Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
NEPHROLOGY / BASIC RESEARCH
Exploring causality between peripheral blood B cell subtypes and membranous nephropathy: a two-sample Mendelian randomization study
1
Department of Nephrology, The Sixth Medical Center of PLA General Hospital, Beijing, China
These authors had equal contribution to this work
Submission date: 2025-01-03
Final revision date: 2025-04-23
Acceptance date: 2025-05-14
Online publication date: 2025-06-24
Publication date: 2026-04-30
Corresponding author
Zhiyong Zhang
Department of Nephrology The Sixth Medical Center of PLA General Hospital Beijing, 100048, China Phone: +86 13601023587
Department of Nephrology The Sixth Medical Center of PLA General Hospital Beijing, 100048, China Phone: +86 13601023587
Arch Med Sci 2026;22(2):991-1000
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Membranous nephropathy (MN) is a glomerular autoimmune disease associated with nephrotic syndrome. This study explored the influence of peripheral blood B cell subtypes on MN using Mendelian randomization (MR).
Material and methods:
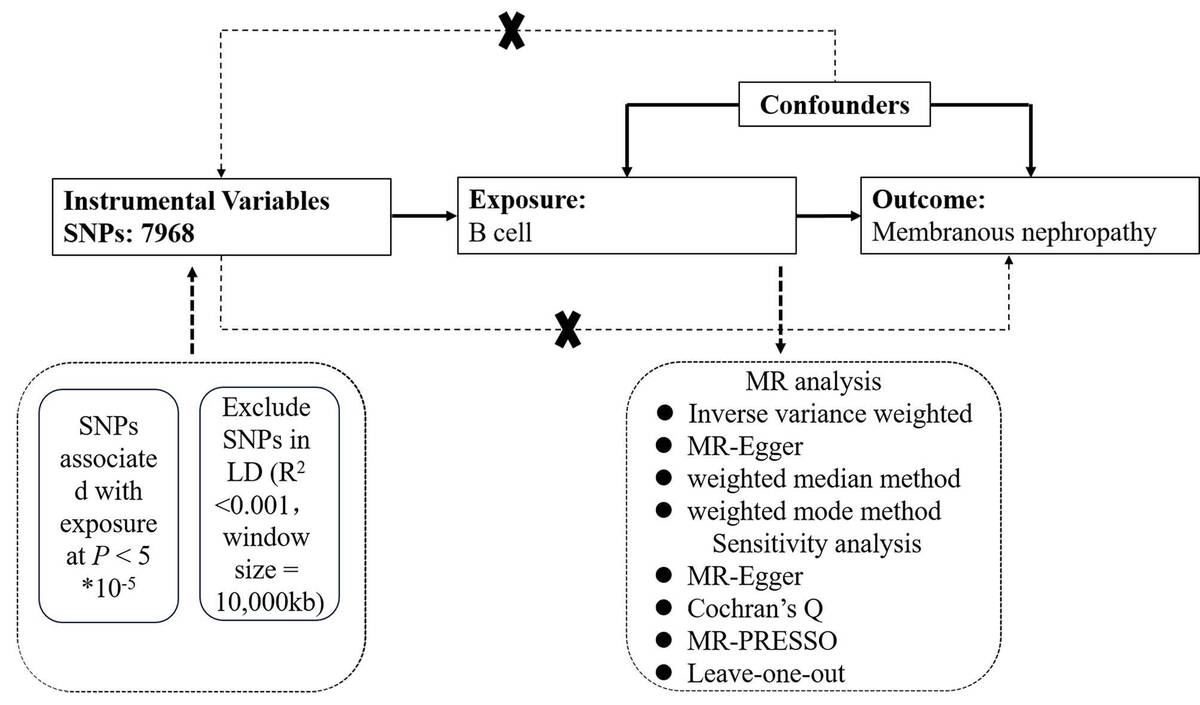
Data on single-nucleotide polymorphisms (SNPs) associated with peripheral blood B cells and MN were obtained from a genome-wide association study (GWAS). Analytical methods included instrumental variable weighted (IVW), weighted median, weighted mode methods, and MR-Egger regression. Sensitivity analyses were conducted using MR-Egger, Mendelian Randomization Pleiotropy RESidual Sum and Outlier (MR-PRESSO) for outlier detection, Cochran’s Q test for heterogeneity, and leave-one-out analysis to assess the robustness of the findings.
