Current issue
Archive
Manuscripts accepted
About the Journal
Editorial office
Editorial board
Section Editors
Abstracting and indexing
Subscription
Contact
Ethical standards and procedures
Most read articles
Instructions for authors
Article Processing Charge (APC)
Regulations of paying article processing charge (APC)
DIABETOLOGY / CLINICAL RESEARCH
The effect of beinaglutide on visceral fat and body weight in obese type 2 diabetic patients
1
Department of Endocrinology and Metabolism, The First Affiliated Hospital of Datong University, Datong, Shanxi, China
Submission date: 2020-06-22
Final revision date: 2020-07-16
Acceptance date: 2020-07-21
Online publication date: 2020-08-18
Publication date: 2026-04-30
Corresponding author
Yin-Bing Wang
Department of Endocrinology and Metabolism The First Affiliated Hospital of Datong University Datong, Shanxi, China 30 Xinhua St Pingcheng District Datong city Shanxi 201321, China. Phone: +86 0352-7122337
Department of Endocrinology and Metabolism The First Affiliated Hospital of Datong University Datong, Shanxi, China 30 Xinhua St Pingcheng District Datong city Shanxi 201321, China. Phone: +86 0352-7122337
Arch Med Sci 2026;22(2):702-707
KEYWORDS
TOPICS
ABSTRACT
Introduction:
Glucagon-like peptide-1 (GLP-1) analogues could induce clinically significant weight loss in obese patients with type 2 diabetes mellitus (T2DM). Beinaglutide is a GLP-1 analogue that is fully homologous to human GLP-1. This study aims to investigate the clinical efficacy of beinaglutide in visceral fat, weight loss and blood glucose in patients with T2DM and obesity.
Material and methods:
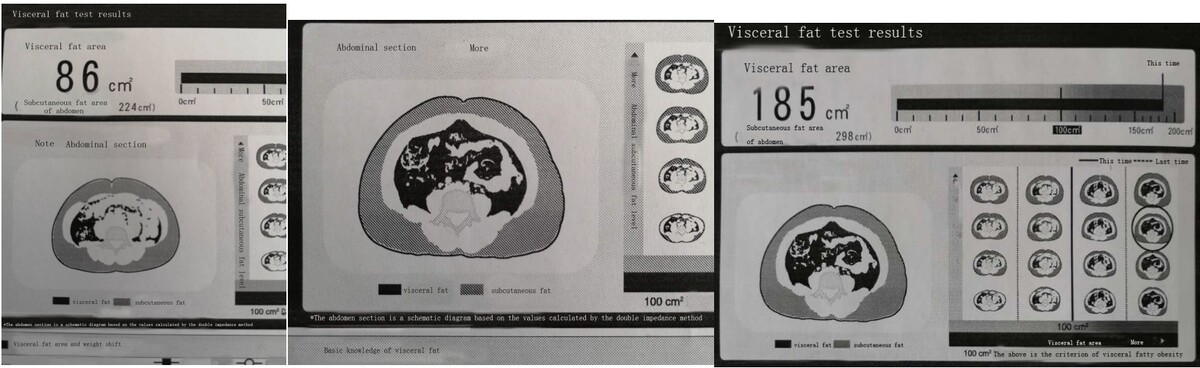
One hundred and seven obese patients with T2DM, according to the World Health Organisation’s (WHO) diagnostic criteria, were treated with beinaglutide. After 12 weeks, changes in visceral fat (VF) were analysed using DUALSCAN. Body weight, body mass index (BMI), glycated haemoglobin (HbA1c), free fatty acids, and blood pressure were also assessed after the 12-week treatment.
Results:
The baselines for BMI and VF areas were 32.8 ±5.2 kg/m2 and 150.4 ±36.2 cm2, respectively. The mean HbA1c level at baseline was 8.8 ±2.3%. After 12 weeks, beinaglutide treatment showed significant decreases in VF areas (150.4 ±36.2 cm2 vs. 115.2 ±36.8 cm2, p < 0.001), body weight (90.7 ±15.8 kg vs. 84.2 ±15.6 kg, p < 0.001), HbA1c (8.8 ±2.3% vs. 7.1 ±1.7%, p < 0.001) and insulin resistance index (HOMA-IR) (5.8 ±4.3 vs. 4.2 ±2.9, p < 0.001). No changes in free fatty acids were observed. Daily doses of beinaglutide varied widely, and 74% of patients ranged from 0.24 mg to 0.30 mg each day, but the appropriate dosage significantly reduced adverse effects.
Conclusions:
Beinaglutide effectively reduced VF, body weight, and blood glucose in obese patients with T2DM. Beinaglutide doses should be individualised because appropriate doses varied widely. A lack of GLP-1 might be responsible for the onset of obesity in patients with T2DM. T2DM and obesity are related not only to insulin deficiency/insulin resistance but also to GLP-1 deficiency, which may cause obesity in patients with T2DM.
