Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
OBSTETRICS AND GYNAECOLOGY / RESEARCH LETTER
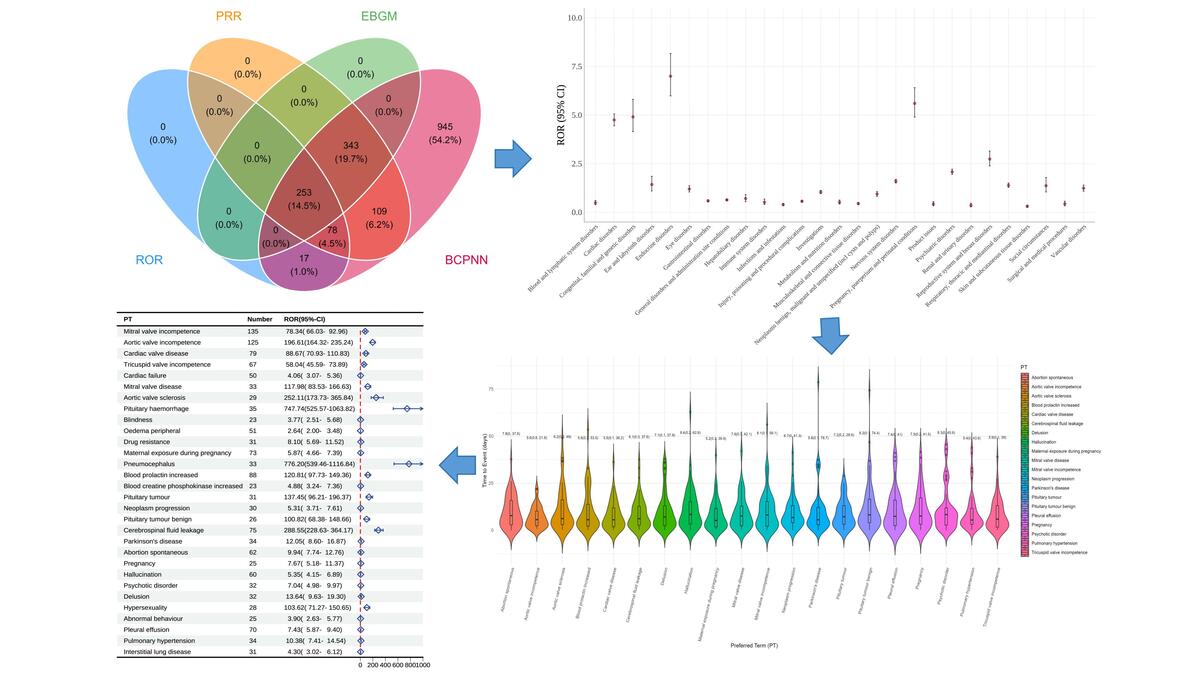
A real-world pharmacovigilance analysis of cardiac, endocrine, and neurological adverse events associated with cabergoline: a disproportionality study
1
Department of Pharmacy, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou, China
2
Department of Pharmacy, The Fourth Affiliated Hospital of School of Medicine, and International School of Medicine, International Institutes of Medicine, Zhejiang University, Yiwu, China
3
Department of Pharmacy, The Quzhou Affiliated Hospital of Wenzhou Medical University, Quzhou People’s Hospital, Quzhou, China
4
Department of Pharmacy, The First People’s Hospital of Wuyi County, Jinhua, China
These authors had equal contribution to this work
Submission date: 2026-02-05
Final revision date: 2026-04-03
Acceptance date: 2026-04-26
Online publication date: 2026-04-30
Publication date: 2026-04-30
Corresponding author
Jun-Jie Xu
Department of Pharmacy The First People’s Hospital of Wuyi County No. 2 Nanmen Street Jinhua 321200, China
Department of Pharmacy The First People’s Hospital of Wuyi County No. 2 Nanmen Street Jinhua 321200, China
Kai-Li Mao
Department of Pharmacy The Quzhou Affiliated Hospital of Wenzhou Medical University Quzhou People’s Hospital No. 100 Minjiang Avenue Kecheng District Quzhou 324000, China
Department of Pharmacy The Quzhou Affiliated Hospital of Wenzhou Medical University Quzhou People’s Hospital No. 100 Minjiang Avenue Kecheng District Quzhou 324000, China
Arch Med Sci 2026;22(2):1224-1233
TOPICS
REFERENCES (29)
1.
Uygur MM, Menotti S, Santoro S, Giustina A. Modern approach to bone comorbidity in prolactinoma. Pituitary 2024; 27: 802-12.
2.
Haider SA, Levy S, Rock JP, Craig JR. Prolactinoma: medical and surgical considerations. Otolaryngol Clin North Am 2022; 55: 305-14.
3.
Castinetti F, Albarel F, Amodru V, et al. The risks of medical treatment of prolactinoma. Ann Endocrinol (Paris) 2021; 82: 15-9.
4.
Henkel A, Johnson SA, Reeves MF, Cahill EP, Blumenthal PD, Shaw KA. Cabergoline for lactation inhibition after second-trimester abortion or pregnancy loss: a randomized controlled trial. Obstet Gynecol 2023; 141: 1115-23.
5.
Witwit SJ. Improving pregnancy rate in infertile patients with polycystic ovarian syndrome receiving clomiphene citrate and cabergoline in euprolactinomic women in single cycle treatment. Ginekol Pol 2023; 94: 456-62.
6.
Tang H, Mourad SM, Wang A, Zhai SD, Hart RJ. Dopamine agonists for preventing ovarian hyperstimulation syndrome. Cochrane Database Syst Rev 2021; 4: CD008605.
