Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
HEMATOLOGY / BASIC RESEARCH
UBC9 silencing-mediated PPARa deSUMOylation induces inhibition of cell proliferation by ferroptosis in acute myeloid leukemia
1
Cancer center, Department of Hematology, Zhejiang Provincial People’s Hospital, Affiliated People’s Hospital, Hangzhou Medical College, Hangzhou, Zhejiang, China
Submission date: 2024-02-04
Final revision date: 2024-06-26
Acceptance date: 2025-04-22
Online publication date: 2025-06-08
Publication date: 2026-04-30
Corresponding author
Jianping Lan
Cancer Center Department of Hematology Zhejiang Provincial People’s Hospital Affiliated People’s Hospital Hangzhou Medical College 158 Shangtang Road Hangzhou, Zhejiang Province 310014, China Phone: 86-571-87666666
Cancer Center Department of Hematology Zhejiang Provincial People’s Hospital Affiliated People’s Hospital Hangzhou Medical College 158 Shangtang Road Hangzhou, Zhejiang Province 310014, China Phone: 86-571-87666666
Arch Med Sci 2026;22(2):1074-1083
KEYWORDS
TOPICS
ABSTRACT
Introduction:
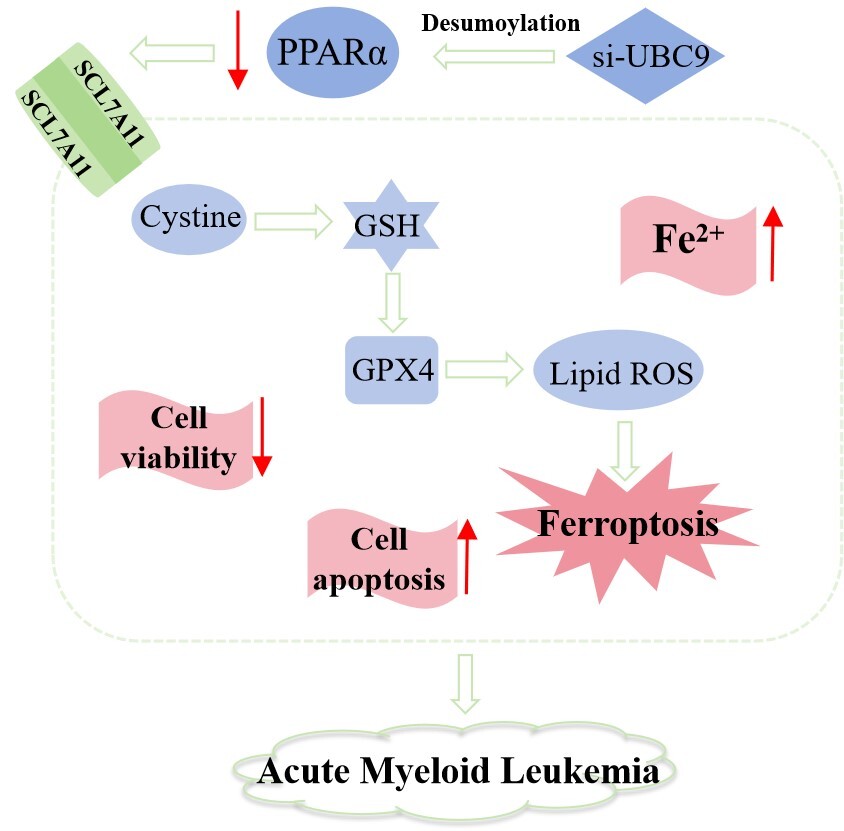
Inhibited acute myeloid leukemia (AML) proliferation is accompanied by downregulated peroxisome proliferator-activated receptor a (PPARa), which however can be stabilized via SUMOylation. This study investigated how PPARa SUMOylation impacts AML cell growth.
Material and methods:
Human AML HL-60 and Tohoku Hospital Pediatrics-1 (THP-1) cells were treated with the PPARa inhibitor GW6471 (10 µM) for 24 and 48 h. THP-1 cells were exposed to the PPARa agonist pirinixic acid (10 µM) following manipulation of the expression of the small ubiquitin-like modifier protein (SUMO)-conjugating enzyme UBC9. The interaction between PPARa and SUMO1 was detected by immunoprecipitation assay. HL-60 and THP-1 cell viability, apoptosis, and ferroptosis were measured via Cell Counting Kit-8 assay, flow cytometry, BODIPY-C11 staining and/or colorimetric assay. UBC9, glutathione peroxidase 4 (GPX4), recombinant solute carrier family 7, member 11(SLC7A11) and PPARa expression levels were analyzed by qRT-PCR or Western blot.
Results:
GW6471 treatment for 24 and 48 h suppressed viability, promoted apoptosis and lipid peroxidation, increased the level of Fe2+, and decreased the expression of GPX4, SLC7A11 and PPARa in HL-60/THP-1 cells. PPARa antibody induced enrichment of PPARa and SUMO1 in THP-1 cells, which was attenuated after UBC9 silencing. UBC9 silencing resulted in viability decrease, apoptosis and lipid peroxidation promotion, Fe2+ upregulation, and GPX4, SLC7A11, and PPARa downregulation in THP-1 cells, which were all counteracted by pirinixic acid.