Glucagon-like peptide-1 (GLP-1) analogues could induce clinically significant weight loss in obese patients with type 2 diabetes mellitus (T2DM). Beinaglutide is a GLP-1 analogue that is fully homologous to human GLP-1. This study aims to investigate the clinical efficacy of beinaglutide in visceral fat, weight loss and blood glucose in patients with T2DM and obesity.
Material and methods:
One hundred and seven obese patients with T2DM, according to the World Health Organisation’s (WHO) diagnostic criteria, were treated with beinaglutide. After 12 weeks, changes in visceral fat (VF) were analysed using DUALSCAN. Body weight, body mass index (BMI), glycated haemoglobin (HbA1c), free fatty acids, and blood pressure were also assessed after the 12-week treatment.
Results:
The baselines for BMI and VF areas were 32.8 ±5.2 kg/m2 and 150.4 ±36.2 cm2, respectively. The mean HbA1c level at baseline was 8.8 ±2.3%. After 12 weeks, beinaglutide treatment showed significant decreases in VF areas (150.4 ±36.2 cm2 vs. 115.2 ±36.8 cm2, p < 0.001), body weight (90.7 ±15.8 kg vs. 84.2 ±15.6 kg, p < 0.001), HbA1c (8.8 ±2.3% vs. 7.1 ±1.7%, p < 0.001) and insulin resistance index (HOMA-IR) (5.8 ±4.3 vs. 4.2 ±2.9, p < 0.001). No changes in free fatty acids were observed. Daily doses of beinaglutide varied widely, and 74% of patients ranged from 0.24 mg to 0.30 mg each day, but the appropriate dosage significantly reduced adverse effects.
Conclusions:
Beinaglutide effectively reduced VF, body weight, and blood glucose in obese patients with T2DM. Beinaglutide doses should be individualised because appropriate doses varied widely. A lack of GLP-1 might be responsible for the onset of obesity in patients with T2DM. T2DM and obesity are related not only to insulin deficiency/insulin resistance but also to GLP-1 deficiency, which may cause obesity in patients with T2DM.
REFERENCES (36)
1.
Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract 2019; 157: 107843.
2.
Wang L, Gao P, Zhang M, et al. Prevalence and ethnic pattern of diabetes and prediabetes in China in 2013. JAMA 2017; 24: 2515-23.
3.
Wharton S, Pedersen SD, Lau DCW, Sharma AM. Weight management in diabetes. Can J Diabetes 2018; 42 Suppl 1: S124-9.
4.
Verkouter I, Noordam R, de Roos A, et al. Adult weight change in relation to visceral fat and liver fat at middle age: the Netherlands epidemiology of obesity study. Int J Obesity 2019; 4: 790-9.
5.
Haberka M, Stolarz-Skrzypek K, Biedron M, et al. Obesity, visceral fat, and hypertension-related complications. Metab Syndr Relat Disord 2018; 10: 521-9.
6.
Barkai L, Kiss Z, Rokszin G, et al. Changes in the incidence and prevalence of type 1 and type 2 diabetes among 2 million children and adolescents in Hungary between 2001 and 2016 – a nationwide population-based study. Arch Med Sci 2020; 16: 1734-41.
7.
Chen GC, Arthur R, Iyengar NM, et al. Association between regional body fat and cardiovascular disease risk among postmenopausal women with normal body mass index. Eur Heart J 2019; 40: 2849-55.
8.
Rubenstein AH. Obesity: a modern epidemic. Trans Am Clin Climatol Assoc 2005; 116: 103-13.
9.
Sironi AM, Petz R, de Marchi D, et al. Impact of increased visceral and cardiac fat on cardiometabolic risk and disease. Diabet Med 2012; 29: 622-7.
10.
Hotamisligil GS. Molecular mechanisms of insulin resistance and the role of the adipocyte. Int J Obesity 2000; 24 Suppl 4: S23-7.
11.
Hajer GR, van Haeften TW, Visseren FLJ. Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur Heart J 2008; 29: 2959-71.
12.
Mahabadi AA, Massaro JM, Rosito GA, et al. Association of pericardial fat, intrathoracic fat, and visceral abdominal fat with cardiovascular disease burden: the Framingham Heart Study. Eur Heart J 2009; 30: 850-6.
13.
Sandeep S, Gokulakrishnan K, Velmurugan K, Deepa M, Mohan V. Visceral and subcutaneous abdominal fat in relation to insulin resistance and metabolic syndrome in non-diabetic south Indians. Indian J Med Res 2010; 131: 629-35.
14.
