Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
DIABETOLOGY / BASIC RESEARCH
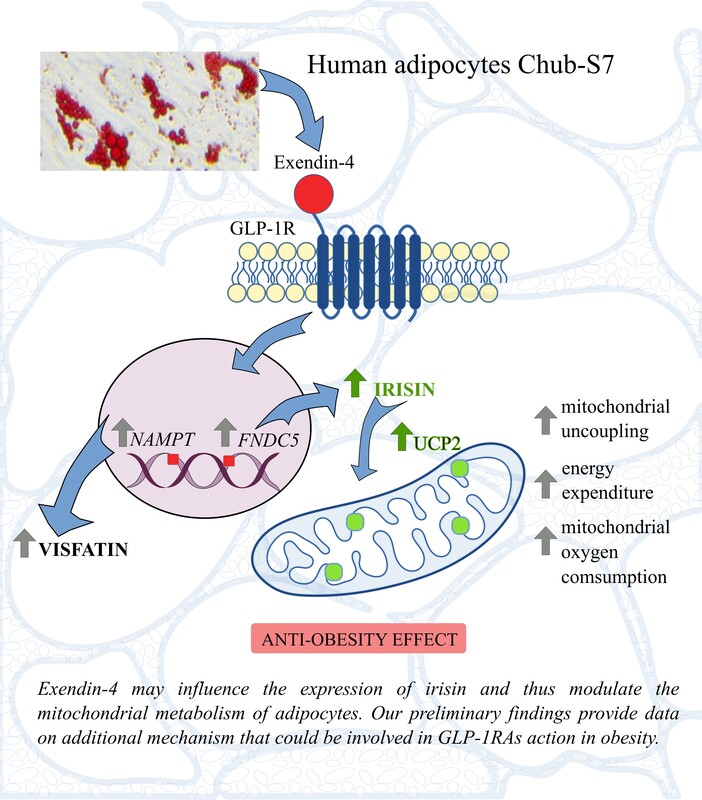
Irisin may be involved in exendin-4 mitochondrial action in human adipocytes
1
Department of Clinical Biochemistry, Jagiellonian University Medical College, Krakow, Poland
Submission date: 2025-07-04
Final revision date: 2025-11-25
Acceptance date: 2025-11-25
Online publication date: 2026-04-03
Publication date: 2026-04-30
Corresponding author
Joanna Góralska
Department of Clinical Biochemistry Jagiellonian University Medical College Krakow, Poland
Department of Clinical Biochemistry Jagiellonian University Medical College Krakow, Poland
Arch Med Sci 2026;22(2):958-967
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Disturbed mitochondrial activity in adipocytes has been proposed as one of the mechanisms involved in metabolic dysfunction in obesity. Glucagon-like peptide receptor agonists (GLP-1RAs) are used to normalize glucose level and reduce body weight. GLP-1 activates intracellular pathways similar to those of irisin, a peptide that modulates metabolism by stimulating the ‘browning’ of adipocytes. The aim of the study was to investigate the mechanisms of action of the GLP-1RA exendin-4 at the mRNA, protein, and mitochondrial levels in human adipocytes.
Material and methods:
Human Chub-S7 preadipocytes were differentiated in vitro to mature adipocytes and then stimulated with exendin-4 at 100 nM for 24 h. Expression levels of mRNA and proteins (irisin, adiponectin, visfatin/NAMPT) were measured. Oxygen consumption rates and intracellular ATP content were determined.
Results:
Exendin-4 enhanced the secretion of irisin and visfatin by adipocytes. Upregulated expression of FNDC5, NAMPT, and UCP2 genes was accompanied by modest changes in mitochondrial activity in exendin-4-treated adipocytes. Exendin-4 exerted a similar effect on mitochondrial oxygen consumption rates as irisin, including increased maximum mitochondrial respiration and reserve capacity with unchanged intracellular ATP.