Results:
Higher levels of IgD+ CD24-B cell absolute count (OR = 0.8285, 95% CI: 0.7317–0.9381, p = 0.003), B-cell activating factor receptor (BAFF-R) on IgD+ CD24-B cells (OR 0.9045, 95% CI: 0.8275-0.9886, p = 0.0269), BAFF-R on IgD+ CD38dim B cells (OR = 0.9057, 95% CI: 0.8277–0.991, p = 0.0311), BAFF-R on IgD- CD27-B cells (OR = 0.9134, 95% CI: 0.8404–0.9928, p = 0.0332), CD19 on IgD-CD24-B cells (OR = 0.884, 95% CI: 0.7906–0.9886, p = 0.0306), CD24 on switched memory B cells (OR = 0.8927, 95% CI: 0.8133–0.9798, p = 0.0169), and CD25 on switched memory B cells (OR = 0.8768, 95% CI: 0.7745–0.9927, p = 0.0379) were strongly associated with an decreased risk of membranous nephropathy. Sensitivity analyses were conducted to confirm the stability of the findings.
Conclusions:
This MR study supports the possibility of a genetic causal association between peripheral blood B cell subtypes and MN. The results improve our understanding of the immunological basis of MN and may inform the development of personalized medicine.
Membranous nephropathy (MN) is a glomerular autoimmune disease associated with nephrotic syndrome. This study explored the influence of peripheral blood B cell subtypes on MN using Mendelian randomization (MR).
Material and methods:
Data on single-nucleotide polymorphisms (SNPs) associated with peripheral blood B cells and MN were obtained from a genome-wide association study (GWAS). Analytical methods included instrumental variable weighted (IVW), weighted median, weighted mode methods, and MR-Egger regression. Sensitivity analyses were conducted using MR-Egger, Mendelian Randomization Pleiotropy RESidual Sum and Outlier (MR-PRESSO) for outlier detection, Cochran’s Q test for heterogeneity, and leave-one-out analysis to assess the robustness of the findings.
Results:
Higher levels of IgD+ CD24-B cell absolute count (OR = 0.8285, 95% CI: 0.7317–0.9381, p = 0.003), B-cell activating factor receptor (BAFF-R) on IgD+ CD24-B cells (OR 0.9045, 95% CI: 0.8275-0.9886, p = 0.0269), BAFF-R on IgD+ CD38dim B cells (OR = 0.9057, 95% CI: 0.8277–0.991, p = 0.0311), BAFF-R on IgD- CD27-B cells (OR = 0.9134, 95% CI: 0.8404–0.9928, p = 0.0332), CD19 on IgD-CD24-B cells (OR = 0.884, 95% CI: 0.7906–0.9886, p = 0.0306), CD24 on switched memory B cells (OR = 0.8927, 95% CI: 0.8133–0.9798, p = 0.0169), and CD25 on switched memory B cells (OR = 0.8768, 95% CI: 0.7745–0.9927, p = 0.0379) were strongly associated with an decreased risk of membranous nephropathy. Sensitivity analyses were conducted to confirm the stability of the findings.
Conclusions:
This MR study supports the possibility of a genetic causal association between peripheral blood B cell subtypes and MN. The results improve our understanding of the immunological basis of MN and may inform the development of personalized medicine.
REFERENCES (39)
1.
Couser WG. Primary membranous nephropathy. Clin J Am Soc Nephrol 2017; 12: 983-97.
2.
Ronco P, Beck L, Debiec H, et al. Membranous nephropathy. Nat Rev Dis Primers 2021; 7: 69.
3.
Cattran DC, Brenchley PE. Membranous nephropathy: integrating basic science into improved clinical management. Kidney Int 2017; 91: 566-74.
4.
De Vriese AS, Glassock RJ, Nath KA, Sethi S, Fervenza FC. A proposal for a serology-based approach to membranous nephropathy. J Am Soc Nephrol 2017; 28: 421-30.
5.
Kumar V, Ramachandran R, Kumar A, et al. Antibodies to m-type phospholipase A2 receptor in children with idiopathic membranous nephropathy. Nephrology (Carlton) 2015; 20: 572-5.
6.
Debiec H, Ronco P. Immunopathogenesis of membranous nephropathy: an update. Semin Immunopathol 2014; 36: 381-97.
7.
McGrogan A, Franssen CF, de Vries CS. The incidence of primary glomerulonephritis worldwide: a systematic review of the literature. Nephrol Dial Transplant 2011; 26: 414-30.
9.
Claudio P. Primary membranous nephropathy: an endless story. J Nephrol 2023; 36: 563-74.
11.
Rosser EC, Mauri C. Regulatory B cells: origin, phenotype, and function. Immunity 2015; 42: 607-12.
12.
LeBien TW, Tedder TF. B lymphocytes: how they develop and function. Blood 2008; 112: 1570-80.
13.
Wu Y, Jiang H, Hu Y, et al. B cell dysregulation and depletion therapy in primary membranous nephropathy: prospects and potential challenges. Int Immunopharmacol 2024; 140: 112769.
14.