Fain JN, Buehrer B, Bahouth SW, et al. Comparison of messenger RNA distribution for 60 proteins in fat cells vs the nonfat cells of human omental adipose tissue. Metabolism 2008; 57: 1005-15.
15.
Biessels GJ, Staekenborg S, Brunner E, Brayne C, Scheltens P. Risk of dementia in diabetes mellitus: a systematic review. Lancet Neurol 2006; 5: 64-74.
16.
Yoon S, Cho H, Kim J, et al. Brain changes in overweight/obese and normal-weight adults with type 2 diabetes mellitus. Diabetologia 2017; 60: 1207-17.
17.
Franz MJ, Boucher JL, Rutten-Ramos S, VanWormer JJ. Lifestyle weight-loss intervention outcomes in overweight and obese adults with type 2 diabetes: a systematic review and meta-analysis of randomized clinical trials. J Acad Nutr Diet 2015; 115: 1447-63.
18.
Baum A, Scarpa J, Bruzelius E, Tamler R, Basu S, Faghmous J. Targeting weight loss interventions to reduce cardiovascular complications of type 2 diabetes: a machine learning-based post-hoc analysis of heterogeneous treatment effects in the Look AHEAD trial. Lancet Diabetes Endocrinol 2017; 5: 808-15.
19.
Taylor R, Al-Mrabeh A, Sattar N. Understanding the mechanisms of reversal of type 2 diabetes. Lancet Diabetes Endocrinol 2019; 7: 726-36.
20.
Cheung BMY, Cheung TT, Samaranayake NR. Safety of antiobesity drugs. Ther Adv Drug Saf 2013; 4: 171-81.
21.
Healy P, Clarke C, Reynolds I, Arumugasamy M, McNamara D. Complications of bariatric surgery: what the general surgeon needs to know. Surgeon 2016; 14: 91-8.
22.
Contival N, Menahem B, Gautier T, Le Roux Y, Alves A. Guiding the non-bariatric surgeon through complications of bariatric surgery. J Visceral Surg 2018; 155: 27-40.
23.
Wysham C, Blevins T, Arakaki R, et al. Efficacy and safety of dulaglutide added onto pioglitazone and metformin versus exenatide in type 2 diabetes in a randomized controlled trial (AWARD-1). Diabetes Care 2014; 37: 2159.
24.
Davies MJ, Bergenstal R, Bode B, et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: the SCALE diabetes randomized clinical trial. JAMA 2015; 314: 687-99.
25.
Pi-Sunyer X, Astrup A, Fujioka K, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med 2015; 373: 11-22.
26.
Zhang YL, Zhou C, Li XF, et al. Beinaglutide showed significant weight-loss benefit and effective glycaemic control for the treatment of type 2 diabetes in a real-world setting: a 3-month, multicentre, observational, retrospective, open-label study. J Clin Investig 2017; 127: 4217-27.
27.
Pietiläinen KH, Kaye S, Karmi A, Suojanen L, Rissanen A, Virtanen KA. Agreement of bioelectrical impedance with dual-energy X-ray absorptiometry and MRI to estimate changes in body fat, skeletal muscle and visceral fat during a 12-month weight loss intervention. Br J Nutr 2013; 109: 1910-6.
28.
Lim GE, Brubaker PL. Glucagon-like peptide 1 secretion by the l-cell. Diabetes 2006; Suppl 2: S70.
29.
Mayo KE, Miller LJ, Bataille D, et al. International Union of Pharmacology. XXXV. The Glucagon Receptor Family. Pharmacol Rev 2003; 55: 167-94.
30.
Ma CC, Duan CC, Huang RC, Tang HQ. Association of circulating cystatin C levels with type 2 diabetes mellitus: a systematic review and meta-analysis. Arch Med Sci 2020; 16: 648-56.
31.
Shek AB, Alieva RB, Kurbanov RD, Hoshimov SU, Nizamov UI, Ziyaeva AV. Can metformin stabilize PCSK9 level in stable coronary artery disease patients treated with statins? Arch Med Sci Atheroscler Dis 2019; 4: e144-50.
32.
Drucker DJ, Habener JF, Holst JJ. Discovery, characterization, and clinical development of the glucagon-like peptides. J Clin Investig 2017; 127: 4217-27.
33.
Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev 2007; 87: 1409-39.
34.
Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology 2007; 132: 2131-57.
35.
Lockie SH, Heppner KM, Chaudhary N, et al. Direct control of brown adipose tissue thermogenesis by central nervous system glucagon-like peptide-1 receptor signaling. Diabetes 2012; 61: 2753-62.
36.
Ritzel R, Orskov C, Holst JJ, Nauck MA. Pharmacokinetic, insulinotropic, and glucagonostatic properties of GLP-1 [7-36 amide] after subcutaneous injection in healthy volunteers. Dose-response-relationships. Diabetologia 1995; 38: 720-5.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.