Conclusions:
Increasing energy expenditure by exendin-4 may be associated with upregulation of irisin in human adipocytes. Clinical studies are necessary to confirm the hypothesis that nutrients, by stimulating the secretion of GLP-1, may influence the expression of irisin and thus modulate the mitochondrial metabolism of adipocytes.
Disturbed mitochondrial activity in adipocytes has been proposed as one of the mechanisms involved in metabolic dysfunction in obesity. Glucagon-like peptide receptor agonists (GLP-1RAs) are used to normalize glucose level and reduce body weight. GLP-1 activates intracellular pathways similar to those of irisin, a peptide that modulates metabolism by stimulating the ‘browning’ of adipocytes. The aim of the study was to investigate the mechanisms of action of the GLP-1RA exendin-4 at the mRNA, protein, and mitochondrial levels in human adipocytes.
Material and methods:
Human Chub-S7 preadipocytes were differentiated in vitro to mature adipocytes and then stimulated with exendin-4 at 100 nM for 24 h. Expression levels of mRNA and proteins (irisin, adiponectin, visfatin/NAMPT) were measured. Oxygen consumption rates and intracellular ATP content were determined.
Results:
Exendin-4 enhanced the secretion of irisin and visfatin by adipocytes. Upregulated expression of FNDC5, NAMPT, and UCP2 genes was accompanied by modest changes in mitochondrial activity in exendin-4-treated adipocytes. Exendin-4 exerted a similar effect on mitochondrial oxygen consumption rates as irisin, including increased maximum mitochondrial respiration and reserve capacity with unchanged intracellular ATP.
Conclusions:
Increasing energy expenditure by exendin-4 may be associated with upregulation of irisin in human adipocytes. Clinical studies are necessary to confirm the hypothesis that nutrients, by stimulating the secretion of GLP-1, may influence the expression of irisin and thus modulate the mitochondrial metabolism of adipocytes.
REFERENCES (48)
1.
Brauer M, Roth GA, Aravkin AY, et al. Global burden and strength of evidence for 88 risk factors in 204 countries and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 2024; 403: 2162-203.
2.
Horwitz A, Birk R. Adipose tissue hyperplasia and hypertrophy in common and syndromic obesity – the case of bbs obesity. Nutrients 2023; 15: 3445.
3.
de Mello A, Costa AB, Engel JDG, Rezin GT. Mitochondrial dysfunction in obesity. Life Sci 2018; 192: 26-32.
4.
Patti ME, Corvera S. The role of mitochondria in the pathogenesis of type 2 diabetes. Endocr Rev 2010; 31: 364-95.
5.
Liu S, Cui F, Ning K, et al. Role of irisin in physiology and pathology. Front Endocrinol (Lausanne) 2022; 13: 962968.
6.
Al-Amrani A, AbdelKarim M, AlZabin M, Alzoghaibi M. Low expression of brown and beige fat genes in subcutaneous tissues in obese patients. Arch Med Sci 2019; 15: 1113-22.
7.
Flori L, Testai L, Calderone V. The “Irisin System”: from biological roles to pharmacological and nutraceutical perspectives. Life Sci 2021; 267: 118954.
8.
Korta P, Pocheć E, Mazur-Biały A. Irisin as a multifunctional protein: implications for health and certain diseases. Medicina (Lithuania) 2019; 55: 485.
9.
Slate-Romano JJ, Yano N, Zhao TC. Irisin reduces inflammatory signaling pathways in inflammation-mediated metabolic syndrome. Mol Cell Endocrinol 2022; 552: 111676.
10.
Bao JF, She QY, Hu PP, Jia N, Li A. Irisin, a fascinating field in our times. Trends Endocrinol Metab 2022; 33: 601-13.
11.
Huh JY, Dincer F, Mesfum E, Mantzoros CS. Irisin stimulates muscle growth-related genes and regulates adipocyte differentiation and metabolism in humans. Int J Obes 2014; 38: 1538-44.
13.
Nauck MA, Meier JJ. Incretin hormones: their role in health and disease. Diabetes Obes Metab 2018; 20: 5-21.
