Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
ONCOLOGY / BASIC RESEARCH
Evaluating the causal relationship between sex hormones and oral cavity cancer: a Mendelian randomization study
1
Department of Otolaryngology Head and Neck Surgery, Beijing Tiantan Hospital, Capital Medical University, Beijing, China
Submission date: 2025-05-12
Final revision date: 2025-06-09
Acceptance date: 2025-06-16
Online publication date: 2025-06-22
Publication date: 2026-04-30
Corresponding author
Lingzhao Meng
Department of Otolaryngology Head and Neck Surgery Beijing Tiantan Hospital Capital Medical University Beijing 100070, China Phone: +86- 01059976696
Department of Otolaryngology Head and Neck Surgery Beijing Tiantan Hospital Capital Medical University Beijing 100070, China Phone: +86- 01059976696
Arch Med Sci 2026;22(2):1022-1033
KEYWORDS
TOPICS
ABSTRACT
Introduction:
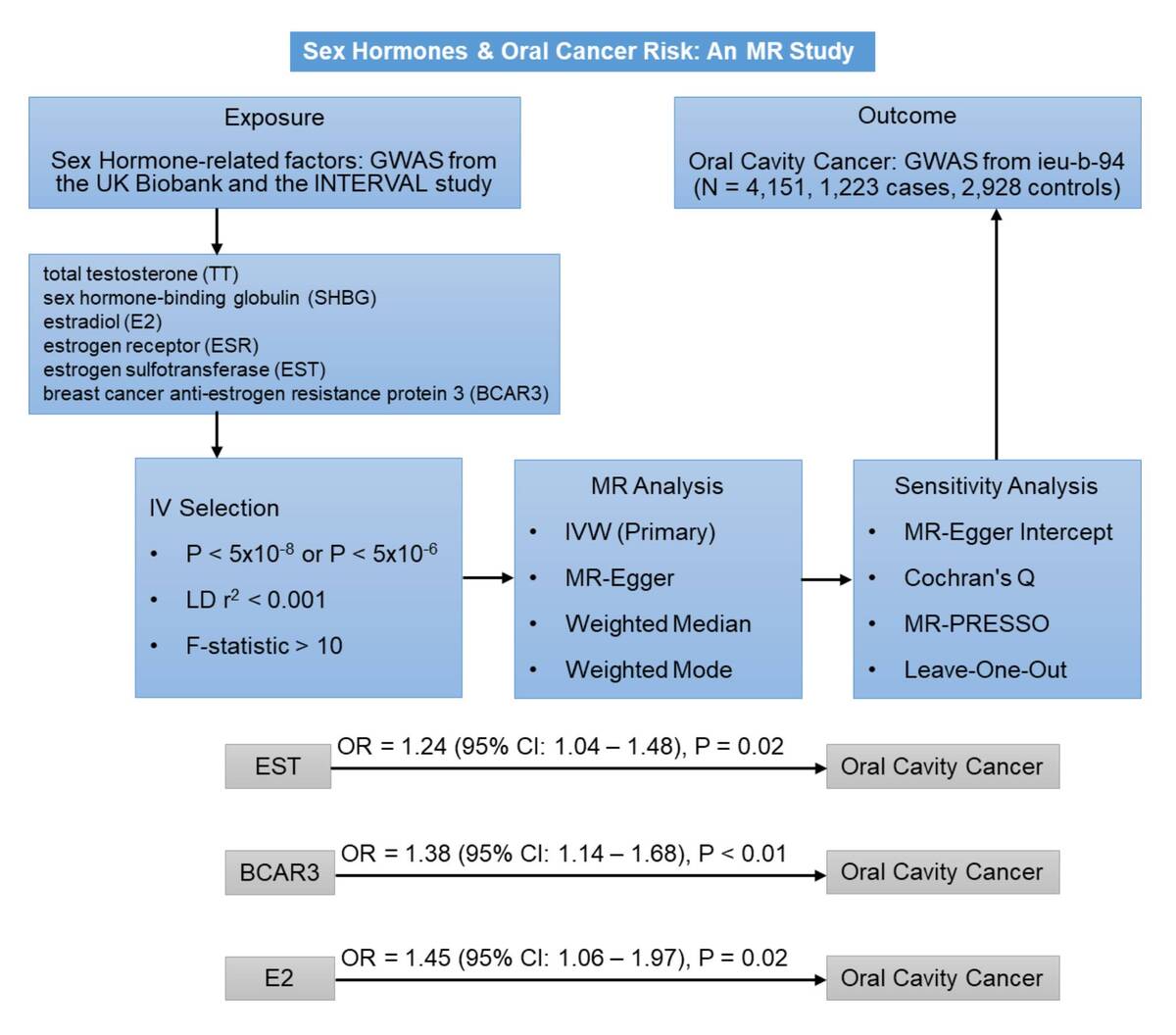
Observational studies on sex hormones and oral cavity cancer show inconsistent results. This two-sample Mendelian randomization (MR) study investigated potential causal associations between genetically predicted levels of six key sex hormone-related factors – total testosterone (TT), sex hormone-binding globulin (SHBG), estradiol (E2), estrogen receptor (ESR), estrogen sulfotransferase (EST), and breast cancer anti-estrogen resistance protein 3 (BCAR3) – and oral cavity cancer risk.
