao Y, Feng Y, et al. MicroRNA-126 participates in lipid metabolism in mammary epithelial cells. Mol Cell Endocrinol 2017; 454: 77-86.
330.
Wada K, Jahan I, Okamoto T, et al. MiR-126-3p and MiR-199a-3p promote lipid accumulation in 3T3-L1 adipocytes via regulating HIF-1a and C/EBPa expression. Shimane J Med Sci 2022; 39: 123-33.
331.
O’Brien K, Breyne K, Ughetto S, Laurent LC, Breakefield XO. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat Rev Mol Cell Biol 2020; 21: 585-606.
332.
Hu M, Huang H, Jia M, et al. A panel of miRNAs in the serum extracellular vesicles serve as novel diagnostic biomarkers for MASLD. Biomed J 2025; 6: 100838.
333.
Tobaruela-Resola AL, Riezu-Boj JI, Milagro FI, et al. Circulating microRNA panels in subjects with metabolic dysfunction-associated steatotic liver disease after following a 2-year dietary intervention. J Endocrinol Investig 2025; 48: 987-1003.
334.
Zhang JW, Ullah K, Khan N, Pan HT. Comprehensive profiling of serum microRNAs in normal and non-alcoholic fatty liver disease (NAFLD) patients. Sci Rep 2025; 15: 3766.
335.
Barchetta I, Zampieri M, Cimini FA, et al. Association between active dna demethylation and liver fibrosis in individuals with metabolic-associated steatotic liver disease (MASLD). Int J Mol Sci 2025; 26: 1271.
336.
Pirola CJ, Scian R, Gianotti TF, et al. Epigenetic modifications in the biology of nonalcoholic fatty liver disease: the role of DNA hydroxymethylation and TET proteins. Medicine 2015; 94: e1480.
337.
Younesian S, Mohammadi MH, Younesian O, Momeny M, Ghaffari SH, Bashash D. DNA methylation in human diseases. Heliyon 2024; 10: e32366.
338.
Li Y, Li L, Zhang Y, et al. Clinical and pathological characteristics of metabolic dysfunction-associated steatotic liver disease and the key role of epigenetic regulation: implications for molecular mechanism and treatment. Ther Adv Endocrinol Metab 2025; 16: 20420188251321602.
339.
Theys C, Vanderhaeghen T, Van Dijck E, et al. Loss of PPARa function promotes epigenetic dysregulation of lipid homeostasis driving ferroptosis and pyroptosis lipotoxicity in metabolic dysfunction associated Steatotic liver disease (MASLD). Front Mol Med 2024; 3: 1283170.
340.
Zeybel M, Hardy T, Robinson SM, et al. Differential DNA methylation of genes involved in fibrosis progression in non-alcoholic fatty liver disease and alcoholic liver disease. Clin Epig 2015; 7: 1-1.
341.
Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, et al. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology 2023; 77: 1797-835.
342.
Papaefthymiou A, Sarrou S, Pateras K, et al. The effect of biologic agents on steatotic liver disease in patients with inflammatory bowel disease: a prospective, open-label comparative trial. Pharmaceuticals 2024; 17: 1432.
343.
Tobaruela-Resola AL, Milagro FI, Mogna-Pelaez P, et al. The use of circulating miRNAs for the diagnosis, prognosis, and personalized treatment of MASLD. J Physiol Biochem 2025; 81: 589-609.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.