14.
Chia CW, Egan JM. Incretins in obesity and diabetes. Ann N Y Acad Sci 2020; 1461: 104-26.
15.
Nauck MA, Quast DR, Wefers J, Pfeiffer AFH. The evolving story of incretins (GIP and GLP-1) in metabolic and cardiovascular disease: a pathophysiological update. Diabetes Obes Metab 2021; 23: 5-29.
16.
Garber AJ. Novel GLP-1 receptor agonists for diabetes. Expert Opin Investig Drugs 2012; 21: 45-57.
17.
Lorza-Gil E, Strauss OD, Ziegler E, et al. Incretin-responsive human pancreatic adipose tissue organoids: a functional model for fatty pancreas research. Mol Metab 2025; 91: 102067.
18.
Zhao Q, Yang Y, Hu J, Shan Z, Wu Y, Lei L. Exendin-4 enhances expression of neurod1 and glut2 in insulin-producing cells derived from mouse embryonic stem cells. Arch Med Sci 2016; 12: 199-207.
19.
Alhomoud IS, Talasaz AH, Chandrasekaran P, Brown R, Mehta A, Dixon DL. Incretin hormone agonists: current and emerging pharmacotherapy for obesity management. Pharmacotherapy 2024; 44: 738-52.
20.
Liu J, Hu Y, Zhang H, Xu Y, Wang G. Exenatide treatment increases serum irisin levels in patients with obesity and newly diagnosed type 2 diabetes. J Diabetes Complications 2016; 30: 1555-9.
21.
Nauck MA, Meier JJ. the incretin effect in healthy individuals and those with type 2 diabetes: physiology, pathophysiology, and response to therapeutic interventions. Lancet Diabetes Endocrinol 2016; 4: 525-36.
22.
Perakakis N, Triantafyllou GA, Fernández-Real JM, et al. Physiology and role of irisin in glucose homeostasis. Nat Rev Endocrinol 2017; 13: 324-37.
23.
Marrano N, Biondi G, Borrelli A, et al. Irisin and incretin hormones: similarities, differences, and implications in type 2 diabetes and obesity. Biomolecules 2021; 11: 286.
24.
Darimont C, Zbinden I, Avanti O, et al. Reconstitution of telomerase activity combined with HPV-E7 expression allow human preadipocytes to preserve their differentiation capacity after immortalization. Cell Death Differ 2003; 10: 1025-31.
25.
Gnaiger E, Renner-Sattler K. Phosphorylation Control Protocol with Intact Cells: ROUTINE, LEAK, ETS, ROX. Mitochondrial Physiol Network 2011: 1-14.
26.
Dunham-Snary KJ, Sandel MW, Westbrook DG, Ballinger SW. A method for assessing mitochondrial bioenergetics in whole white adipose tissues. Redox Biol 2014; 2: 656-60.
27.
Gnaiger E. Oroboros O2k-FluoRespirometer High-Resolution Respirometry O2k-Calibration by DatLab. Mitochondrial Physiol Network 2016; 19: 1-12.
28.
Gnaiger E. Bioenergetics at low oxygen: dependence of respiration and phosphorylation on oxygen and adenosine diphosphate supply. Respir Physiol 2001; 128: 277-97.
29.
Góralska J, Śliwa, A, Gruca, A, et al. Glucagon-like peptide-1 receptor agonist stimulates mitochondrial bioenergetics in human adipocytes. Acta Biochim Pol 2017; 64: 423-9.
30.
Zhang S, Wu X, Wang J, et al. Adiponectin/AdiopR1 signaling prevents mitochondrial dysfunction and oxidative injury after traumatic brain injury in a SIRT3 dependent manner. Redox Biol 2022; 54: 102390.
31.
Shackelford R, Hirsh S, Henry K, Abdel-Mageed A, Kandil E, Coppola D. Nicotinamide phosphoribosyltransferase and SIRT3 expression are increased in well-differentiated thyroid carcinomas. Anticancer Res 2013; 33: 3047-52.