Material and methods:
Summary-level data from genome-wide association studies (GWAS) for exposures and oral cavity cancer, obtained via the IEU OpenGWAS, were analyzed using inverse variance weighted (IVW) as the primary method, complemented by weighted median, weighted mode, and MR-Egger regression. Sensitivity analyses included MR-Egger regression (directional pleiotropy), Cochran’s Q (heterogeneity), and MR-PRESSO (outlier detection/correction).
Results:
IVW analysis linked genetically predicted higher EST (OR = 1.24, 95% CI: 1.04–1.48, p = 0.02) and BCAR3 (OR = 1.38, 95% CI: 1.14–1.68, p < 0.01) to increased oral cavity cancer risk. After removing one outlier SNP, a significant association was also observed between E2 and oral cavity cancer (OR = 1.45, 95% CI: 1.06–1.97, p = 0.02). No significant relationships were found for TT, ESR, and SHBG. These findings were consistent across weighted median, weighted mode, and MR-Egger regression. MR-Egger regression did not indicate significant directional pleiotropy. For E2, initial heterogeneity was resolved after outlier correction, while EST and BCAR3 showed no significant heterogeneity.
Conclusions:
To our knowledge, this is the first comprehensive MR study linking sex hormone-related factors (E2, EST, BCAR3) to increased oral cavity cancer risk. Further validation is needed to explore prevention and treatment implications.
Observational studies on sex hormones and oral cavity cancer show inconsistent results. This two-sample Mendelian randomization (MR) study investigated potential causal associations between genetically predicted levels of six key sex hormone-related factors – total testosterone (TT), sex hormone-binding globulin (SHBG), estradiol (E2), estrogen receptor (ESR), estrogen sulfotransferase (EST), and breast cancer anti-estrogen resistance protein 3 (BCAR3) – and oral cavity cancer risk.
Material and methods:
Summary-level data from genome-wide association studies (GWAS) for exposures and oral cavity cancer, obtained via the IEU OpenGWAS, were analyzed using inverse variance weighted (IVW) as the primary method, complemented by weighted median, weighted mode, and MR-Egger regression. Sensitivity analyses included MR-Egger regression (directional pleiotropy), Cochran’s Q (heterogeneity), and MR-PRESSO (outlier detection/correction).
Results:
IVW analysis linked genetically predicted higher EST (OR = 1.24, 95% CI: 1.04–1.48, p = 0.02) and BCAR3 (OR = 1.38, 95% CI: 1.14–1.68, p < 0.01) to increased oral cavity cancer risk. After removing one outlier SNP, a significant association was also observed between E2 and oral cavity cancer (OR = 1.45, 95% CI: 1.06–1.97, p = 0.02). No significant relationships were found for TT, ESR, and SHBG. These findings were consistent across weighted median, weighted mode, and MR-Egger regression. MR-Egger regression did not indicate significant directional pleiotropy. For E2, initial heterogeneity was resolved after outlier correction, while EST and BCAR3 showed no significant heterogeneity.
Conclusions:
To our knowledge, this is the first comprehensive MR study linking sex hormone-related factors (E2, EST, BCAR3) to increased oral cavity cancer risk. Further validation is needed to explore prevention and treatment implications.
REFERENCES (49)
1.
Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021; 71: 209-49.
2.
Tan Y, Wang Z, Xu M, et al. Oral squamous cell carcinomas: state of the field and emerging directions. Int J Oral Sci 2023; 15: 44.
3.