Conclusions:
UBC9 silencing-induced PPARa deSUMOylation induces suppression of AML cell growth by ferroptosis.
Inhibited acute myeloid leukemia (AML) proliferation is accompanied by downregulated peroxisome proliferator-activated receptor a (PPARa), which however can be stabilized via SUMOylation. This study investigated how PPARa SUMOylation impacts AML cell growth.
Material and methods:
Human AML HL-60 and Tohoku Hospital Pediatrics-1 (THP-1) cells were treated with the PPARa inhibitor GW6471 (10 µM) for 24 and 48 h. THP-1 cells were exposed to the PPARa agonist pirinixic acid (10 µM) following manipulation of the expression of the small ubiquitin-like modifier protein (SUMO)-conjugating enzyme UBC9. The interaction between PPARa and SUMO1 was detected by immunoprecipitation assay. HL-60 and THP-1 cell viability, apoptosis, and ferroptosis were measured via Cell Counting Kit-8 assay, flow cytometry, BODIPY-C11 staining and/or colorimetric assay. UBC9, glutathione peroxidase 4 (GPX4), recombinant solute carrier family 7, member 11(SLC7A11) and PPARa expression levels were analyzed by qRT-PCR or Western blot.
Results:
GW6471 treatment for 24 and 48 h suppressed viability, promoted apoptosis and lipid peroxidation, increased the level of Fe2+, and decreased the expression of GPX4, SLC7A11 and PPARa in HL-60/THP-1 cells. PPARa antibody induced enrichment of PPARa and SUMO1 in THP-1 cells, which was attenuated after UBC9 silencing. UBC9 silencing resulted in viability decrease, apoptosis and lipid peroxidation promotion, Fe2+ upregulation, and GPX4, SLC7A11, and PPARa downregulation in THP-1 cells, which were all counteracted by pirinixic acid.
Conclusions:
UBC9 silencing-induced PPARa deSUMOylation induces suppression of AML cell growth by ferroptosis.
REFERENCES (44)
1.
Pelcovits A, Niroula R. Acute myeloid leukemia: a review. R I Med J (2013) 2020; 103: 38-40.
2.
Sasaki K, Ravandi F, Kadia T M, et al. De novo acute myeloid leukemia: a population-based study of outcome in the United States based on the Surveillance, Epidemiology, and End Results (SEER) database, 1980 to 2017. Cancer 2021; 127: 2049-61.
3.
Döhner H, Weisdorf DJ, Bloomfield CD. Acute myeloid leukemia. N Engl J Med 2015; 373: 1136-52.
4.
Bakhtiyari M, Liaghat M, Aziziyan F, et al. The role of bone marrow microenvironment (BMM) cells in acute myeloid leukemia (AML) progression: immune checkpoints, metabolic checkpoints, and signaling pathways. Cell Commun Signal 2023; 21: 252.
5.
Thol F, Ganser A. Treatment of relapsed acute myeloid leukemia. Curr Treat Options Oncol 2020; 21: 66.
6.
Xu T, Ding W, Ji X, et al. Molecular mechanisms of ferroptosis and its role in cancer therapy. J Cell Mol Med 2019; 23: 4900-12.
7.
Sun Y, Chen P, Zhai B, et al. The emerging role of ferroptosis in inflammation. Biomed Pharmacother 2020; 127: 110108.
8.
Gaschler MM, Andia AA, Liu H, et al. FINO2 initiates ferroptosis through GPX4 inactivation and iron oxidation. Nat Chem Biol 2018; 14: 507-15.
9.
Koppula P, Zhang Y, Zhuang L, et al. Amino acid transporter SLC7A11/xCT at the crossroads of regulating redox homeostasis and nutrient dependency of cancer. Cancer Commun 2018; 38: 12.
10.
Birsen R, Larrue C, Decroocq J, et al. APR-246 induces early cell death by ferroptosis in acute myeloid leukemia. Haematologica 2022; 107: 403-16.
11.
Yu Y, Xie Y, Cao L, et al. The ferroptosis inducer erastin enhances sensitivity of acute myeloid leukemia cells to chemotherapeutic agents. Mol Cell Oncol 2015; 2: e1054549.
12.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol 2021; 22: 266-82.
14.
Rezzola S, Sigmund EC, Halin C, et al. The lymphatic vasculature: an active and dynamic player in cancer progression. Med Res Rev 2022; 42: 576-614.
15.
Pawlak M, Lefebvre P, Staels B. Molecular mechanism of PPARa action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J Hepatol 2015; 62: 720-33.
16.
Lee JY, Nam M, Son HY, et al. Polyunsaturated fatty acid biosynthesis pathway determines ferroptosis sensitivity in gastric cancer. Proc Natl Acad Sci USA 2020; 117: 32433-42.