Cantarelli C, Jarque M, Angeletti A, et al. A comprehensive phenotypic and functional immune analysis unravels circulating anti-phospholipase A2 receptor antibody secreting cells in membranous nephropathy patients. Kidney Int Rep 2020; 5: 1764-76.
15.
Shi M, Wang Y, Zhang H, et al. Single-cell RNA sequencing shows the immune cell landscape in the kidneys of patients with idiopathic membranous nephropathy. Front Immunol 2023; 14: 1203062.
16.
Cohen CD, Calvaresi N, Armelloni S, et al. CD20-positive infiltrates in human membranous glomerulonephritis. J Nephrol 2005; 18: 328-33.
17.
Wang ZF, Cheng YC, Li YQ, Liu L, Ge SW, Xu G. Characteristics and prognostic value of tertiary lymphoid organs in membranous nephropathy: a retrospective study. Front Med (Lausanne) 2021; 8: 803929.
18.
Emdin CA, Khera AV, Kathiresan S. Mendelian randomization. JAMA 2017; 318: 1925-6.
19.
Skrivankova VW, Richmond RC, Woolf BAR, et al. Strengthening the reporting of observational studies in epidemiology using mendelian randomization: the STROBE-MR Statement. JAMA 2021; 326: 1614-21.
20.
Davies NM, Holmes MV, Davey Smith G. Reading Mendelian randomisation studies: a guide, glossary, and checklist for clinicians. BMJ 2018; 362: k601.
21.
Sanderson E, Glymour MM, Holmes MV, et al. Mendelian randomization. Nat Rev Methods Primers 2022; 2: 6.
22.
Xie J, Liu L, Mladkova N, et al. The genetic architecture of membranous nephropathy and its potential to improve non-invasive diagnosis. Nat Commun 2020; 11: 1600.
23.
Pierce BL, Ahsan H, Vanderweele TJ. Power and instrument strength requirements for Mendelian randomization studies using multiple genetic variants. Int J Epidemiol 2011; 40: 740-52.
24.
Davey Smith G, Hemani G. Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet 2014; 23: R89-98.
25.
Pierce BL, Burgess S. Efficient design for Mendelian randomization studies: subsample and 2-sample instrumental variable estimators. Am J Epidemiol 2013; 178: 1177-84.
26.
Hemani G, Bowden J, Davey Smith G. Evaluating the potential role of pleiotropy in Mendelian randomization studies. Hum Mol Genet 2018; 27: R195-208.
27.
Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 2015; 44: 512-25.
28.
Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol 2016; 40: 304-14.
29.
Bowden J, Del Greco MF, Minelli C, Davey Smith G, Sheehan N, Thompson J. A framework for the investigation of pleiotropy in two-sample summary data Mendelian randomization. Stat Med 2017; 36: 1783-802.
30.
Cai A, Meng Y, Zhou H, et al. Podocyte pathogenic bone morphogenetic protein-2 pathway and immune cell behaviors in primary membranous nephropathy. Adv Sci (Weinh) 2024; 11: e2404151.
31.
Wu G, Peng YM, Liu H, et al. Expression of CD19(+)CD5(+)B cells and IgA1-positive cells in tonsillar tissues of IgA nephropathy patients. Ren Fail 2011; 33: 159-63.
32.
Wu G, Peng YM, Liu FY, Xu D, Liu C. The role of memory B cell in tonsil and peripheral blood in the clinical progression of IgA nephropathy. Hum Immunol 2013; 74: 708-12.
33.
Deng B, Deng L, Liu M, et al. Elevated circulating CD19(+)CD24(hi)CD38(hi) B cells display pro-inflammatory phenotype in idiopathic membranous nephropathy. Immunol Lett 2023; 261: 58-65.
34.
Su Z, Jin Y, Zhang Y, et al. The diagnostic and prognostic potential of the B-cell repertoire in membranous nephropathy. Front Immunol 2021; 12: 635326.
35.
So BYF, Yap DYH, Chan TM. B cells in primary membranous nephropathy: escape from immune tolerance and implications for patient management. Int J Mol Sci 2021; 22: 13560.
36.
Han SS, Yang SH, Jo HA, et al. BAFF and APRIL expression as an autoimmune signature of membranous nephropathy. Oncotarget 2018; 9: 3292-302.
37.
Netti GS, Infante B, Spadaccino F, et al. Serum levels of BAFF and APRIL predict clinical response in anti-PLA2R-positive primary membranous nephropathy. J Immunol Res 2019; 2019: 8483650.
38.
Sakkas LI, Daoussis D, Mavropoulos A, Liossis SN, Bogdanos DP. Regulatory B cells: new players in inflammatory and autoimmune rheumatic diseases. Semin Arthritis Rheum 2019; 48: 1133-41.
39.
Lutz J. Pathophysiology, diagnosis, and treatment of membranous nephropathy. Nephrol Ther 2021; 17s: S1-10.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.