Chen CJ, Sung WW, Lin YM, et al. Gender difference in the prognostic role of interleukin 6 in oral squamous cell carcinoma. PLoS One 2012; 7: e50104.
4.
Montero PH, Patel SG. Cancer of the oral cavity. Surg Oncol Clin N Am 2015; 24: 491-508.
5.
Rumgay H, Shield K, Charvat H, et al. Global burden of cancer in 2020 attributable to alcohol consumption: a population-based study. Lancet Oncol 2021; 22: 1071-80.
6.
Gupta I, Badrzadeh F, Tsentalovich Y, Gaykalova DA. Connecting the dots: investigating the link between environmental, genetic, and epigenetic influences in metabolomic alterations in oral squamous cell carcinoma. J Exp Clin Cancer Res 2024; 43: 239.
7.
Gaxiola-Rubio A, Jave-Suárez LF, Hernández-Silva CD, et al. The G-protein-coupled estrogen receptor agonist G-1 mediates antitumor effects by activating apoptosis pathways and regulating migration and invasion in cervical cancer cells. Cancers (Basel) 2024; 16: 3292.
8.
Lafront C, Germain L, Campolina-Silva GH, et al. The estrogen signaling pathway reprograms prostate cancer cell metabolism and supports proliferation and disease progression. J Clin Invest 2024; 134: e170809.
9.
Sabit H, Abouelnour S, Hassen BM, et al. Anticancer potential of prebiotics: targeting estrogen receptors and PI3K/AKT/mTOR in breast cancer. Biomedicines 2025; 13: 990.
10.
Thakare E, Chaudhary M, Gadbail A. A prospective study of circulating estrogen in oral leukoplakia and oral squamous cell carcinoma. J Cancer Res Ther 2024; 20: 1370-5.
11.
Colella G, Izzo G, Carinci F, et al. Expression of sexual hormones receptors in oral squamous cell carcinoma. Int J Immunopathol Pharmacol 2011; 24: 129-32.
12.
Aquino G, Collina F, Sabatino R, et al. Sex hormone receptors in benign and malignant salivary gland tumors: prognostic and predictive role. Int J Mol Sci 2018; 19: 399.
13.
Ojanotko-Harri A, Forssell H, Laine M, Hurttia H, Bläuer M, Tuohimaa P. Immunohistochemical detection of androgen receptors in human oral mucosa. Arch Oral Biol 1992; 37: 511-4.
14.
Liang G, Miao D, Du C. Causal associations between blood metabolites and breast cancer. Arch Med Sci 2025; 21: 206-14.
15.
Emdin CA, Khera AV, Kathiresan S. Mendelian randomization. JAMA 2017; 318: 1925-6.
16.
Sekula P, Del Greco MF, Pattaro C, Köttgen A. Mendelian randomization as an approach to assess causality using observational data. J Am Soc Nephrol 2016; 27: 3253-65.
17.
Birney E. Mendelian randomization. Cold Spring Harb Perspect Med 2022; 12: a041302.
18.
Burgess S, Davey Smith G, Davies NM, et al. Guidelines for performing Mendelian randomization investigations: update for summer 2023. Wellcome Open Res 2019; 4: 186.
19.
Skrivankova VW, Richmond RC, Woolf BAR, et al. Strengthening the reporting of observational studies in epidemiology using mendelian randomization: the STROBE-MR statement. JAMA 2021; 326: 1614-21.
20.
Lesseur C, Diergaarde B, Olshan AF, et al. Genome-wide association analyses identify new susceptibility loci for oral cavity and pharyngeal cancer. Nat Genet 2016; 48: 1544-50.
21.
Ruth KS, Day FR, Tyrrell J, et al. Using human genetics to understand the disease impacts of testosterone in men and women. Nat Med 2020; 26: 252-8.
22.
Schmitz D, Ek WE, Berggren E, Höglund J, Karlsson T, Johansson Å. Genome-wide association study of estradiol levels and the causal effect of estradiol on bone mineral density. J Clin Endocrinol Metab 2021; 106: e4471-86.
23.
Sun BB, Maranville JC, Peters JE, et al. Genomic atlas of the human plasma proteome. Nature 2018; 558: 73-9.
24.
Hemani G, Zheng J, Elsworth B, et al. The MR-Base platform supports systematic causal inference across the human phenome. Elife 2018; 7: e34408.
25.