7.
Mohammad MM, Alidrisi HA, Mansour AA. Impulse control disorders in southern iraqi patients medicated with cabergoline for prolactinoma. Cureus 2024; 16: e58516.
8.
Yüksel RN, Elyas Kaya Z, Dilbaz N, Cingi Yirün M. Cabergoline-induced manic episode: case report. Ther Adv Psychopharmacol 2016; 6: 229-31.
9.
De Vecchis R, Esposito C, Ariano C. Cabergoline use and risk of fibrosis and insufficiency of cardiac valves. Meta-analysis of observational studies. Herz 2013; 38: 868-80.
10.
Delgado V, Biermasz NR, van Thiel SW, et al. Changes in heart valve structure and function in patients treated with dopamine agonists for prolactinomas, a 2-year follow-up study. Clin Endocrinol (Oxf) 2012; 77: 99-105.
11.
Scavone C, Stelitano B, Rafaniello C, Rossi F, Sportiello L, Capuano A. Drugs-induced pathological gambling: an analysis of Italian Spontaneous Reporting System. J Gambl Stud 2020; 36: 85-96.
12.
Sridharan K. Drugs associated with tachyphylaxis: results from a retrospective pharmacovigilance study using disproportionality analysis. Eur J Hosp Pharm 2026; 33: 183-6.
13.
Li LC, Zhang ZH, Chen MM, Xu F, Mao KL. Bleeding and antiplatelet therapies: a combined pharmacovigilance analysis of the FDA Adverse Event Reporting System and Japanese Adverse Drug Event Report databases, and the role of pharmacists. Int J Surg 2026; 112: 3612-24.
14.
Ogrinc G, Davies L, Goodman D, Batalden P, Davidoff F, Stevens D. SQUIRE 2.0 (Standards for QUality Improvement Reporting Excellence): revised publication guidelines from a detailed consensus process. BMJ Qual Saf 2016; 25: 986-92.
15.
Agha RA, Mathew G, Rashid R, et al. Transparency In The reporting of Artificial INtelligence – the TITAN guideline. Premier J Sci 2025; 10: 100082.
16.
Kekewska A, Hübner H, Gmeiner P, Pertz HH. The bulky N6 substituent of cabergoline is responsible for agonism of this drug at 5-hydroxytryptamine 5-HT2A and 5-HT2B receptors and thus is a determinant of valvular heart disease. J Pharmacol Exp Ther 2011; 338: 381-91.
17.
Otis AS, Brochet MS, Tremblay Z, Balayla J, Dahdouh EM. Cabergoline use and pregnancy outcomes: a systematic review. Birth Defects Res 2025; 117: e2464.
18.
Harris K, Murphy KE, Horn D, MacGilivray J, Yudin MH. Safety of cabergoline for postpartum lactation inhibition or suppression: a systematic review. J Obstet Gynaecol Can 2020; 42: 308-15.e20.
19.
Ma K, Ma L, Huang T, et al. The effectiveness and safety of aripiprazole, bromocriptine, and cabergoline in the treatment of hyperprolactinemia: a systematic review and network meta-analysis. Expert Opin Drug Saf 2025; 24: 773-86.
20.
Boucoiran I, Roy M, Poliquin V, et al. Evaluation of cabergoline for lactation inhibition in women living with HIV. Int J STD AIDS 2021; 32: 654-61.
21.
Dayan-Schwartz A, Yefet E, Massalha M, et al. The efficiency of cabergoline vs pyridoxine for lactation inhibition-a randomized controlled trial. Am J Obstet Gynecol 2024; 230: 561.e1-e8.
22.
Lai JS, Gangwani RA. Medication-induced acute angle closure attack. Hong Kong Med J 2012; 18: 139-45.
23.
Elabd SS, Ahmad MM, Qetab SQ, Almalki MH. Cabergoline-induced pneumocephalus following treatment for giant invasive macroprolactinoma presenting with spontaneous cerebrospinal fluid rhinorrhea. Clin Med Insights Endocrinol Diabetes 2018; 11: 1179551418758640.
24.
Lorenc A, Howell A, Jawad H, et al. The education process for pharmacists in Poland: standards and proposed changes. Arch Med Sci 2024; 20: 1057-62.
25.
Li LC, Sun W, Lv XQ, Xu YY, Hu Y, Shi JN. Escitalopram-induced sinus bradycardia in coronary heart disease combined with depression: a case report and review of literature. Front Cardiovasc Med 2023; 10: 1133662.
26.
Li LC, Zhang S, Sun JB, Xu YY, Mao KL. Case report: practice of whole-course pharmaceutical care: a case in hypertrophic cardiomyopathy and a review of the pharmacist’s role. Front Cardiovasc Med 2025; 12: 1512784.
27.
Wang YZ, Chen J, Pei SQ, et al. Treatment strategies and pharmacist-led medication management for Helicobacter pylori infection. Drug Dev Res 2023; 84: 326-36.
28.
Janani Buddhika RB, Pathirana D, Chathumali VGI, Wickraramasinghe NDD. Knowledge, attitudes, and practices among oncology pharmacists on cytotoxic drug reconstitution in Sri Lanka: a cross-sectional study. J Oncol Pharm Pract 2025: 10781552251361801.
29.
Lin YH, Wu YC, Lin YT, et al. Associations of preoperative Oswestry Disability Index and EuroQol-5D with long-term all-cause mortality in patients undergoing percutaneous vertebroplasty. Arch Med Sci 2025; 21: 1381-7.
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.