17.
Venkatesh D, O’Brien NA, Zandkarimi F, et al. MDM2 and MDMX promote ferroptosis by PPARa-mediated lipid remodeling. Genes Dev 2020; 34: 526-43.
18.
Han ZJ, Feng YH, Gu BH, et al. The post-translational modification, SUMOylation, and cancer (Review). Int J Oncol 2018; 52: 1081-94.
20.
Liu Y, Dou X, Zhou WY, et al. Hepatic small ubiquitin-related modifier (SUMO)-specific protease 2 controls systemic metabolism through SUMOylation-dependent regulation of liver-adipose tissue crosstalk. Hepatology 2021; 74: 1864-83.
21.
Zhang J, Huang FF, Wu DS, et al. SUMOylation of insulin-like growth factor 1 receptor, promotes proliferation in acute myeloid leukemia. Cancer Lett 2015; 357: 297-306.
22.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001; 25: 402-8.
23.
Vago L, Gojo I. Immune escape and immunotherapy of acute myeloid leukemia. J Clin Invest 2020; 130: 1552-64.
24.
Carter JL, Hege K, Yang J, et al. Targeting multiple signaling pathways: the new approach to acute myeloid leukemia therapy. Signal Transduct Target Ther 2020; 5: 288.
25.
Kayser S, Levis MJ. Advances in targeted therapy for acute myeloid leukaemia. Br J Haematol 2018; 180: 484-500.
26.
Zhang Y, Xing Z, Liu T, et al. Targeted therapy and drug resistance in thyroid cancer. Eur J Med Chem 2022; 238: 114500.
27.
Michalik L, Desvergne B, Wahli W. Peroxisome-proliferator-activated receptors and cancers: complex stories. Nat Rev Cancer 2004; 4: 61-70.
28.
Kersten S. Integrated physiology and systems biology of PPARa. Mol Metab 2014; 3: 354-71.
29.
Leng J, Li H, Niu Y, et al. Low-dose mono(2-ethylhexyl) phthalate promotes ovarian cancer development through PPARa-dependent PI3K/Akt/NF-kB pathway. Sci Total Environ 2021; 790: 147990.
30.
Luo Y, Xie C, Brocker CN, et al. Intestinal PPARa protects against colon carcinogenesis via regulation of methyltransferases DNMT1 and PRMT6. Gastroenterology 2019; 157: 744-59.e4.
31.
Luo X, Zhong L, Yu L, et al. TRIB3 destabilizes tumor suppressor PPARa expression through ubiquitin-mediated proteasome degradation in acute myeloid leukemia. Life Sci 2020; 257: 118021.
32.
Wang YX, Lee CH, Tiep S, et al. Peroxisome-proliferator-activated receptor delta activates fat metabolism to prevent obesity. Cell 2003; 113: 159-70.
33.
Kok T, Wolters H, Bloks VW, et al. Induction of hepatic ABC transporter expression is part of the PPARalpha-mediated fasting response in the mouse. Gastroenterology 2003; 124: 160-71.
34.
Kersten S. Peroxisome proliferator activated receptors and lipoprotein metabolism. PPAR Res 2008; 2008: 132960.
35.
Doll S, Proneth B, Tyurina YY, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat Chem Biol 2017; 13: 91-8.
36.
Sun N, Shen C, Zhang L, et al. Hepatic Krüppel-like factor 16 (KLF16) targets PPARa to improve steatohepatitis and insulin resistance. Gut 2021; 70: 2183-95.
37.
Xie Y, Hou W, Song X, et al. Ferroptosis: process and function. Cell Death Differ 2016; 23: 369-79.
38.
Maiorino M, Conrad M, Ursini F. GPx4, lipid peroxidation, and cell death: discoveries, rediscoveries, and open issues. Antioxid Redox Signal 2018; 29: 61-74.
39.
Wu X, Li Y, Zhang S, et al. Ferroptosis as a novel therapeutic target for cardiovascular disease. Theranostics 2021; 11: 3052-9.
40.
Liu DS, Duong CP, Haupt S, et al. Inhibiting the system x(C)(-)/glutathione axis selectively targets cancers with mutant-p53 accumulation. Nat Commun 2017; 8: 14844.
41.
Linkermann A, Stockwell BR, Krautwald S, et al. Regulated cell death and inflammation: an auto-amplification loop causes organ failure. Nat Rev Immunol 2014; 14: 759-67.
42.
Li Q, Su R, Bao X, et al. Glycyrrhetinic acid nanoparticles combined with ferrotherapy for improved cancer immunotherapy. Acta Biomater 2022; 144: 109-20.
43.
Du Y, Bao J, Zhang MJ, et al. Targeting ferroptosis contributes to ATPR-induced AML differentiation via ROS-autophagy-lysosomal pathway. Gene 2020; 755: 144889.
44.
Hendriks IA, Vertegaal AC. A comprehensive compilation of SUMO proteomics. Nat Rev Mol Cell Biol 2016; 17: 581-95.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.