Ference BA, Majeed F, Penumetcha R, Flack JM, Brook RD. Effect of naturally random allocation to lower low-density lipoprotein cholesterol on the risk of coronary heart disease mediated by polymorphisms in NPC1L1, HMGCR, or both: a 2 × 2 factorial Mendelian randomization study. J Am Coll Cardiol 2015; 65: 1552-61.
26.
Abecasis GR, Altshuler D, Auton A, et al. A map of human genome variation from population-scale sequencing. Nature 2010; 467: 1061-73.
27.
Wu L, Liao F, Guo X, Li N. The causal effect of adipose tissue on Hodgkin’s lymphoma: two-sample Mendelian randomization study and validation. Front Immunol 2024; 15: 1400756.
28.
Burgess S, Thompson SG. Avoiding bias from weak instruments in Mendelian randomization studies. Int J Epidemiol 2011; 40: 755-64.
29.
Slob EAW, Burgess S. A comparison of robust Mendelian randomization methods using summary data. Genet Epidemiol 2020; 44: 313-29.
30.
Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 2015; 44: 512-25.
31.
Hartwig FP, Davey Smith G, Bowden J. Robust inference in summary data Mendelian randomization via the zero modal pleiotropy assumption. Int J Epidemiol 2017; 46: 1985-98.
32.
Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol 2016; 40: 304-14.
33.
Greco MF, Minelli C, Sheehan NA, Thompson JR. Detecting pleiotropy in Mendelian randomisation studies with summary data and a continuous outcome. Stat Med 2015; 34: 2926-40.
34.
Burgess S, Thompson SG. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol 2017; 32: 377-89.
35.
Verbanck M, Chen CY, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet 2018; 50: 693-8.
36.
Burgess S, Bowden J, Fall T, Ingelsson E, Thompson SG. Sensitivity analyses for robust causal inference from mendelian randomization analyses with multiple genetic variants. Epidemiology 2017; 28: 30-42.
37.
Tran QK. Reciprocality between estrogen biology and calcium signaling in the cardiovascular system. Front Endocrinol 2020; 11: 568203.
38.
Ishida H, Wada K, Masuda T, et al. Critical role of estrogen receptor on anoikis and invasion of squamous cell carcinoma. Cancer Sci 2007; 98: 636-43.
39.
Kranjčević JK, Čonkaš J, Ozretić P. The role of estrogen and estrogen receptors in head and neck tumors. Cancers (Basel) 2024; 16: 1575.
41.
Diaz-Ruano AB, Martinez-Alarcon N, Perán M, et al. Estradiol and estrone have different biological functions to induce NF-kB-driven inflammation, EMT and stemness in ER+ cancer cells. Int J Mol Sci 2023; 24: 1221.
42.
Yi M, Negishi M, Lee SJ. Estrogen sulfotransferase (SULT1E1): its molecular regulation, polymorphisms, and clinical perspectives. J Pers Med 2021; 11: 194.
43.
Wang J, Feng Y, Liu B, Xie W. Estrogen sulfotransferase and sulfatase in steroid homeostasis, metabolic disease, and cancer. Steroids 2024; 201: 109335.
44.
Hirata H, Hinoda Y, Okayama N, et al. CYP1A1, SULT1A1, and SULT1E1 polymorphisms are risk factors for endometrial cancer susceptibility. Cancer 2008; 112: 1964-73.
45.
Choi JY, Lee KM, Park SK, et al. Genetic polymorphisms of SULT1A1 and SULT1E1 and the risk and survival of breast cancer. Cancer Epidemiol Biomarkers Prev 2005; 14: 1090-5.
46.
Cross AM, Wilson AL, Guerrero MS, et al. Breast cancer antiestrogen resistance 3-p130(Cas) interactions promote adhesion disassembly and invasion in breast cancer cells. Oncogene 2016; 35: 5850-9.
47.
Moon DO. Deciphering the role of BCAR3 in cancer progression: gene regulation, signal transduction, and therapeutic implications. Cancers (Basel) 2024; 16: 1674.
48.
Zhang Z, Wang Y, Wang Y, et al. BCAR3 promotes head and neck cancer growth and is associated with poor prognosis. Cell Death Discov 2021; 7: 316.
49.
Mungenast F, Aust S, Vergote I, et al. Clinical significance of the estrogen-modifying enzymes steroid sulfatase and estrogen sulfotransferase in epithelial ovarian cancer. Oncol Lett 2017; 13: 4047-54.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.