32.
Simental-Mendía LE, Sánchez-García A, Linden-Torres E, Simental-Mendía M. Impact of glucagon-like peptide-1 receptor agonists on adiponectin concentrations: a meta-analysis of randomized controlled trials. Br J Clin Pharmacol 2021; 87: 4140-9.
33.
Kim Chung LT, Hosaka T, Yoshida M, et al. Exendin-4, a GLP-1 receptor agonist, directly induces adiponectin expression through protein kinase a pathway and prevents inflammatory adipokine expression. Biochem Biophys Res Commun 2009; 390: 613-8.
34.
Sommer G, Garten A, Petzold S, et al. Visfatin/PBEF/Nampt: structure, regulation and potential function of a novel adipokine. Clin Sci 2008; 115: 13-23.
35.
Liu R, Ding X, Wang Y, Wang M, Peng Y. Glucagon-like peptide-1 upregulates visfatin expression in 3T3-L1 adipocytes. Hormone Metab Res 2013; 45: 646-51.
36.
Lee J, Hong SW, Chae SW, et al. Exendin-4 improves steatohepatitis by increasing Sirt1 expression in high-fat diet-induced obese C57BL/6J mice. PLoS One 2012; 7: e31394.
37.
Yamaguchi S, Yoshino J. Adipose tissue NAD+ biology in obesity and insulin resistance: from mechanism to therapy. BioEssays 2017; 39: 10.1002/bies.201600227.
38.
Stromsdorfer KL, Yamaguchi S, Yoon MJ, et al. NAMPT-mediated NAD+ biosynthesis in adipocytes regulates adipose tissue function and multi-organ insulin sensitivity in mice. Cell Rep 2016; 16: 1851-60.
39.
Nagahisa T, Kosugi S, Yamaguchi S. Interactions between intestinal homeostasis and NAD+ biology in regulating incretin production and postprandial glucose metabolism. Nutrients 2023; 15: 1494.
40.
Sreedhar A, Zhao Y. Uncoupling protein 2 and metabolic diseases. Mitochondrion 2017; 34: 135-40.
41.
Moreno-Navarrete JM, Ortega F, Serrano M, et al. Irisin is expressed and produced by human muscle and adipose tissue in association with obesity and insulin resistance. J Clin Endocrinol Metab 2013; 98: E8769-78.
42.
Ge Y, Wu X, Cai Y, et al. FNDC5 prevents oxidative stress and neuronal apoptosis after traumatic brain injury through SIRT3-dependent regulation of mitochondrial quality control. Cell Death Dis 2024; 15: 364.
43.
Maak S, Norheim F, Drevon CA, Erickson HP. Progress and challenges in the biology of FNDC5 and irisin. Endocr Rev 2021; 42: 436-56.
44.
Wen P, Sun Z, Yang D, et al. Irisin regulates oxidative stress and mitochondrial dysfunction through the UCP2-AMPK pathway in prion diseases. Cell Death Dis 2025; 16: 66.
45.
Estell E, Ichikawa T, Giffault P, Bonewald L, Spiegelman B, Rosen C. Irisin enhances mitochondrial function in osteoclast progenitors during differentiation. Biomedicines 2023; 11: 3311.
46.
Jansen KM, Dahdah N, Gama-Perez P, Schots PC, Larsen TS, Garcia-Roves PM. Impact of GLP-1 receptor agonist versus omega-3 fatty acids supplement on obesity-induced alterations of mitochondrial respiration. Front Endocrinol (Lausanne) 2023; 14: 1098391.
47.
Bennett CF, Latorre-Muro P, Puigserver P. Mechanisms of mitochondrial respiratory adaptation. Nat Rev Mol Cell Biol 2022; 23: 817-35.
48.
Ejarque M, Guerrero-Pérez F, de la Morena N, et al. Role of adipose tissue GLP-1R expression in metabolic improvement after bariatric surgery in patients with type 2 diabetes. Sci Rep 2019; 9: 6274.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.